ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2012; 8(5):685-696. doi:10.7150/ijbs.4401 This issue Cite
Research Paper
Interactions of Several Lipid-Related Gene Polymorphisms and Cigarette Smoking on Blood Pressure Levels
1. Department of Cardiology, Institute of Cardiovascular Diseases, the First Affiliated Hospital, Guangxi Medical University, Nanning, Guangxi, People's Republic of China;
2. Department of Cardiology, the Third Affiliated Hospital, Guangxi Medical University, Nanning, Guangxi, People's Republic of China;
3. Department of Cardiology, the People's Hospital of Guilin, Guilin, Guangxi, People's Republic of China;
4. Department of Internal Medicine, Worker's Hospital of Guangxi Liuzhou Iron and Steel (Group) Company, Liuzhou, Guangxi, People's Republic of China.
Abstract
The interactions of single nucleotide polymorphisms (SNPs) and cigarette smoking on blood pressure levels are limited. The present study was undertaken to detect nine lipid-related SNPs and their interactions with cigarette smoking on blood pressure levels. Genotyping of ATP-binding cassette transporter A1 (ABCA-1) V825I, acyl-CoA:cholesterol acyltransferase-1 (ACAT-1) rs1044925, low density lipoprotein receptor (LDL-R) AvaⅡ, hepatic lipase gene (LIPC) -250G>A, endothelial lipase gene (LIPG) 584C>T, methylenetetrahydrofolate reductase (MTHFR) 677C>T, proprotein convertase subtilisin-like kexin type 9 (PCSK9) E670G, peroxisome proliferator-activated receptor delta (PPARD) +294T>C, and Scavenger receptor class B type 1 (SCARB1) rs5888 was performed in 935 nonsmokers and 845 smokers. The interactions were detected by factorial regression analysis. The frequencies of genotypes (ACAT-1 and LIPG), alleles (ABCA-1), and both genotypes and alleles (LDL-R, LIPC, PPARD and SCARB1) were different between nonsmokers and smokers (P < 0.05-0.001). The levels of pulse pressure (PP, ABCA-1), and systolic, diastolic blood pressure (SBP, DBP) and PP (LIPC) in nonsmokers were different among the genotypes (P < 0.01-0.001). The levels of SBP (ABCA-1, ACAT-1, LIPG and PCSK9), DBP (ACAT-1, LDL-R, LIPC, PCSK9 and PPARD), and PP (LIPC, LIPG, MTHFR and PCSK9) in smokers were different among the genotypes (P < 0.01-0.001). The SNPs of ABCA-1, ACAT-1 and PCSK9; ACAT-1, LDL-R, MTHFR and PCSK9; and ABCA-1, LIPC, PCSK9 and PPARD were shown interactions with cigarette smoking to influence SBP, DBP and PP levels (P < 0.05-0.001); respectively. The differences in blood pressure levels between the nonsmokers and smokers might partly result from different interactions of several SNPs and cigarette smoking.
Keywords: blood pressure, hypertension, genetic polymorphism, cigarette smoking, interaction.
Introduction
Hypertension is a complex disease that affects 1 billion people and causes more than 7.1 million premature deaths a year worldwide [1]. It is well-recognized that blood pressure is modulated by multiple genetic loci as well as environmental and lifestyle factors including diet, smoking habits, obesity and physical inactivity [2,3], and their interactions [4-9]. Many previous genome-wide association studies (GWAS) in different populations have simultaneously explored a large number of genomic loci associated with blood pressure and hypertension, but the results of these association studies conducted with blood pressure traits have been inconsistent [10-18]. A major reason for inconsistency among studies may be different environmental modifiers that interact with genes to influence blood pressure and hypertension.
Although the deleterious effects of cigarette smoking on the cardiovascular system have been clearly documented, the relationship between cigarette smoking and blood pressure and hypertension has not been fully characterized [19]. Some studies have reported that cigarette smoking is associated with increased blood pressure [20-22], others reported associations with decreased blood pressure [23-25], while some found no effect at all [26,27]. However, cigarette smoking has proven to be an important mediator of genetic effects in association studies of blood pressure and hypertension [4-9].
