ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2009; 5(1):82-96. doi:10.7150/ijbs.5.82 This issue Cite
Research Paper
Aberrant p63 and WT-1 expression in myoepithelial cells of pregnancy-associated breast cancer: implications for tumor aggressiveness and invasiveness
1. Department of Thyroid and Breast Surgery, China-Japan Union Hospital, Jilin University, Changchun, Jilin, China;
2. Mammalian Genetics Section, GDDB, NIDDK, National Institutes of Health, Bethesda, MD, USA;
3. Department of Gynecologic and Breast Pathology, Armed Forces Institute of Pathology and American Registry of Pathology, Washington DC, USA
Abstract
Our recent studies revealed that focal alterations in breast myoepithelial cell layers significantly impact the biological presentation of associated epithelial cells. As pregnancy-associated breast cancer (PABC) has a significantly more aggressive clinical course and mortality rate than other forms of breast malignancies, our current study compared tumor suppressor expression in myoepithelial cells of PABC and non-PABC, to determine whether myoepithelial cells of PABC may have aberrant expression of tumor suppressors. Tissue sections from 20 cases of PABC and 20 cases of stage, grade, and age matched non-PABC were subjected to immunohistochemistry, and the expression of tumor suppressor maspin, p63, and Wilms' tumor 1 (WT-1) in calponin positive myoepithelial cells were statistically compared. The expression profiles of maspin, p63, and WT-1 in myoepithelial cells of all ducts encountered were similar between PABC and non-PABC. PABC, however, displayed several unique alterations in terminal duct and lobular units (TDLU), acini, and associated tumor tissues that were not seen in those of non-PABC, which included the absence of p63 and WT-1 expression in a vast majority of the myoepithelial cells, cytoplasmic localization of p63 in the entire epithelial cell population of some lobules, and substantially increasing WT-1 expression in vascular structures of the invasive cancer component. All or nearly all epithelial cells with aberrant p63 and WT-1 expression lacked the expression of estrogen receptor and progesterone receptor, whereas they had a substantially higher proliferation index than their counterparts with p63 and WT-1 expression. Hyperplastic cells with cytoplasmic p63 expression often adjacent to, and share a similar immunohistochemical and cytological profile with, invasive cancer cells. To our best knowledge, our main finings have not been previously reported. Our findings suggest that the functional status of myoepithelial cells may be significantly associated with tumor aggressiveness and invasiveness.
Keywords: Pregnancy-associated breast cancer, myoepithelial cells, p63, WT-1, tumor stem cells.
Introduction
The normal and pre-invasive human breast epithelium, which is the histological origin of over 80% of breast malignancies, is physically separated from the stroma by both the basement membrane and myoepithelial cells. The basement membrane is composed of type IV collagen, laminins, and other molecules, forming a continuous lining surrounding and attaching to myoepithelial cells [1-2]. Myoepithelial cells are joined by intercellular junctions and adhesion molecules, forming a continuous sheet that completely surrounds the normal duct system and a vast majority of the lobular units [3-4]. The predominant and phenotypic structural constituent of myoepithelial cells is microfilament, which could be elucidated by immunohistochemistry with a number of commercially available antibodies, including those to calponin, smooth muscle actin, smooth muscle myosin heavy chain, H-caldesmon, P-cadherin, and Cytokeratins 5, 7, 14, and 17 [5]. In H&E stained sections, myoepithelial cells are in cuboid or spindle shape with pale cytoplasm and nucleus. Under an electronic microscope, normal myoepithelial cells have a substantially fewer or no distinct rough reticulum endoplasm, Golgi complex, secretory granules, and a very low level of proliferating activities, compared to the associated epithelial cells [3]. Thus, myoepithelial cells have been traditionally regarded as an idle and passive physical barrier with no significant active functions [1-4].
Recent studies, however, have suggested that myoepithelial cells may possess a number of active functions. One of the functions is the production of tumor suppressers, including maspin, Wilms' tumor 1 (WT-1), p63, and p73 [5-9]. In vitro and in vivo studies have shown that these tumor suppressors have significant paracrine inhibitory functions against tumor growth and invasion [5-9]. Our recent studies revealed that about 90% of morphologically distinct myoepithelial cells in normal and hyperplastic ducts had distinct expression of these tumor suppressors, whereas only about 70% of the myoepithelial cells in ducts with in situ breast cancer expressed these tumor suppressors [5,9]. Our studies also revealed that a subset of normal, hyperplastic, and pre-invasive ducts and acini harbored focal disruptions (the absence of myoepithelial cells resulting a gap larger than a combined size of at least three myoepithelial cells) in their surrounding myoepithelial cell layers, and that a subset of morphologically distinct myoepithelial cells completely lacked the expression of all myoepithelial cell phenotypic markers [10-19]. Ducts and acini with focally disrupted myoepithelial cell layers or myoepithelial cell layers lacked expression of tumor suppressors had a significantly higher rate of cell proliferation, ER negativity, and expression of invasion related molecules than their morphologically similar counterparts with normal myoepithelial cell layers [10-19]. Together, these findings suggest that a reduction of tumor suppressor expression and focal disruption of the myoepithelial cell layers may significantly contribute to tumor aggressiveness and invasiveness.
Pregnancy-associated breast cancer (PABC) occurs during pregnancy or within one year of delivery. Although the incidence of PABC is low, it is the second most common cancer in pregnant women [20-23]. It has been well documented that a normal full-term of pregnancy is one of the most effective means of decreasing the lifetime-risk of breast cancer, whereas PABC is consistently associated with the most aggressive clinical course and highest mortality rate among breast malignancies [20-23]. These seemingly conflicting impacts have been largely attributed to proliferation and differentiation of stem cells, and remodeling of the cellular microenvironment and extracellular matrix [22-23]. The specific molecular or cellular mechanism, or unique molecules that are directly linked to aggressiveness and invasiveness, however, have not been identified. As the myoepithelial cell is the sole source of several tumor suppressors that have significant paracrine inhibitory function on tumor cell growth and invasion [5-9], our current study attempted to determine whether the myoepithelial cell layer of PABC would have aberrant tumor suppressor expression.
Materials and Methods
Formalin-fixed, paraffin-embedded tissue blocks from 20 patients with PABC, 20 cases of stage, grade, and age matched non-PABC, and 20 reduction mammoplasties of individuals with no family history of breast cancer, and no mammographic or histological breast abnormalities were selected from the cases sent to our institute for consultation with patients' consent. All selected PABC and non-PABC cases contained normal, hyperplastic, in situ, or invasive components. Consecutive sections at 4-5 μm thickness were prepared and placed on positively charged slides. The first and last sections from each case were stained with hematoxylin and eosin (H&E) for morphological classification, based on our published criteria [24], which confirmed all the initial diagnosis.
