ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2008; 4(1):48-57. doi:10.7150/ijbs.4.48 This issue Cite
Research Paper
ParaHox genes in pancreatic cell cultures: effects on the insulin promoter regulation
Departament de Genètica, Facultat de Biologia, Universitat de Barcelona, Barcelona, Spain
* Present address: Barcelona Center for International Health and Research(CRESIB), Laboratorio de Malaria (311 IDIBAPS), Facultat Medicina, Hospital Clinic, Barcelona, Spain
Abstract
The gene encoding PDX1 (pancreatic duodenum homeobox 1), the main transcription factor regulating the glucose-dependent transactivation of the insulin promoter in pancreatic β-cells, clusters with two closely related homeobox genes (Gsh1 and Cdx2/3), all of them belonging to the ParaHox gene family. The ParaHox gene evolutionary history in the vertebrate lineage involved duplications of the cluster and subsequent loss of some members, so that eventually, the human and murine genomes contain only 6 ParaHox genes. The crucial role of PDX1 in pancreas development, beta-cell formation and insulin transcription regulation has long been established. There is some data on CDX2/3 function in α-cells, but remarkably, nothing is known on the role of the other ParaHox genes, which are also expressed in the endocrine pancreas. Homeobox transcription factors that belong to the same family show high conservation of the homeodomain and share similar target sites and oligomeric partners, and thus may act redundantly, synergistically or antagonistically on the same promoters. Therefore, we explored the effects of the Parahox proteins (GSH1, GSH2, CDX1, CDX2/3 and CDX4) on the regulation of the insulin promoter in transfected α- and β- cultured cell lines at different glucose concentrations and compared them to those of PDX1. Noticeably, several ParaHox transcription factors are able to transactivate or inhibit the insulin promoter, depending on the cell type and glucose concentration, thus suggesting their possible participation in the regulation of similar target genes, such as insulin, either by silencing or activating them, in the absence of PDX1.
Keywords: ParaHox genes, endocrine pancreas, α-cells, β-cells, insulin promoter, Pdx1
1. INTRODUCTION
PDX1 is a crucial pancreatic transcription factor. It functions both in the early commitment of the primitive gut to pancreatic fate and in the later maturation of β-cells. In the adult, PDX1 is mainly expressed in β-cells where it is a major transactivator of the insulin gene (reviewed in [1]). Notably, heterozygous mutations of the PDX1 gene are linked to a type of autosomal dominant diabetes mellitus known as maturity onset diabetes of the young (MODY4) [2]. Finely tuned regulation of the insulin gene transcription relies on the recognition of several promoter sequence motifs by a combination of ubiquitous and islet-specific transcriptions factors [3], whose characterization would contribute to our current understanding not only of insulin gene expression, but also of endocrine pancreas development and function (reviewed in [1, 4]). Several of these key cis-acting enhancer elements show a considerably degree of conservation among vertebrate species, suggesting that the regulatory mechanisms controlling insulin expression are evolutionarily conserved [5]. Among them, several key motifs are recognised and bound by homeodomain-containing proteins [6], the most relevant being the pancreatic duodenal homeobox-1 (PDX1) transcription factor. Similarly to other homeodomain transcription factors, PDX1 form oligomers with additional homeodomain proteins and trasncriptional regulators [7], acting in a synergistic fashion. Hence, the net activity of the promoter is subtly tuned by the concentration and function of any of the proteins in the complex, as well as by glucose, which regulates insulin gene transcription through multiple effects on several of the proteins in the transcription activation complex [8-13]. Although Pdx1 is pivotal for pancreas formation and insulin regulation, the knockout mouse for Pdx1 still maintains some insulin-producing cells, thus pointing to a degree of flexibility on the formation of the promoter complex, where other transcription factors, most probably with an homeodomain, could substitute its function (reviewed in [14]).
The Pdx1 gene belongs to the ParaHox gene cluster, an array of genes containing a Hox-like homeobox. This cluster includes Gsh and Cdx genes in addition to Pdx1. The ParaHox cluster is an ancient evolutionary sister group of the well-known Hox cluster [15]. These two gene complexes suffered several rounds of large genome duplications in the vertebrate lineage that eventually produced, in the case of the ParaHox genes in mammals, four syntenic segments. Currently, only one cluster remains fully intact [16], most probably because these clustered genes share common or interdigitated control regions [17]. Although the HOX cluster has been a long-standing paradigm, there is still controversy regarding the conserved structure of the clusters and the intriguing fact of the spatio-temporal colinearity in their expression patterns (reviewed in [15]). There is a wealth of information on the transcriptional targets of most HOX genes, although so far, comprehensive networks that encompass the HOX, the hundreds of potential target genes, and their interrelations is still lacking [18, 19]. Much less is known on the polemic ParaHox cluster, which has been mostly overlooked. This scarcity of data is probably due to several reasons: i) the ParaHox gene number is smaller, ii) only one of the clusters remained in mammals, iii) the apparent lack of colinearity in their expression, and iv) the prominent role of PDX1 in pancreas organogenesis and function has diverted most of the attention to function instead than organization.