Mouse monoclonal antibodies against human calponin (clone: CALP)), a most commonly used myoepithelial cell phenotypic marker, and maspin (clone: EAW24) were purchased from Novocastra Laboratories Ltd (Newcastle, UK). Mouse monoclonal antibodies against human WT-1 protein (clone: 6F-H2) and p63 protein (clone: 4A4) were purchased from Cell Marque (Hot Springs, AR). A mouse monoclonal antibody for proliferation marker Ki-67 (Clone: MM1) was purchased from DAKO (Capterria, CA). Mouse monoclonal antibodies against human estrogen receptor (ER; clone: ER-6F11) and progesterone receptor (PR; clone: PGR-312) were purchased from Novocastra Laboratories Ltd (Newcastle, UK). The secondary antibody, the ABC detection kit, and the DAB chromogen kit were purchased from Vector Laboratories (Burlingame, CA). A red chromogen, AP red, was purchased from Zymed Laboratories (South San Francico, CA). Sets of four adjacent sections were subjected to immunohistochemical staining using the protocol provided by the manufacturers.
To assess the specificity of the immunostaining, three technical approaches were used. First, different negative controls were used, which included (1) the substitution of the primary antibody with normal serum, (2) the omission of the secondary antibody from the immunostaining sequence, (3) serial dilutions of the primary antibody. Second, the same immunostaining protocol was used on the same cases, but substituting with different detection system and substrates. Third, the immunostaining procedure was repeated at least twice using the same protocol and under the same condition and immunostained sections were independently evaluated by at least two investigators. A given cell was considered immunoreactive to a given antibody when distinct immunoreactivities were consistently seen in either its cytoplasm nucleus, whereas all negative controls were devoid of distinct immunostaining.
To further confirm the specificity of calponin and WT-1 immunostaining, which occasionally cross-react with fiberoblasts and endothelial cells, a set of three adjacent sections from each case were subjected to immunohistochemical staining with molecular antibodies to cytokeratin (CK) 5 (clone: XM26), 14 (clone: LL002), and 17 (clone: E3; Novocastra Laboratories Ltd, Newcastle, UK), which are exclusively expressed in myoepithelial cells and cells with the epithelial origin.
From each case, 4-5 randomly selected duct and acinar clusters of each tissue component were photographed, and enlarged printers were made. As calponin is uniformly expressed in all or nearly all morphologically identifiable myoepithelial cells in H&E stained sections, the number of myoepithelial cells in a given duct or acinus was first determined in calponin stained sections. Using the obtained number as references, the number of positive and negative myoepithelial cells for each tumor suppressor in each duct or acinus was counted, and the numbers in different ducts and acini were added and averaged. The averaged percentages of tumor suppressor expressing cells in morphologically similar tissue components of PABC and non-PABC were statistically compared with the Pearson's Chi-squared test. The proliferation indexes in morphologically similar ducts or acini with and without aberrant p63 and WT-1 expression were similarly compared. Statistical significance was defined as the p-value < 0.05.
Results
Distinct immunoreactivities to calponin, p63, maspin, and WT-1 were elusively or preferentially seen in myoepithelial cells. WT-1, however, was also seen in endothelial cells and duct and acinar lumen. The negative controls were devoid of immunoreactivities to any of these molecules. Immunoreactivities to calponin, maspin, and WT-1 were predominantly seen in the cytoplasm. Immunoreactivities to Ki-67, ER, and PR were seen exclusively in the nuclei. Immunoreactivities to p63 were predominantly seen in the nuclei, but also seen in the epithelial cells of some cases (see below). Immunoreactivities to cytokeratin 5, 14, and 17 were exclusively present in the cytoplasm of myoepithelial cells.
In reduction mammoplasties and the morphologically clear-cut normal tissue component of both PABC and non-PABC, epithelial and myoepithelial cells were roughly equal in number, and all or nearly all the myoepithelial cells were similar in shape and size. Over 90% of morphologically distinct myoepithelial cells were uniformly immunoreactive to the myoepithelial cell phenotypic marker calponin, and about 90% of calponin positive myoepithelial cells co-expressed maspin, WT-1, and p63 (Fig. 1).
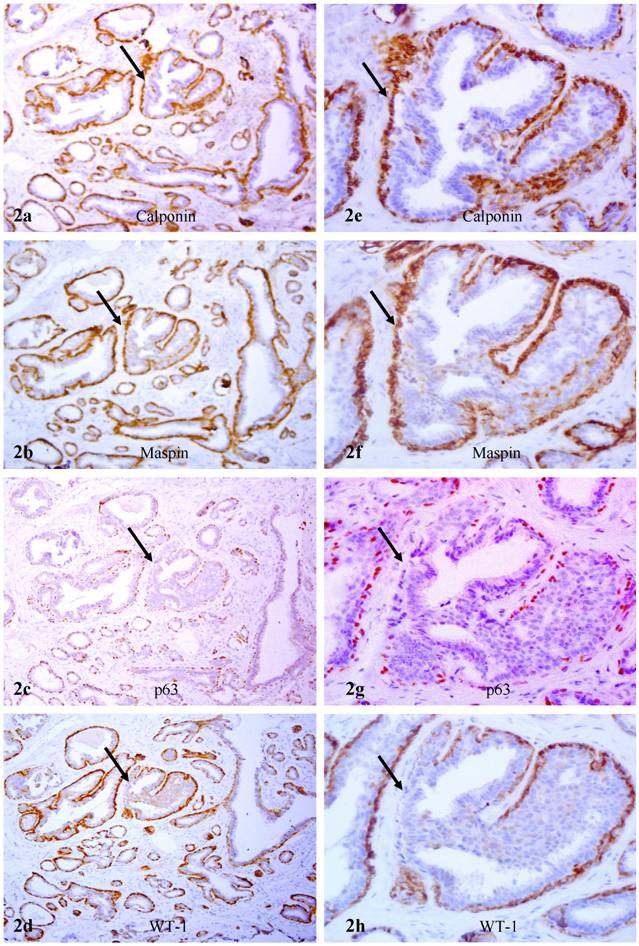
In hyperplastic and in situ malignant tissue components of non-PABC, the relative number of myoepithelial cells was significantly fewer than that of epithelial cells, and the shape and size of myoepithelial cells varied substantially. Over 90% of morphologically distinct myoepithelial cells, however, were uniformly and strongly immunoreactive to calponin immunostaining. Some ducts with malignant or hyperplastic lesions, however, harbored morphologically distinct myoepithelial cells that were strongly immunoreactive to calponin, whereas lacked the expression of p63 and WT-1 (Fig. 2).
Calponin and tumor suppressor expression in reduction mammoplasties. A set of four adjacent sections were immunostained for calponin and mapin, p63, and WT-1. Arrows identify myoepithelial cell layers. Note that about 90% of the myoepithelial cells in a small lobule are positive for calponin, and also co-express maspin, p63, and WT-1. a, c, e, and g: 100X; b, d, f, and h: a higher (300X) magnification of a, c, e, and g, respectively.
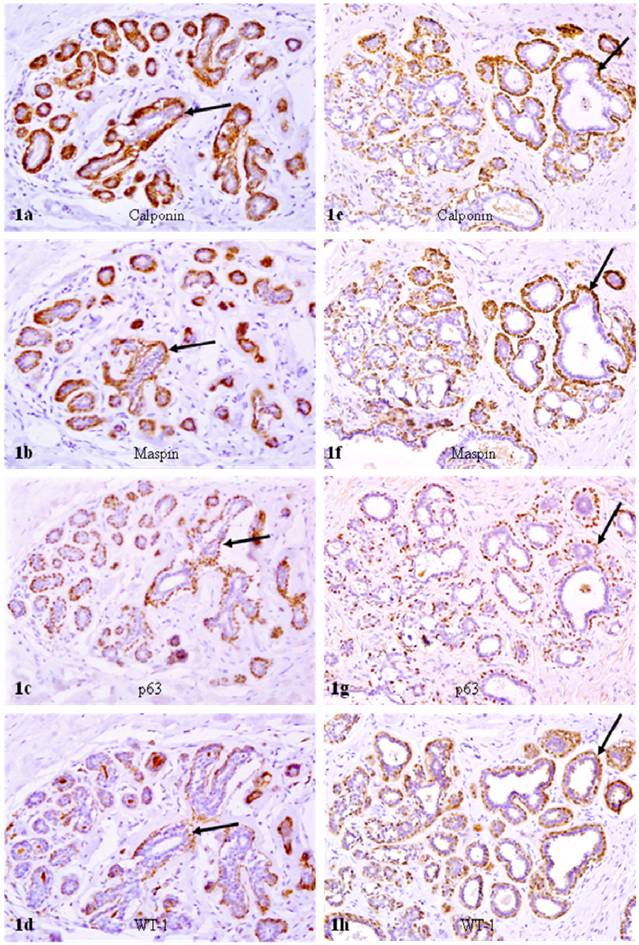
Lack of p63 and WT-1 in calponin positive myoepithelial cells A set of four adjacent sections were immunostained for calponin and mapin, p63, and WT-1. Arrows identify myoepithelial cell layers. Note that in a hyperplastic duct, a number of myoepithelial cells lack p63 and WT-1 expression, whereas they are positive for calponin and maspin. a, c, e, and g: 100X; b, d, f, and h: a higher (400X) magnification of a, c, e, and g, respectively.
In hyperplastic and in situ malignant components of PABC, the overall pattern of myoepithelial cell alterations and tumor suppressor expression was similar to that of non-PABC in all ducts encountered. PABC, however, displayed several unique properties that were not seen in their non-PABC counterparts:
1. Significantly fewer dusts encountered
In all 20 cases, normal or pathologically altered ducts accounted for only about 10% of the total epithelial cell population. Most ducts were distributed in the inter-lobular space.
2. Significantly decreased p63 and WT-1 expression in acini and terminal duct and lobular units (TDLU)
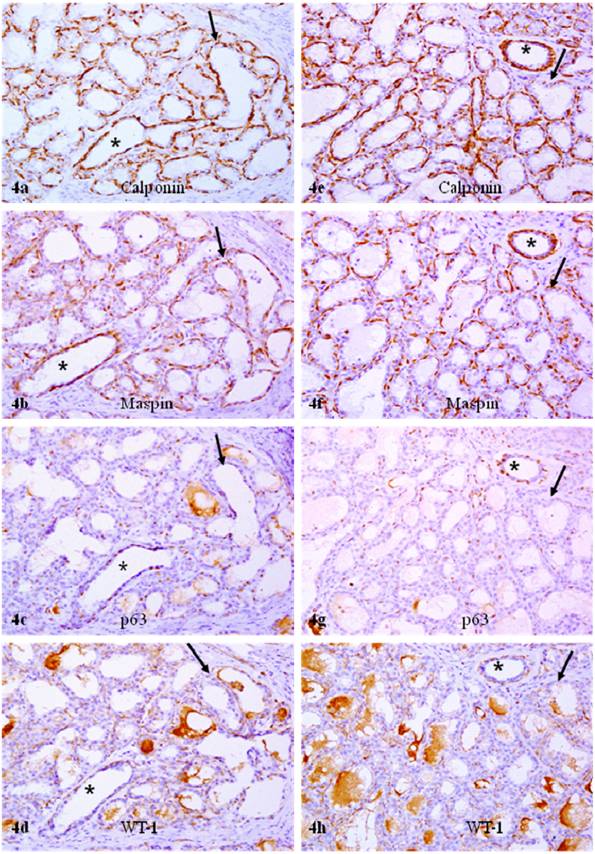
In sets of four immediate consecutive sections, calponin and maspin were uniformly expressed in all or nearly all morphologically distinct myoepithelial cells (Fig. 3a-3b). In sharp contrast, in about 80% of the acini and TDLU, only about 10% and 30% of calponin positive myoepithelial cells showed WT-1 and p63 expression, respectively (Figs. 3-4; Table 1). Under high magnification, all or nearly all acini and TDLU with reduced p63 and WT-1 expression showed fragmentations or small focal disruptions in the surrounding myoepithelial cell layers. In about 20% of the acini and TDLU, however, all or nearly all morphologically distinct myoepithelial cells uniformly expressed all four markers. These acini and TDLU were arranged as morphologically distinct lobules with variable sizes. These lobules were generally separated by stromal tissues from adjacent lobules that showed significantly reduced p63 and WT-1 expression (data not shown).
Reduced p63 and WT-1 expression in calponin and maspin positive myoepithelial cells of PABC. Two sets of four adjacent sections were immunostained for calponin and mapin, p63, and WT-1. Asterisks identify normal ducts in the inter-lobular space. Arrows identify myoepithelial cell layers. Note that a vast majority of the myoepithelial cells in normal ducts are positive for all four markers, whereas a vast majority of the myoepithelial cells in the acini lack p63 and WT-1 expression. 200X.
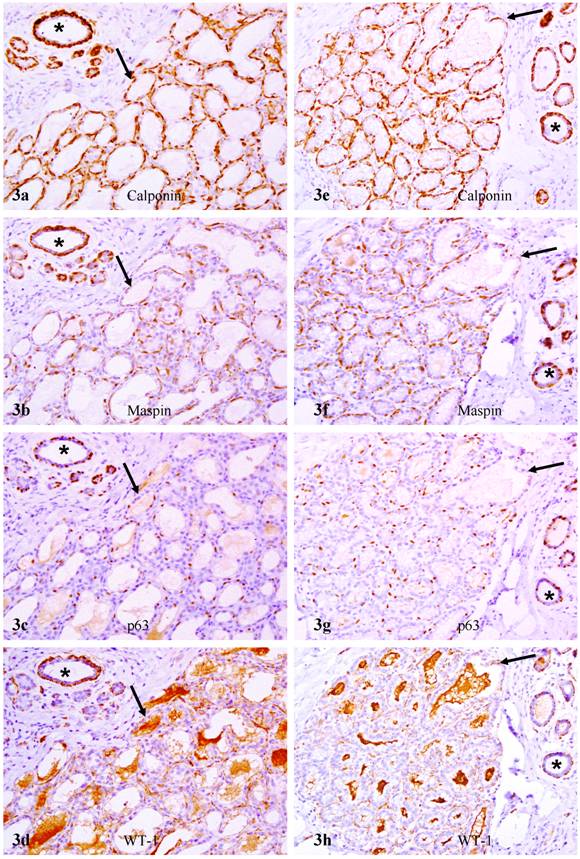
Reduced p63 and WT-1 expression in calponin and maspin positive myoepithelial cells of PABC. Two sets of four adjacent sections were immunostained for calponin and mapin, p63, and WT-1. Asterisks identify normal ducts in the inttra-lobular space. Arrows identify myoepithelial cell layers. Note that a vast majority of the myoepithelial cells in normal ducts are positive for all four markers, whereas a vast majority of the myoepithelial cells in the acini lack p63 and WT-1 expression. 200X.