All ParaHox genes belong to the same family and share a very similar homeodomain, hence they may bind related or even identical target sites, as it is the case with other homeodomain trancription factors. Then, the specificity and the affinity of the binding to the promoter, as well as the effect (inhibition of transactivation) on transcription, would depend on the interacting partners (reviewed in [20]). Thus, conceivably, other ParaHox proteins could bind to Pdx1 targets in vivo, given that adequate partners are present. This has not been, to our knowledge, explored elsewhere.
Besides Pdx1, not much data has been gathered on Parahox expression in pancreas. CDX2/3 is expressed in α-cells, where it binds to the glucagon promoter and activates its transcription [21]. In addition, and although this factor is not β cell-specific, CDX2/3 can recognise enhacer elements of the rat insulin I gene promoter usually bound by PDX1 (German et al., 1992) and transactivate endogenous and recombinant rat insulin I and II promoters [22, 23]. Moreover, most studies on Cdx and Gsh transcription factors focus on their roles during embryonic development and tumorigenesis, and very scarce data is available on their biological function in adult tissues. We have recently shown that all ParaHox genes are expressed in the murine endocrine pancreas, in particular within the glucagon-secreting α-cells, except Pdx1, which is mainly detected in β- and δ-cells [24]. These results prompted us to further evaluate whether the ParaHox transcription factors might be involved in the regulation of similar target genes.
Given that: i) all the ParaHox genes are evolutionary related, ii) share similar homeodomains and thus, may bind to similar target motifs; iii) some of the ParaHox genes are linked to Pdx1, retaining the original genomic clustering and potentially sharing common regulatory elements and, iv) their recently reported expression in endocrine pancreas, we aimed to evaluate the role of the ParaHox proteins as potential transcriptional regulators of the insulin gene, as proof of principle. To this end, we analysed the effect of Parahox gene overexpression on the transactivation of a luciferase reporter gene driven by the –600INS mouse promoter region in transiently transfected α- and β- pancreatic-derived mouse cells (αTC1 and βTC6).
Our results show that evolutionarily related homeodomain transcription factors can share the same target and provide unexpected insights into the regulation of the insulin promoter by homeodomain proteins other than PDX1, which are also expressed in the endocrine pancreas. We surmise that this type of results may be extrapolated to other homeodomain transcription factors and target genes.
2. MATERIALS AND METHODS
Cell culture
The mouse βTC6 and αTC1 cell lines were obtained from ATCC (references CRL-11506 and CRL-2350, respectively), and all experiments were performed with cells between passage numbers 1 to 20. Cells were maintained following the manufacturer's recommendations. βTC6 cells were cultured in Dulbecco's Modified Eagle's Medium with 4 mM L-glutamine, 1.5 g/l sodium bicarbonate, supplemented with 15% heat-inactivated fetal bovine serum, penicillin and streptomycin. αTC1 cells were cultured in Dulbecco's Modified Eagle's Medium with 1.5 g/l sodium bicarbonate and supplemented with 15 mM HEPES, 0.1 mM non-essential amino acids, 0.02 % bovine albumin serum (BSA) and 10 % heat-inactivated fetal bovine serum. Two different glucose concentrations were used: 4.5 g/l (25mM) and 0.1 g/l (0.5 mM) depending on whether high or low glucose concentration was required for each experiment.
DNA constructs
It is well known that mRNA stability, nucleus/cytoplasma transport and even, mRNA translation efficiency, are related to mRNA maturation processes, in particular to the splicing events [25, 26]. Accordingly, many expression vectors designed to improve protein production in cultured cells include introns in the 5' or 3' UTRs. Alternatively, the expression of coding sequences derived from genomic amplification (so that introns are embedded in their original location) warrants the appropiate processing by the cellular splicing machinery. Considering all this information, we decided to use several strategies in order to include at least one intron (either the original or synthetic) in the expression constructs.
The final expression vectors were constructed as follows. The PDX1 and CDX2/3 vectors were generated by insertion of the full-length genomic sequences from the ATG to the STOP codons (including the corresponding genomic introns) into the pcDNA3.1 vector (Invitrogen). The expression vector encoding CDX4 was generated by insertion of the full-length coding sequence plus genomic intron 2 to the pcDNA3 vector. To this end, we used mouse genomic DNA as a template to amplify the first exon (from the initial ATG to the end of the first exon) and fused it to a second amplification product that stretched from the beginning of the second exon, including intron 2, until the end of the third exon. The strategy to clone the coding sequences of CDX1, GSH1 and GSH2 was as follows: we amplified each single coding exon, and subsequently, fused them in-frame; the reconstituted full-length cDNA was cloned into pcDNA3 and a synthetic intron from pIRESneo (BD Biosciences) was additionally cloned after the STOP codon at the 3' UTR of the expression vector.
A plasmid containing a –645 bp fragment of the mouse Insulin II promoter was cloned into the pGL3 basic vector (Promega) upstream of the luciferase reporter gene, using standard cloning methods. This promoter segment contains all the necessary and sufficient cis-regulatory elements required for the correct transcriptional regulation of the insulin gene in β-cells [6, 27].
All the constructs were verified by DNA sequencing using the ABI3700 sequencer (AP Biosystems).
Transfections
Cells grown at about 80% confluence in twelve-well plates were transfected using 6 μl of Lipofectamine 2000 transfection reagent (Invitrogen Life Technologies) following the manufacturer's recommendations with a total of 2.5 μg of DNA, in serum-free Optimem medium (Gibco, Invitrogen Corporation). For standard transfections, and unless otherwise stated, cells were harvested 48 h after transfection.
In all transfections, the quantity of the mouse insulin II promoter –645INS-LUC reporter construct was maintained constant at 0.5 μg. However, to analyse the effect of the overexpression of the ParaHox transcription factors, the expression constructs were cotransfected in increasing quantities: 0.25 μg, 0.5 μg, 0.8 μg, 1.2 μg, 1.5 μg, 1.8 μg and 2 μg for the βTC6 cell line and at 0.25 μg, 0.5 μg, 0.8 μg, 1.5 μg and 2 μg for the αTC1 cell line. The pSV-βGal vector (0.2 μg) was also cotransfected to normalise the transfection efficiency. For each sample, at least four independently replicates were transfected, and duplicate values for each replica were obtained. Cells transfected with the –645INS-LUC vector but no transcription factor construct were used as positive controls and the value obtained was considered as the reference unit. Cells transfected with empty vectors were considered as negative controls. The β-galactosidase and luciferase activities were quantified using the ß-Gal Reporter Gene Assay chemiluminescent (Roche) and the Luciferase Assay Systems E1500 (Promega) kits, respectively, following the manufacturer's recommendations. Each luminescence value, the ratio, mean average value and the standard deviation were obtained using the FB12 luminometer (Berthold Detection Systems) and the FB12 Sirius SW software (Berthold Detection Systems). Statistical significance was assessed by the Mann-Whitney test (p<0.05).
cDNA obtention and RT-PCR
Cells were seeded in 12-well plates, transfected with 0.8 μg of each ParaHox construct as detailed above and grown on high glucose (25 mM) supplemented media. Cells (at least three replicates for each construct were pooled together) were harvested 48 hours post-transfection using the “Cells-to-cDNA II” kit (Ambion) following the manufacturer's instructions. For each ParaHox transcript, a different primer pair spanning different exons was used to perform RT-PCR. A housekeeping gene, GAPDH, was also amplified to allow normalisation and comparison between samples. Primers and PCR conditions are detailed in [24], and are available upon request.
3. RESULTS
Constructs controls to assess the effect of ParaHox gene overexpression on the insulin promoter transactivation
The construction of the vectors to allow constitutive expression of GSH1, GSH2, PDX1, CDX1, CDX2/3 and CDX4 in transiently transfected eukaryotic cells is detailed in the Material and Methods section. In brief, the coding sequence of each Parahox gene was amplified from mouse genomic DNA, from the ATG to the STOP codons and cloned into pcDNA3.1. In all the cases, at least one intron (either genomic or synthetic) was included in the expression constructs. The integrity of the final sequence was confirmed by sequencing prior to transfection of murine α- and β- pancreatic cells.
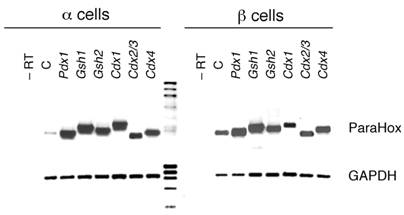
After transfection, the expression levels of each Parahox construct were verified by RT-PCR. Cells from at least three replicates for each construct and cell line were pooled and, total RNA was obtained to synthesise the first strand cDNA with a mixture of poly-d(T) and random hexamers. Subsequently, the cDNA from each Parahox was amplified with specific primers from sequences of different exons (Fig. 1). GAPDH was used as an internal control. As expected, no expression was detected in the reverse transcription negative control (–RT lane, Fig.1), whereas we amplified the endogenously expressed Pdx1 in β-cells transfected with empty-vector (positive control, C lane in Fig.1). Much lower, although detectable, levels of Pdx1 were also observed in αTC1 cells, in agreement with microarray expression reports comparing β- and α-derived cells [28].
Expression of the ParaHox constructs in αTC1 and βTC6 cell lines. Cells were transfected with 0.8 μg of the construct for each transcription factor, allowed to grow in high glucose media (25mM glucose) and harvested 48 hours post-transfections. Cells were harvested from at least three replicates and two different experiments for each construct, and pooled to obtain the first strand cDNA. Specific primers and PCR conditions allowed the detection of each ParaHox gene expression. GAPDH was used as an internal control. –RT: transfected cells with the Pdx1 transcription factor, but in which no reverse transcription was performed (negative control of the RT-PCR, as there is no amplification if no cDNA is previously obtained). C: empty vector transfected cells, which also serve as positive control of the RT-PCR, as Pdx1 is endogenously expressed (at low levels in αTC1 cells and at much higher levels in βTC6 cells, as expected).
To assess the effect of the overexpression of the ParaHox genes on the insulin promoter activity, we relied on the highly sensitive luciferase reporter gene. The proximal mice genomic region upstream of the insulin transcriptional start site (around 400 bp) had already been shown to contain all the regulatory elements necessary to confer glucose-dependent regulation in cultured β-cell derived lines [29]. Hence, we cloned the proximal 645 bp – a slightly larger region than strictly required – from this promoter to drive the luciferase gene transcription.