Comparison of tumor suppressor expression in myoepithelial cells of PABC and non-PABC
| Tissue Type | Calponin (+) ME cells | With Maspin (%) | With p63 ((%) | With WT-1 (%) | p |
|---|---|---|---|---|---|
| Non-PABC ducts | 5,000 | 4,637 (92.7%) | 4,582 (91.6%) | 4,512 (90.2%) | |
| Acini or TDLU | 5,000 | 4,201 (84.0%) | 4,120 (82.4%) | 4,034 (80.1%) | >0.05 |
| PABC ducts | 2,000 | 1,806 (90.3%) | 1,796 (89.8%) | 1,724 (86.2%) | |
| Acini or TDLU | 5,000 | 4,015 (80.3%) | 1,560 (31.2%) | 541 (10.8%) | <0.01 |
3. Cytoplasmic p63 expression in the entire epithelial cell population of some lobules
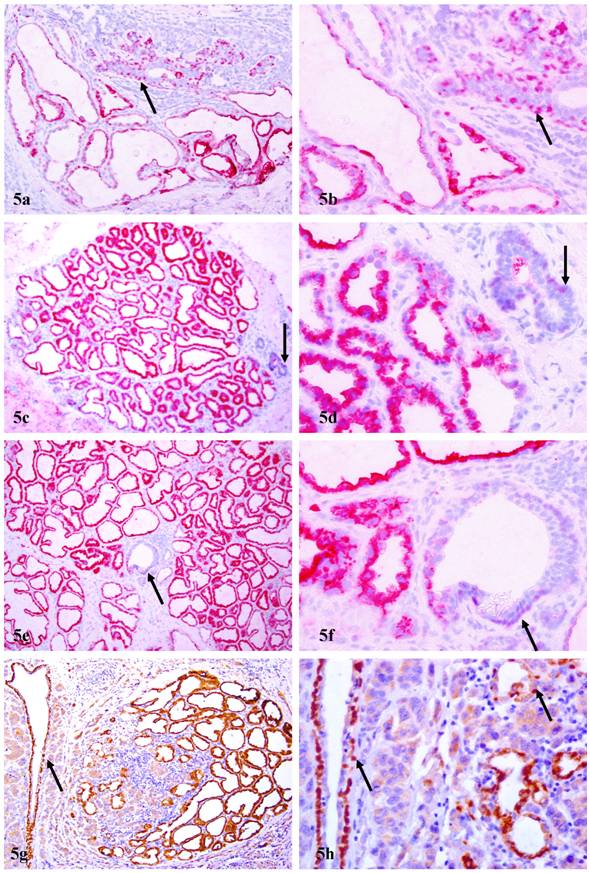
In 8 (40%) cases, distinct cytoplasmic p63 expression was seen in a subset of epithelial cells. The number of cells with cytoplasmic p63 expression among cases varied significantly, from a few acinar clusters to over 30% of the entire epithelial cell population. These normal or hyperplastic p63 expressing cells were arranged as lobules, in which all or nearly all the epithelial cells uniformly expressed high levels of p63, in sharp contrast to adjacent normal ducts, which showed distinct nuclear p63 expression (Fig. 5). These cytoplasmic p63 expressing cells were often adjacent and cytologically similar to invasive cancer cells, which also showed distinct cytoplasmic p63 expression (Figs. 5g-5h).
Cytoplasmic p63 expression in epithelial cells of PABC. Four sections from four different cases were immunostained for p63. Arrows identify myoepithelial cells with nuclear p63 localization in normal ducts. Note that the epithelial cells in some acinar clusters or the entire lobule show cytoplasmic p63 expression. In addition, some lobules with hyperplastic cells that show cytoplasmic p63 expression are immediately adjacent to, and share a similar morphological and immunohistochemical profile with, invasive cancer cells (g-h) a, c, e, and g: 100X; b, d, f, and h: a higher (400X) magnification of a, c, e, and g, respectively.
4. Increasing WT-1 expression in micro-vessels of the malignant tissue component
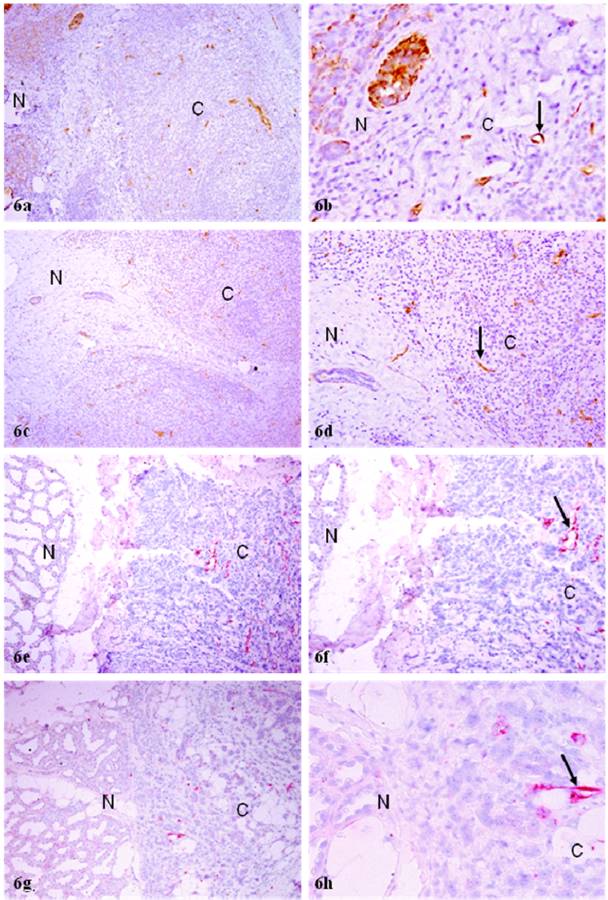
In normal and hyperplastic tissue components, most micro-vessels showed no distinct or very weak. WT-1 expression. In the malignant tissue component, however, all or nearly all morphologically identifiable micro-vessels were strongly immunoreactive to WT-1 (Fig. 6). The expression of WT-1 in vascular structures was confirmed by different immuno-detection systems with different chromogens (Fig. 6), and also by immunohistochemistry with endothelial cell specific markers CD31 (data not shown).