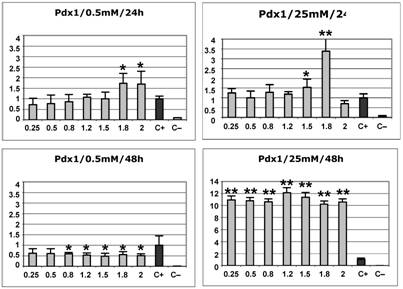
In order to set up the conditions of transfection and overexpression of all the ParaHox constructs, we first evaluated the effects of PDX1 on β-cells under high glucose concentrations, as it is well known that, under these conditions, PDX1 is able to transactivate the insulin promoter [30, 31]. To determine the post-transfection time needed to observe the effects of protein overexpression, we compared the results of PDX1 overexpression in the βTC6 cell line at either 24 h or 48 h post-transfection. As depicted in Fig. 2, the results at 48 h agreed consistently with the reported effects of PDX1 upon the insulin promoter: at low glucose concentration, there is no activation (in fact, the excess of PDX1 inhibits very weakly the promoter), whereas at high glucose concentration, high transactivation levels were robustly detected (more than 10 fold), even at low DNA concentration of the construct expressing the transcription factor.
Effect of PDX1 overexpression on the insulin promoter in the βTC6 cell line at low (0.5 mM) or high (25 mM) glucose concentrations at either 24 h or 48 h post-transfection. The values on the x axis represent the increasing dose (in μg) of the PDX1 expression construct used in the transfections. The y axis represent the arbitrary transactivation values of the insulin promoter with respect to the positive control (c+), which is considered as 1. The standard deviation is also indicated. At least four independently transfected replicates of each sample were analysed. c+ : positive control (cells transfected with the –645INS-LUC construct but no transcription factor). c– : negative control (cells transfected with empty vectors). Values statistically different from the control by the Mann-Whitney test (p<0.05) are indicated by one asterisk. Statistically significant values that show a stronger effect of the transcription factor on the insulin promoter (at least two-fold or more, or half or less, than the positive controls) are indicated by two asterisks.
Taken together, these results confirmed that overexpression of exogenous PDX1 greatly enhances transcription from the insulin promoter in β-derived cells (βTC6 cell line), and show that under our conditions, 48 h post-transfection were required to elicit an appropriate response to different glucose concentrations. Therefore, and in order to compare the effects of the overexpression of all the ParaHox genes, cells were all similarly treated and harvested at 48 h post-transfection.
Overexpression of ParaHox genes in the βTC6 cell line
To evaluate the role of the ParaHox proteins as potential transcriptional regulators of the insulin gene, we overexpressed these proteins in βTC6 cell lines and monitored the activation of a cotransfected reporter plasmid containing the mouse insulin II gene promoter (-645 to –1). The activation of the insulin promoter is very complex and requires a combination of interacting transcription factors specific of endocrine pancreas, which are activated or produced under high glucose concentration. Transfection on β-cells allowed us to test directly the effect of any ParaHox gene in a cell environment that provided all the necessary partners and transcription machinery, including endogenous levels of PDX1. In this context, our positive control, to which we have referred all transactivation values, is the basal transactivation level of the luciferase reporter gene driven by the insulin promoter, obtained without the overexpression of any transcription factor. The positive value observed for this control (c+) showed that the promoter was indeed bound and transactivated by endogenous protein complexes.
The effect on the transactivation or inhibition of a particular promoter by transcription factors is dependent on the amount of transcription factor available, particularly when low affinities or competition for the binding sites are taken into consideration. Although, most of the studies on the insulin promoter have been performed at high concentrations of transcription factors, we decided to test the ParaHox transcription factors in a series of increasing amounts, while maintaining constant the other of parameters. Therefore, we transfected from 0.25 micrograms (low amount) to 2 micrograms (high amount) of construct to study the effect on the insulin gene transcription in a dose dependant manner. The trends on the promoter are therefore more easily and consistently detected.
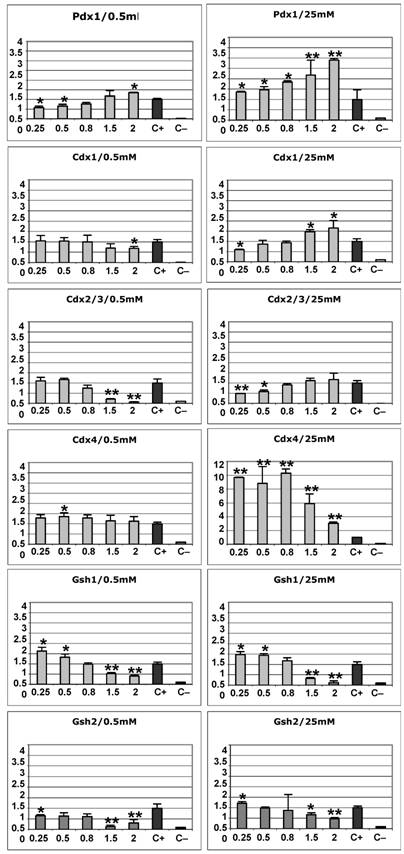
The results are summarised in the histogram diagrams of Fig. 3. All experiments were consistently replicated, and statistical significance of the observed differences was determined using the Mann-Whitney test (p<0.05, indicated by one asterisk). In summary, GSH1 and GSH2 over expression resulted in inhibition of the promoter, more pronounced for GSH2, at high glucose concentration (Fig. 3), whereas CDX1 and CDX2/3 showed a transactivation effect on the insulin promoter. CDX1, which acted in a glucose-independent manner, yielded around 2-fold increase in activity at the highest concentration of protein (Fig. 3). CDX2/3, instead, responded differently depending on the glucose concentration. While having no statitistically significant effect at low glucose levels, it was able to transactivate the insulin promoter at high glucose concentration, in accordance to previous reports [22, 23]. This effect was particularly observed when high amounts of the transcription factor construct were transfected (4.4-fold increase). In contrast, CDX4, which was also sensitive to glucose levels, produced the reverse effect to that of CDX2/3. Whereas at a low glucose concentration CDX4 over expression did not affect the basal transcription of the insulin promoter, at high glucose concentration it readily inhibited transcription to nearly null values in a transcription factor dose-dependent manner (Fig. 3).