Increased cell proliferation in normal appearing epithelial cells near invasive lesions of PABC. Four sections from four different cases were double immunostained for calponin and Ki-67. N: normal; C: invasive cancer. Note that some morphologically normal appearing acinar clusters in the inter-lobular space or near the invasive cancer show substantially higher cell proliferation than their counterparts distant from the invasive lesion. a, c, e, and g: 80X; b, d, f, and h: a higher (200) magnification of a, c, e, and g, respectively.
5. Significantly elevated epithelial cell proliferation
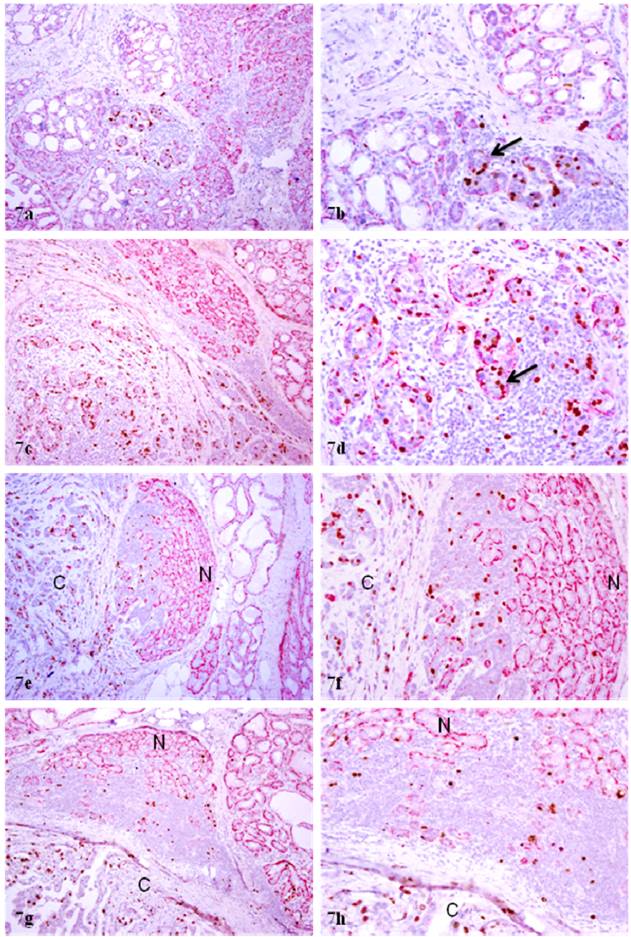
In sections double immunostained with myoepithelial cell and proliferation markers, a vast majority of the proliferating cells were seen in acinar clusters with focally disrupted myoepithelial cell layers, which consistently showed aberrant p63 or WT-1 expression. The size of these acinar clusters varied substantially and all these clusters showed a distinct boundary with their adjacent counterparts (Fig 7a-7b). A vast majority of these acinar clusters were immediately adjacent to invasive cancers (Fig.7c-7h).
Increased WT-1 expression in invasive lesions of PABC. Four sections from four different cases were double immunostained for WT-1. N: normal; C: invasive cancer. Note that the invasive tissue component has substantially more small vessels with WT-1 expression. a, c, e, and g: 80X; b, d, f, and h: a higher (200) magnification of a, c, e, and g, respectively.
6. Significantly reduced ER and PR expression
In non-PABC, about 50% of the acinar and duct cells in over 80% of the cases showed distinct ER or PR expression (data not shown). In sharp contrast, all cells in TDLU and acini with aberrant p63 and WT-1 expression lacked distinct ER or PR expression in all cases (data not shown).
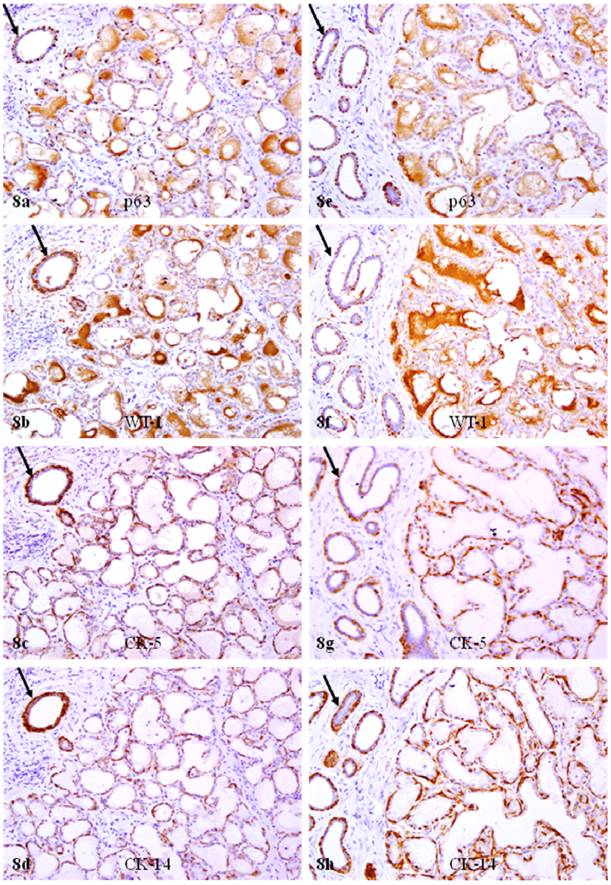
The significant reduction of p63 and WT-1 expression in calponin positive myoepithelial cells of PABC was confirmed by immunohistochemistry for CK 5, 14, and 17. In sets of four immediate adjacent sections, all or nearly all morphologically distinct myoepithelial cells were uniformly immunoreactive to CK 5, 14, and 17, but only about 10% and 30% of CK positive cells showed WT-1 and p63 expression, respectively (Fig 8).
Reduced p63 and WT-1 expression in cytokeratin positive myoepithelial cells of PABC. Two sets of four adjacent sections from two different cases were immunostained for CK 5, CK 14, p63, and WT-1, respectively. Arrows identify myoepithelial cells in normal ducts with expression of different markers. Note that all or nearly all morphologically distinct myoepithelial cells express CK 5 and CK 14, while a vast majority of the CK-positive cells lack p63 and WT-1 expression. 200X.
Discussion and Conclusions
The overall expression pattern of tumor suppressors in myoepithelial cells and biological presentations in epithelial cells seen in our current study are in total agreement with previous reports that the expression of p63 and WT-1 is inversely correlated with breast tumor progression and invasion [5-9]. Our findings of WT-1 expression in the vascular structure are in agreement with a recent study, which reported that “Endothelial WT1 expression was detected 95% of 113 tumors of different origin” [25]. To our best knowledge, our findings of the absence of p63 and WT-1 expression in a vast majority of the myoepithelial cells of acini and TDLU, cytoplasmic localization of p63 in the entire epithelial cell population of some lobules, increasing WT-1 expression in vascular structures of the invasive tissue component, and substantially different p63 and WT-1 expression between ducts and acini or TDLU, in PABC, however, have not been previously reported.