Overexpression of ParaHox genes in the αTC1 cell line
We next intended to evaluate the effects of the over expression of the ParaHox genes on the insulin promoter in an α-derived cell line, which, on the one hand, is of endocrine origin, close to β-cells, sharing many transcriptional pathways and expressing similar transcription factors, but which, on the other hand, respond differently to glucose concentrations. In this respect, other authors have shown that the insulin promoter can be activated in α-cells after ectopic expression of PDX1 under high glucose conditions, thus demonstrating the presence in α-cells of the coactivator partners required for the transcriptional regulation of the insulin gene [32].
The effects of the ParaHox over expression on the regulation of the insulin promoter were assessed by transient transfections on the αTC1 cell line, similarly to those performed in βTC6 cells, and the results are summarised in Fig. 4. Overexpression of PDX1 in α-cells, at low glucose concentrations, resulted in a very low activation of the promoter, and only at high doses of the transcription factor construct (Fig. 4), while at high glucose concentration, the activation of the promoter steadily increased in a dose-dependent manner, reaching around 3-fold increase at the highest amount of transcription factor construct (Fig. 4). This result shows that, in the presence of glucose, PDX1 can transactivate the insulin promoter in α-cells, albeit at a much lower efficiency than in β-cells, in accordance to previous reports [32-34].
Effect of ParaHox overexpression (except PDX1) on the insulin promoter in the βTC6 cell line at low (0.5 mM) or high (25 mM) glucose concentration, 48 h post-transfection. The values on the x axis represent the increasing dose (in micrograms) of each transcription factor expression construct used in the transfections. The y axis represent the arbitrary transactivation values of the insulin promoter with respect to the positive control (c+), which is considered as 1. c+ : positive control (cells transfected with the –645INS-LUC construct but no transcription factor). c– : negative control (cells transfected with empty vectors). Values statistically different from the control by the Mann-Whitney test (p<0.05) are indicated by one asterisk. Statistically significant values that show a stronger effect of the transcription factor on the insulin promoter (at least two-fold or more, or half or less, than the positive controls) are indicated by two asterisks.

Effect of ParaHox overexpression, including PDX1, on the insulin promoter in the αTC1 cell line at low (0.5 mM) or high (25 mM) glucose concentrations, 48 h post-transfection. The values on the x axis represent the increasing dose (in μg) of each transcription factor expression construct used in the transfections. The y axis represent the arbitrary transactivation values of the insulin promoter with respect to the positive control (c+), which is considered as 1. c+: positive control (cells transfected with the –645INS-LUC construct but no transcription factor). c– : negative control (cells transfected with empty vectors). Values statistically different from the control by the Mann-Whitney test (p<0.05) are indicated by one asterisk. Statistically significant values that show a stronger effect of the transcription factor on the insulin promoter (at least two-fold or more, or half or less, than the positive controls) are indicated by two asterisks.
Again, GSH1 and GSH2 effects on the insulin promoter were not glucose dependent, and as shown for β-cells, their over expression caused a strong inhibition of transcription from the insulin promoter in a dose-dependent manner, particularly for GSH1 (Fig. 4). CDX1 over expression did not result in major changes on the insulin promoter activity, though at high glucose concentrations and at high concentration of transcription factor, it weakly increased the activity from the insulin promoter (Fig. 4). Instead, CDX2/3 over expression inhibited the insulin promoter at low glucose concentrations to nearly null values at high concentrations of the transcription factor (Fig. 4). Interestingly, CDX4 again rendered the most unexpected result, as it showed sensitivity to glucose concentration and, at high glucose concentration, highly transactivated the insulin promoter, reaching 10-fold the value of the control. This effect was more pronounced at lower transcription factor doses. Noticeably, the effect of CDX4 over expression in β-cells at high glucose concentrations was just the reverse, while again not detectable effects were observed at low levels of glucose.
In a more physiological context, given that: i) all ParaHox genes are expressed in glucagon-producing α cells of the endocrine pancreas, ii) α cells do not express insulin and, iii) taking into consideration the above results, we suggest that the inhibitory effects of GSH1, GSH2 and CDX2/3 may contribute to the regulation of the insulin promoter in α cells.