The absence of p63 and WT-1 expression in most myoepithelial cells in acini and TDLU of PABC is likely to result from two correlated mechanisms: (A) Genetic defects in myoepithelial cell self-renewal-related genes. As both p63 and WT-1 play significant roles in the early developmental of several organs and systems [27-29], it is possible that at the early stage of breast morphogenesis, the breasts of these patients may have exposed to certain external or internal insults that may have caused permanent damages in DNA structures of some primitive stem cells. These DNA structural damages resulted in the inactivation of, or defects in, myoepithelial cell renewal-related genes, which impaired the myoepithelial cell replenishment process to replace the aged or injured myoepithelial cells, resulting in a “senesced” myoepithelial cell population with significantly reduced p63 and WT-1 expression. Consistent with this possibility is the fact that the expression of maspin, a tumor suppressor that is not involved in early development [6], is not affected in all PABC cases in this study. (B) Simultaneous activation and proliferation of multiple tumor stem cells. The DNA structural damages may have also caused the inactivation of, or defects in, cell cycle control-and apoptosis-related genes in the epithelial cell population, which allow these cells to proliferate to form different acinar clusters or TDLU with the same genetic defects. These acinar clusters or TDLU may progress rapidly after external or internal insults, leading to the early occurrence of breast cancer at young ages. On the other hand, these clusters may become maturation-arrested after a few cycles of cells divisions, but remain the potential for unlimited proliferation and multi-lineage differentiation. During the last few weeks of pregnancy, these clusters are activated and simultaneously proliferating in responding to a sharp increase of estrogen, progesterone, and other hormones [21-23]. As the myoepithelial cell population lacks the receptors for these hormones, it may not be able to undergo vigorous proliferations to match the dramatic increase of epithelial cells, which could result in the over-stretch or even focal disruptions of the myoepithelial cell layers. Consistent with this possibility is the following facts: (1) the proliferation index is significantly higher in acini and TDLU with aberrant p63 and WT-1 expression, (2) the morphological and immunohistochemical profiles differ substantially among lobules, and (3) the entire cell population of some morphologically normal appearing lobules shared the same malignant cytological features and proliferation index with their adjacent invasive counterparts (as shown in Fig 7). This possibility is also supported by recent studies in human prostate tumors, which detected a DNA phenotype that is identical to that of invasive cancer in some “healthy” men, and in morphologically normal prostate tissues adjacent to prostate cancer [30-33].
Cytoplasmic p63 expression in the entire epithelial cell population of some lobules is likely to results from aberrant transportation of the p63 protein. Similar to the p53 protein, p63 is synthesized in the cytoplasm and constantly transported through the nuclear pore complex into the nuclei [34-35]. A number of factors, including alterations of the elements and molecules involved in p63 nucleo-cytoplasmic transportation could significantly impact the p63 cellular trafficking, and consequently alter its sub-cellular localization [34-35]. A previous study from our lab showed that only cells exhibiting secretory changes or secretory carcinomas had cytoplasmic p63 expression [36]. It is possible that the secretory products in PABC may form protein-protein complexes with p63, which prevent the import of p63 protein into the nuclei. Consistent with this possibility is the fact that some proteins could form protein-protein complexes with p53, which results in accumulation of p53 in the cytoplasm [37-38]. Although the clinical significance of cytoplasmic p63 expression in human breast is unknown, a similar morphological and immunohistochemical profile between cytoplasmic p63 expressing hyperplastic and adjacent invasive cancer cells (as shown in Figs. 5g-5h) suggests the possibility of direct transformation of hyperplastic cells to invasive lesions. Consistent with this possibility are the facts that cutoplasmic localization of p63 is associated with poor patient survival in lung cancer and is also associated with tumor grade in meningiomas [39-40].
The differential WT-1 expression in myoepithelial and endothelial cells of the same case could potentially result from focal degeneration of aged or injured myoepithelial cells and the resultant auto-immunoreactions for the following reasons. First, myoepithelial cells belong to a self-renewal population that has to consistently proliferate and differentiate to replace the aged and injured myoepithelial cells. Any internal and external insults that impair the replenishment process could result in a “senesced” myoepithelial cell population that is morphologically distinct, whereas may loss the expression of tumor suppressors and other markers. Second, the degradation products of degenerated myoepithelial cells and the diffusible molecules secreted by the overlying tumor cells could function as self-epitopes to attract the infiltration of immunoreactive cells, resulting in localized chronic inflammation, which could directly incite angiogenesis or cause cellular and genomic damages [41-42]. WT-1 expression in endothelial cells could potentially result from or reflect the recapitulation of the early events of morphogenesis, which undergo extensive cell proliferation and vascularization. As the WT-1 encodes a transcription factor that binds to GC-rich sequences and regulates the expression of several genes of the growth factor family [43-47], it is likely that WT-1 might also regulate the expression of the vascular growth factor. Consistent with our speculation is the fact that the normal development and tumor invasion share the same mechanism for epithelial-branching and migration. It is also possible that, elevated WT-1 expression in endothelial cells may result from alternative splicing that results in an isoform of WT-1 mRNA, which has high affinity to bind to the vascular growth factor receptor. Consistent with our speculation is the fact that a recent study has shown that WT-1 17AA(-)/KTS isoform could induce morphological changes, and promotes cell migration and invasion in vitro, whereas other isoforms failed to induce similar changes under the same condition [48].
The specific cellular or molecular mechanism accounting for a potential correlation between tumor suppressor expression in myoepithelial cells and biological behavior in tumor cells is unknown, but is likely to result from the resultant consequences of aberrant tumor suppressor expression and focal disruptions in the myoepithelial cell layers. As the epithelium is normally devoid of both blood vessels and lymphatic ducts, and thus, totally relies on the stroma for its needed materials for normal functions and even survival, a focal myoepithelial cell layer disruption (FMCLD) could have several consequences, including (1) an increase of permeability for oxygen, nutrients, and growth factors, which selectively triggers the exit of stem or progenitor cells from quiescence [49-50], (2) the exposure of luminal cells to different cytokines, which facilitates vasculogenic mimicry and tumor angiogenesis [51-52], (3) The physical contact between luminal and stromal cells, which augments the expression of stromal MMP and facilitates epithelial-mesenchymal transition (EMT) and cell motility [53-54], and (4) The physical contact between luminal and immunoreactive cells, which directly cause genomic or cellular damages that trigger a cascade reaction of malignant transformation [41-42]. These alterations could individually or collectively trigger elevated proliferation in acinar clusters with aberrant tumor suppressor expression or with focally disrupted myoepithelial cell layers, which leads to the enlargement of FMCLD and stretched-out of the residual myoepithelial cells. Eventually, the entire epithelial cell population in these clusters completely loses their surrounding structures and transform directly to a new microenvironment that favors epithelial- mesenchymal transition and cell motility. Although these cells might not possess all the properties of invasive cancer cells, the changed microenvironment may act as the second “hit” to trigger a cascade reaction of malignant transformation [22-23]. As some of those acinar clusters with focally disrupted myoepithelial cell layers may represent a population of mutation-arrested stem cells and retain the potential for unlimited proliferation and multi-lineage differentiation, they may rapidly progress to invasive or metastatic breast lesions. In addition, hyperplastic acinar cells with cytoplasmic p63 expression may directly transform into invasive lesions.