4. DISCUSSION
The over expression of proteins in mammalian cells, is highly dependent on mRNA stability and nucleocytoplasmic export. In this context, our strategy to include either one genomic or synthetic intron in each expression construct has guaranteed the over expression of the transcription factor, and not merely that of its RNA (Figure 1, [25, 26]). We also set the conditions to assay the effects of the over expression of all ParaHox genes after time-course experiments with PDX1. Most probably, the higher response at two days was due to the higher time of mRNA production or high protein stability, which may accumulate and hence, induce a higher transactivation of the reporter gene. Besides, high PDX1 production during such a period of time may also activate, in a positive feedback loop, other interacting transcription factors that synergistically act upon the insulin promoter [35, 36], thus eliciting a stronger and robust promoter response [14]. The fact that the overexpression of one single transcription factor, PDX1, is sufficient to transactivate the insulin promoter, even when the binding of other coactivators are required to promote transcription, suggests that PDX1 may be a limiting transactivating factor in β cells, consistent with its finely-tuned regulation.
PDX1 was also able to activate the insulin promoter in a glucose-dependent manner in α cells, indicating that many of the proteins required for transactivation and regulation of the insulin promoter are shared between α- and β-cells. However, the effect was much less pronounced than in β-cells, in accordance to previous reports [32-34]. Concerning the other the ParaHox genes and comparing the results on β and α cells, in general, and not unexpectedly, the effects of their overexpression, either inhibiting or transactivating, are much more pronounced at high glucose concentration. This probably reflects the availability of other transcription factors or coactivators in these cells that respond to glucose concentration, and bind to the insulin promoter. Given that all ParaHox genes share a very similar homeodomain sequence, and probably bind similar sequences, our results are more easily interpreted in the view of differences in affinity binding to the target site and/or in partner recruitment.
GSH1 and GSH2 did not appear to affect overtly the regulation of the insulin promoter, whereas the over expression of CDX proteins resulted in a more complex regulation pattern. Overall, GSH2 overexpression resulted in the inhibition of the promoter in β cells, depending on the transcription factor dose, but irrespective of glucose concentration. This inhibitory effect could be due to GSH2 interfering with target recognition by endogenous factors and/or sequestering of other endogenous coactivators and main transactivators, amongst them, PDX1. In fact, homeodomain-containing transcription factors usually act by assembling into homo- or hetero-oligomers. Different partners would shift recognition and affinity among different DNA motifs [14]. A similar inhibitory effect was observed when over expressing GSH1 in α cells, again irrespective of glucose concentration
CDX1 over expression did not visibly affect the transcription from the insulin promoter in α cells, while a two-fold transactivation response was clearly detected in β cells, even at low concentration of transfected construct, suggesting a conserved ability of CDX1 to recognise and bind targets at the insulin promoter. The weak transactivation effect and the fact that it is glucose-independent, points to recruitment of glucose-independent, and probably non-synergic, transcription factors that are available in β- but not in α-cells.
CDX2/3 over expression rendered a different result in α or β cells, depending on glucose concentration. In α cells, the insulin promoter was clearly inhibited at low glucose concentration, whereas it was transactivated in β cells at high glucose concentration. These findings suggest that CDX2/3 binds to - specific insulin promoter motifs (in accordance to the reported binding to the A-boxes of the insulin promoter [22]), resulting in either inhibition or transactivation, depending on the recruitment and interaction to other transcription factors, which differ between these two cell lines. However, the ability to bind and transactivate the insulin promoter in β cells at high glucose concentration, when compared to that of PDX1, is moderate and highly dependent on the transcription factor dose, thus pointing to a reduced affinity for either the promoter targets or limited synergy with other required transcription factors.
Remarkably, CDX4 elicited a strong and completely reverse response compared to that of CDX2/3 in the two cell lines, or that of PDX1 in β cells. This effect of CDX4 over expression was highly dependent on glucose concentration and the cell type. In β cells, and only at high glucose concentrations, CDX4 strongly inhibited the insulin promoter to null activity. In contrast, CDX4 transactivated very efficiently (10 fold) the same promoter in α cells, while not affecting it, whatsoever, at low glucose concentrations. This effect was more pronounced at low doses of the transcription factor. Several scenarios would account for these results. First, CDX4 may recognise and bind directly one or several targets in the insulin promoter. In β cells this binding would either prevent the binding of the endogenous transactivation factors, disrupt or sequester activating proteins or protein complexes essentially required for transactivation of the insulin promoter, or recruit inhibitory proteins. In this context, as PDX1 is a limiting factor in β cells, oligomerization with excess of CDX4 could effectively deplete the PDX1 endogenous pools. Conversely, the response that CDX4 elicited in α cells, again indicated binding to particular motifs and, in this case, recruitment of α-cell specific transcription factors or coactivators, thus strongly transactivating the insulin promoter. In response to high glucose concentration, the transactivation effect of CDX4 over expression in α cells reached the same levels of that of PDX1 in β cells. However, CDX4 showed a saturating effect at high transcription factor amounts, which was not observed with PDX1.
In summary, in β cells, no other ParaHox gene can substitute efficiently PDX1 on the transactivation of the insulin promoter in response to glucose concentration, as CDX2/3 was able to do so only at high amounts of transcription factor construct, and CDX1 transactivated it moderately, but irrespectively of glucose levels. Instead, if we consider the results on α-cells, CDX4 effectively transactivates the insulin promoter, providing a strong response dependent on glucose concentration, remarkably similar to that of PDX1 in β cells. Our results provide some proof of principle that distinct ParaHox transcription factors can bind to the same promoter, in this case, the insulin promoter, the final transactivating or inhibiting effect depending on the strength and affinity to the target sites and their interaction with other co-factor partners. Consistently, both PDX1 and CDX2 have been shown to form heteromeric complexes with PBX1, PDX1 in β and δ cells [37, 38], and CDX2, in α cells [21]. The rest of ParaHox proteins may also partially share some common cofactor networks, targets and responses to particular physiological conditions and might well be responsible for unaccounted events, such as the few insulin producing cells in the Pdx1 knockout. These may hold true for other transcription factors belonging to the same family and with a conserved homology.