No definite conclusions, however, could be drawn at present, since our sample size is small, and the clinical follow-up data are not available. On the other hand, if confirmed by further studies, our findings are likely to have several significant scientific and clinical implications. First, as a vast majority of the breast malignancies are originated from TDLU, the development of more applicable methods to quantitatively assess the expression of p63 and WT-1 expression in this site may be more effective than calponin immunostaining for early detection of breast cancer. Second, the elucidation of the mechanism of cytoplasmic p63 expression may lead to the development of new therapeutic agents to target cancer-related molecules in the cytoplasm or nuclei. Third, the development of different agents to target WT-1 in endothelial cells may have significant therapeutic value. Fourth, the development therapeutic agent to stimulate myoepithelial cell self-renewal or to prevent myoepithelial cell degradation may be more effective than the traditional approaches for breast cancer intervention and prevention.
Acknowledgements
This study was supported in part by grant 2006CB910505 from the Ministry of Chinese Science and Technology Department, grant 30801176 from The National Natural Science Foundation of China, grants DAMD17-01-1-0129, DAMD17-01-1-0130, PC051308 from Congressionally Directed Medical Research Programs, and grant BCTR0706983 from The Susan G. Komen Breast Cancer Foundation.
The opinions and assertions contained herein represent the personal views of the authors and are not to be construed as official or as representing the views of the Department of the Army or the Department of Defense.
Conflict of Interest
The authors have declared that no conflict of interest exists.
References
1. Guelstein VI, Tehypysheva TA, Ermilova VD, Liubimov AV. Myoepithelial and besement membrane antigens in benign and malignant human breast tumors. In J Cancer. 1993;53:269-77
2. Miosge N. The ultrastructural composition of basement membrane in vivo. Histol Histopathol. 2001;16:1239-48
3. Jolicoeur F, Seemayer TA, Gabbiani G. Multifocal, nascent, and invasive myoepithelial carcinoma (malignant myoepithelioma) of the breast: an immunohistochemical and ultrastructural study. Int J Surg Pathol. 2002;10:281-91
4. Gottlieb C, Raiu U, Greenwald KA. Myoepithelial cells in the differential diagnosis of complex benign and malignant breast lesions: an immunohistochemical study. Mod Pathol. 1990;3:135-40
5. Man YG, Sang QXA. The significance of focal myoepitehlial cell layer disruptions in breast tumor invasion: a paradigm shift from the “protease-centered” hypothesis. Exp Cell Res. 2004;301:103-18
6. Zou Z, Anisowicz A, Hendrix MJ, Thor A, Neveu M, Sheng S, Rafidi K, Seftor E, Sager R. Maspin, a serpin with tumor-suppressing activity in human mammary epithelial cells. Science. 1994;263:526-29
7. Barbareschi M, Pecciarini L, Cangi MG. et al. p63, a p53 homologue, is a selective nuclear marker of myoepithelial cells of the human breast. Am J Surg Pathol. 2001;25:1954-60
8. Sternlight MD, Barsky SH. The myoepithelial defense: a host defense against cancer. Med Hypotheses. 1997;48:37- 46
9. Li JH, Man YG. Dual usages of single wilms' tumor 1 immunohistochemistry in evaluation of breast tumors. Cancer Biomarkers. in press
10. Zhang R, Man YG, Vang RS, Saenger JS, Barner R, Wheeler D, Liang CY, Vinh TN, Bratthauer GL. A subset of morphologically distinct mammary myoepithelial cells lacks corresponding immunophenotypic markers. Breast Cancer Res. 2003;5:R151-6
11. Man YG, Tai L, Barner R, Vang R, Saenger JS, Shekitka KM. et al. Cell clusters overlying focally disrupted mammary myoepithelial cell layers and adjacent cells within the same duct display different immunohistochemical and genetic features: implications for tumor progression and invasion. Breast Cancer Res. 2003;5:R231-241
12. Yousefi M, Mattu R, Gao C, Man YG. Mammary ducts with and without focal myoepithelial cell layer disruptions show a different frequency of white blood cell infiltration and growth pattern: Implications for tumor progression and invasion. AIMM. 2005;13:30-37
13. Man YG, Zhang Y, Shen T, Vinh TN, Zeng X, Tauler J, Mulshine JL, Strauss BL. cDNA expression profiling identifies elevated expressions of tumor progression and invasion related genes in cell clusters of in situ breast tumors. Breast Cancer Res Treat. 2005;89:199-208
14. Man YG, Shen T, Weisz J, Berg PE, Schwartz AM, Mulshine JL, Sang QXA, Nieburgs HE. A subset of in situ breast tumor cell clusters lacks expression of proliferation and progression related markers but shows signs of stromal and vascular invasion. Cancer Detect Prev. 2005;29:323-31
15. Man YG, Zhao CQ, Wang J. Breast tumor cell clusters and their budding derivatives show different immunohistochemical profiles during stromal invasion: implications for hormonal and drug therapies. Cancer Therapy. 2006;4:193-204
16. Man YG, Nieburgs HE. A subset of cell clusters with malignant features in morphologically normal and hyperplastic breast tissues. Cancer Detect Prev. 2006;30(3):239-47
17. Man YG. Focal degeneration of aged or injured myoepithelial cells and the resultant auto- immunoreactions are trigger factors for breast tumor invasion. Medical Hypotheses. 2007;69(6):1340-57
18. Zhang XC, Hashemi SS, Yousefi M, Gao CL, Sheng J, Mason J, Man YG. Atypical expression of c-erbB2 in cell clusters overlying focally disrupted breast myoepithelial cell layers: a potential sign for increasing cell motility and invasion. Int J Biol Scien. 2008;4:259-69
19. Man YG. Bad seeds produce bad crops: a single step-process of breast carcinogenesis and Progression. Bioscience Hypotheses. 2008;1:147-55
20. Mathelin C, Annane K, Treisser A, Chenard MP, Tomasetto C, Bellocq JP, Rio MC. Pregnancy and post-partum breast cancer: a prospective study. Anticancer Res. 2008;28(4C):2447-52
21. Rodriguez AO, Chew H, Cress R, Xing G, McElvy S, Danielsen B, Smith L. Evidence of poorer survival in pregnancy-associated breast cancer. Obstet Gynecol. 2008;112(1):71-8