We are aware that some of these transactivation effects of the ParaHox overexpression on the insulin promoter in cultured α- or β- cells could be non-physiological. For starters, only Pdx1 is endogenously expressed in β cells, while the rest of ParaHox is endogenously expressed in α cells [24]. Nonetheless, on these grounds it is remarkable that CDX2/3, GSH1 and GSH2 have strong inhibitory effects on the insulin promoter, and thus may contribute to the silence of the insulin gene in α cells under physiological conditions.
Finally, given the prevalence of pancreas disorders in humans, that all ParaHox genes are expressed in adult pancreas and, particularly, the results on CDX4 ability to transactivate the insulin promoter in α -cells, our data may open new venues to explore the physiological role of the ParaHox genes in α-cell-based therapy regarding glucose homeostasis.
Acknowledgements
We thank Pedro Herrera (University of Geneve) for encouraging discussions. We also thank the Servei Científico-Tècnics de la Universitat de Barcelona for the use of the ABI PRISM 3700 Sequencer. This work was partially supported by 990610 (Fundació La Marató de TV3) to J. G.-F. and G. M. and BFU04-01343 (Ministerio de Educación y Ciencia, España) to G. M.
CONFLICT OF INTEREST
The authors have declared that there is no conflict of interest.
References
1. Babu DA, Deering TG, Mirmira RG. A feat of metabolic proportions: Pdx1 orchestrates islet development and function in the maintenance of glucose homeostasis. Mol Genet Metab. 2007;92:43-55
2. Stoffers DA, Ferrer J, Clarke WL, Habener JF. Early-onset type-II diabetes mellitus (MODY4) linked to IPF1. Nat Genet. 1997;17:138-139
3. Shieh SY, Tsai MJ. Cell-specific and ubiquitous factors are responsible for the enhancer activity of the rat insulin II gene. J Biol Chem. 1991;266:16708-16714
4. Melloul D, Marshak S, Cerasi E. Regulation of insulin gene transcription. Diabetologia. 2002;45:309-326
5. Hay CW, Docherty K. Comparative analysis of insulin promoters: implications for diabetes research. Diabetes. 2006;55:3201-3213
6. German M, Ashcroft S, Docherty K, Edlund H, Edlund T, Goodison S, Imura H, Kennedy G, Madsen O, Melloul D. The insulin gene promoter. A simplified nomenclature. Diabetes. 1995;44:1002-1004
7. Massari ME, Murre C. Helix-loop-helix proteins: regulators of transcription in eukaryotic organisms. Mol Cell Biol. 2000;20:429-440
8. German MS, Moss LG, Rutter WJ. Regulation of insulin gene expression by glucose and calcium in transfected primary islet cultures. J Biol Chem. 1990;265:22063-22066
9. Melloul D, Ben-Neriah Y, Cerasi E. Glucose modulates the binding of an islet-specific factor to a conserved sequence within the rat I and the human insulin promoters. Proc Natl Acad Sci USA. 1993;90:3865-3869
10. German MS, Wang J. The insulin gene contains multiple transcriptional elements that respond to glucose. Mol Cell Biol. 1994;14:4067-4075
11. Peers B, Leonard J, Sharma S, Teitelman G, Montminy MR. Insulin expression in pancreatic islet cells relies on cooperative interactions between the helix loop helix factor E47 and the homeobox factor STF-1. Mol Endocrinol. 1994;8:1798-1806
12. Odagiri H, Wang J, German MS. Function of the human insulin promoter in primary cultured islet cells. J Biol Chem. 1996;271:1909-1915
13. Sander M, Griffen SC, Huang J, German MS. A Novel glucose-responsive element in the human insulin gene functions uniquely in primary cultured islets. Proc Natl Acad Sci USA. 1998;95:11572-11577
14. Ohneda RG, Mirmira J, Wang JD, Johnson MS, German MS. The homeodomain of PDX-1 mediates multiple protein-protein interactions in the formation of a transcriptional activation complex on the insulin promoter. Mol Cell Biol. 2000;20:900-911
15. Garcia-Fernàndez J. The genesis and evolution of the homeobox gene clusters. Nature Rev Genetics. 2005;6:881-892
16. Ferrier DE, Dewar K, Cook A, Chang JL, Hill-Force A, Amemiya C. The chordate ParaHox cluster. Curr Biol. 2005;15:R820-822
17. Mulley JF, Chiu CHY, Holland PW. Breakup of a homeobox cluster after genome duplication in teleosts. Proc Natl Acad Sci USA. 2006;103:10369-10372
18. Martinez P, Amemiya CT. Genomics of the HOX gene cluster. Comp Biochem Physiol B. 2002;133:571-580
19. Pearson JC, Lemons D, McGinnis W. Modulating Hox gene functions during animal body patterning. Nature Rev Genetics. 2005;6:893-904