22. Polyak K. Pregnancy and breast cancer: the other side of the coin. Cancer Cell. 2006;9(3):151-3
23. Hu M, Polyak M. Molecular characterization of the tumor microenvironment in breast cancer. Eur J Cancer. 2008 [Epub ahead of print]
24. Tavassoli FA, Man YG. Morphofunctional features of intraductal hyperplasia, atypical hyperplasia, and various grades of intraductal carcinoma. The Breast J. 1995;1(3):155-62
25. Wagner N, Michiels JF, Schedl A, Wagner KD. The Milms' tumour suppressor WT1 is involved in endothelial cell proliferation and migration:expression in tumo vessels in vivo. Oncogene. 2008;27:3662-72
26. Tomkova K, Tomka M, Zajae V. Contribution of p53, p63, and p73 to the developmental diseases and cancer. Neoplasm. 2008;55(3):177-81
27. Kurita T, Cunha GR, Robboy SJ, Mills AA, Medina RT. Differential expression of p63 in female reproductive organs. Mech Dev. 2005;122(9):1043-55
28. Scharnhorst V, van der Eb AJ, Jochemsen AG. WT1proteins: functions in growth and differentiation. Gene. 2001;273(2):141-61
29. Hirose M. The role of Wilms' tumor genes. J Med Invest. 1999;46(3-4):130-40
30. Malins DC, Polissar NL, Su Y, Gardner HS, Gunselman SJ. A new structural analysis of DNA using statistical models of infrared sepctra. Nat Med. 1997;3:927-30
31. Malins DC, Johnson PM, Barker EA. et al. Cancer-related changes in prostate DNA as men age and early identification of metastasis in prostate tumors. Proc Natl Acad Sci USA. 2003;100:5401-6
32. Malins DC, Anderson KM, Gilman NK. et al. Development of a cancer DNA phenotype prior to tumor formation. Proc Natl Acad Sci Sci USA. 2004;101:10721-5
33. Malins DC, Gilman NK, Green VM. et al. A DNA phenotype in healthy prostates, conserved in tumors and adjacent normal cells, implies a relationship to carcinogenesis. Proc Natl Acad Sci USA. 2005;102:19093-6
34. Barbieri CE, Pietenpol JA. p63 and epithelial biology. Exp Cell Res. 2006;312(6):695-706
35. Westfall MD, Pietenpol JA. p63: Molecular complexity in development and cancer. Carcinogenesis. 2004;25(6):857-64
36. Bratthauer GL, Saenger JS, Strauss BL. Antibodies targeting p63 react specifically in the cytoplasm of breast epithelial cells exhibiting secretory differentiation. Histopathology. 2005;47(6):611-16
37. Liang SH, Clarke MF. Regulation of p53 localization. Eur J Biochem. 2001;268(10):2779-83
38. O'Keefe K, Li H, Zhang Y. Nucleocytoplasmic shuttling of p53 is essential for MDM2-mediated cytoplasmic degradation but not ubiquitination. Mol Cell Biol. 2003;23(18):6396-405
39. Narahashi T, Niki T, Wang T, Goto A, Matsubara D, Funata N, Fukayama M. Cytoplasmic localization of p63 is associated with poor patient survival in lung adenocarcinoma. Histopathology. 2006;49(4):349-57
40. Rushing EJ, Olsen C, Man YG. Correlation of p63 immunoreactivity with tumor grade in meningiomas. Int J Surg Pathol. 2008;16(1):38-42
41. Peek RM Jr, Mohla S, DuBois RN. Inflammation in the genesis and perpetuation of cancer: summary and recommendations from a national cancer institute-sponsored meeting. Cancer Res. 2005;65:8583-6
42. MacLennan GT, Eisenberg R, Fleshman RL. et al. The influence of chronic inflammation in prostatic carcinogenesis: a 5-year follow-up study. J Urol. 2006;176:1012-6
43. Scharnhorst V, van der Eb AJ, Jochemsen AG. WT1 proteins: functions in growth and differentiation. Gene. 2001;273(2):141-61
44. Re GG, Hazen-Martin DJ, Sens DA, Garvin AJ. Nephroblastoma (Wilms' tumor): a model system of aberrant renal development. Semin Diagn Pathol. 1994;11(2):126-35
45. Haber DA, Buckler AJ. WT1: a novel tumor suppressor gene inactivated in Wilms' tumor. New Biol. 1992;4(2):97-106
46. Armstrong JF, Pritchard-Jones K, Bickmore WA, Hastie ND, Bard JB. The expression of the Wilms' tumor gene, WT1, in the developing mammalian embryo. Mech Dev. 1993;40(1-2):85-97
47. Scharnhorst V, Menke AL, Attema J, Haneveld JK, Riteco N, van Steenbrugge GL, der Eb AJ, Jochemsen AG. EGR-1 enhances tumor growth and modulates the effect of the Wilms' tumor 1 gene products on tumorgenicity. Oncogene. 2000;19(6):791-800
48. Jomgeow T, Oji Y, Tsuji N, Ikeda Y, Ito K, Tsuda A. et al. Wilms' tumor gene WT1 17AA(-)/KTS(-) isoform induces morphological changes and promotes cell migration and invasion in vitro. Cancer Sci. 2006;97:259-70
49. Chakravarthy MV, Spangenhurg EE, Booth FW. Culture in low levels of oxygen enhances in vitro proliferation potential of satellite cells from old skeletal muscles. Cell Mol Life Sci. 2001;58:1150-58
50. Csete M, Walikonis J, Slawny N. et al. Oxygen-mediated regulation of skeletal muscle satellite cell proliferation and adipogrnesis in culture. J Cell Physical. 2001;189:189-96
51. Klos KS, Wyszomierski SL, Sun M. et al. c-erbB2 increases vascular endothelial growth factor protein synthesis via activation of mammalian target of rapamycin/p70S6K leading to increased angiogenesis and spontaneous metastasis of human breast cancer cells. Cancer Res. 2006;64:2028-37
52. Hendrix MJ, Seftor EA, Hess AR, Seftor RE. Vasculogenic mimicry and tumor-cell plasticity: lessons from melanoma. Nat Rev Cancer. 2003;3(6):411-21
53. Kang Y, Massague J. Epithelial-mesenchymal transition: twist in development and metastasis. Cell. 2004;118:277-9
54. Sato T, Sakai T, Noguchi Y, Takita M, Hirakawa S, Ito A. Tumor-stromal cell contact promotes invasion of human uterine cervical carcinoma cells by augmenting the expression and activation of stromal matrix metalloproteinases. Gynecol Oncol. 2004;92:47-56
Author contact
![]() Correspondence to: Yan-gao Man, MD., PhD. Director of Gynecologic and Breast Research Laboratory Department of Gynecologic and Breast Pathology Armed Forces Institute of Pathology and American Registry of Pathology 6825 16th Street, NW Washington DC 20306-6000 Tel: 202-782-1612; Fax: 202-782-3939; E-mail: manosd.mil
Correspondence to: Yan-gao Man, MD., PhD. Director of Gynecologic and Breast Research Laboratory Department of Gynecologic and Breast Pathology Armed Forces Institute of Pathology and American Registry of Pathology 6825 16th Street, NW Washington DC 20306-6000 Tel: 202-782-1612; Fax: 202-782-3939; E-mail: manosd.mil
Received 2008-12-23
Accepted 2009-1-8
Published 2009-1-9