20. Akin ZN, Nazarali AJ. Hox genes and their candidate downstream targets in the developing central nervous system. Cell Mol Neurobiol. 2005;25:697-741
21. Liu T, Branch DR, Jin T. Pbx is a cofactor for Cdx-2 in regulating proglucagon gene expression in pancreatic A cells. Mol Cell Endocrin. 2006;249:140-149
22. German MS, Wang J, Chadwick RB, Rutter WJ. Synergistic activation of the insulin gene by a LIM-homeo domain protein and a basic helix-loop-helix protein: building a functional insulin minienhancer complex. Genes Dev. 1992;6:2165-2176
23. Zhao Y, Liu T, Zhang N, Yi F, Wang Q, Fantus IG, Jin T. Role of Cdx-2 in insulin and proglucagon gene expression: a study using the RIN-1056A cell line with an inducible gene expression system. Journal Endocrinol. 2005;186:179-192
24. Rosanas-Urgell A, Marfany G, Garcia-Fernàndez J. Pdx1 related-homeodomain transcription factors are distinctly expressed in adult pancreatic islets. Mol Cell Endocrinol. 2005;237:59-66
25. Le Hir H, Nott A, Moore MJ. How introns influence and enhance eukaryotic gene expression. Trends Biochem Sci. 2003;28:215-220
26. Nott A, Le Hir H, Moore MJ. Splicing enhances translation in mammalian cells: an additional function of the exon junction complex. Genes Dev. 2004;18:210-222
27. Karlsson O, Walker MD Rutter WJ, Edlund T . Individual protein-binding domains of the insulin gene enhancer positively activate β-cell-specific transcription. Mol Cell Biol. 1989;9:823-827
28. Mizusawa N, Hasegawa T, Ohigashi I, Tanaka-Kosugi C, Harada N, Itakura M, Yoshimoto K. Differentiation pehnotypes of pancreatic islet β- and α- cells are closely related with homeotic genes and a group of differentially expressed genes. Gene. 2004;331:53-63
29. Petersen HV, Serup P, Leonard J, Michelsen BK, Madsen OD. Transcriptional regulation of the human insulin gene is dependent on the homeodomain protein STF1/IPF1 acting through the CT boxes. Proc Natl Acad Sci USA. 1994;91:10465-10469
30. Macfarlane WM, Read ML, Gilligan M, Bujalska I, Docherty K. Glucose modulates the binding activity of the β-cell transcription factor IUF1 in a phosphorylation-dependent manner. Biochem J. 1994;303:625-631
31. Marshak S, Totary H, Cerasi E, Melloul D. Purification of the β-cell glucose-sensitive factor that transactivates the insulin gene differentially in normal and transformed islet cells. Proc Natl Acad Sci USA. 1996;93:15057-15062
32. Campbell SC, Macfarlane WM. Regulation of the pdx1 gene promoter in pancreatic β-cells. Biochem Biophys Res Commun. 2002;299:277-284
33. Serup P, Jensen J, Andersen FG, Jorgensen MC, Blume N, Holst JJ, Madsen OD. Induction of insulin and islet amyloid polypeptide production in pancreatic islet glucagonoma cells by insulin promoter factor 1. Proc Natl Acad Sci USA. 1996;93:9015-9020
34. Watada H, Kajimoto Y, Miyagawa J, Hanafusa T, Hamaguchi K, Matsuoka T, Yamamoto K, Matsuzawa Y, Kawamori R, Yamasaki Y. PDX-1 induces insulin and glucokinase gene expressions in αTC1 clone 6 cells in the presence of betacellulin. Diabetes. 1996;45:1826-1831
35. Gerrish K, Cissell MA, Stein R. The role of hepatic nuclear factor 1α and PDX-1 in transcriptional regulation of the pdx-1 gene. J Biol Chem. 2001;276:47775-47784
36. Chakrabarti SK, James JC, Mirmira RG. Quantitative assessment of gene targeting in vitro and in vivo by the pancreatic transcription factor, Pdx1. Importance of chromatin structure in directing promoter binding. J Biol Chem. 2002;277:13286-13293
37. Kim SK, Selleri L, Lee JS, Zhang AY, Gu X, Jacobs Y, Cleary ML. Pbx1 inactivation disrupts pancreas development and in Ipf-1 defficient mouse promotes diabetes mellitus. Nat Genet. 2002;30:430-435
38. Peers B, Sharma S, Johnson T, Kamps M, Montminy M. The pancreatic islet factor STF-1 binds cooperatively with Pbx to a regulatory element in the somatostatin promoter: importance of the FPWMK motif and of the homeodomain. Mol Cell Biol. 1995;15:7091-7097
Author contact
![]() Correspondence to: Departament de Genètica, Facultat de Biologia, Universitat de Barcelona, Avda. Diagonal, 645, E-08028 Barcelona, Spain. Tel: +34 934021502; fax: +34 934034420. E-mail address: gmarfanyedu
Correspondence to: Departament de Genètica, Facultat de Biologia, Universitat de Barcelona, Avda. Diagonal, 645, E-08028 Barcelona, Spain. Tel: +34 934021502; fax: +34 934034420. E-mail address: gmarfanyedu
Received 2008-1-10
Accepted 2008-2-4
Published 2008-2-6