There are 56 ethnic groups in China. Yao is the eleventh largest minority among the 55 minority groups according to the population size. Bai Ku Yao (White-trouser Yao), an isolated subgroup of the Yao minority, is named so because all of men wear white knee-length knickerbockers. The population size is about 30 000. Because of isolation from the other ethnic groups, the special customs and cultures including their clothing, intra-ethnic marriages, dietary patterns, and corn wine and rum intakes are still completely preserved to the present day. In previous epidemiological studies, we found that the prevalence of hypertension was higher in smokers than in nonsmokers [2,3]. Since hypertension is found to occur more often than expected in families with familial combined hyperlipidemia and other types of familial lipid syndromes [28], genes involved in lipid metabolism may be involved in the genetic component of the development of hypertension. Therefore, the aim of the present study was to detect nine single nucleotide polymorphisms (SNPs) in different lipid-related genes and evaluate their interactions with cigarette smoking on blood pressure levels in the Guangxi Bai Ku Yao population.
Materials and Methods
Subjects
A total of 1780 unrelated subjects of Bai Ku Yao who reside in Lihu and Baxu villages in Nandan County, Guangxi Zhuang Autonomous Region, People's Republic of China were randomly selected from our previous stratified randomized cluster samples [2,3]. The age of the subjects ranged from 15 to 80 years, with an average age of 41.60 ± 16.43 years. There were 935 nonsmokers (285 males and 650 females) and 845 smokers (709 males and 136 females). All of the subjects were rural agricultural workers. The subjects had no evidence of diseases related to atherosclerosis, coronary artery disease and diabetes. None of them had been treated with antihypertensive drugs, such as nifedipine and/or catopril and lipid-lowering drugs. The study design was approved by the Ethics Committee of the First Affiliated Hospital, Guangxi Medical University. All participants have given written informed consent.
Epidemiological survey
The survey was done according to standardized methods. Information on personal and demographic characteristics, personal and family history of disease, current pharmacotherapy, cigarette smoking, alcohol consumption, and physical activity was obtained at interview. Self-reported information on cigarette smoking was recorded. All participants were asked, “Have you smoked at least 100 cigarettes in your entire life?” and “Do you now smoke cigarettes every day, some days or not at all?” Smoking status was re-coded as: “Smokers” or “current smokers” if the respondents reported having smoked at least 100 cigarettes in their lifetime and were current smokers either every day or some days; “nonsmokers” or “never smokers” if they reported they had not smoked at least 100 cigarettes in their lifetime; and “former smokers” if they reported having smoked 100 cigarettes in their lifetime but did not currently smoke [29,30]. “Former smokers” were not included in this study. Sitting blood pressure was measured three times by a well-trained physician with the use of a standard mercury sphygmomanometer after the subject rested 5 minutes, and the average of the three measurements was used for the level of blood pressure. Systolic blood pressure (SBP) was determined by the first Korotkoff sound, and diastolic blood pressure (DBP) by the fifth Korotkoff sound. Pulse pressure (PP) was calculated as the systolic minus the diastolic blood pressure. Body weight, to the nearest 50 grams, was measured using a portable balance scale. Subjects were weighed without shoes and in a minimum of clothing. Height was measured, to the nearest 0.5 cm, using a portable steel measuring device. From these two measurements body mass index (BMI, kg/m2) was calculated. Waist circumference was measured with a nonstretchable measuring tape, at the level of the smallest area of the waist, to the nearest 0.1 cm.
Measurements of lipids and apolipoproteins
A venous blood sample of 5 mL was obtained from all subjects after at least 12 hours of fasting. A part of the sample (2 mL) was collected into glass tubes and used to determine serum lipid levels. Another part of the sample (3 mL) was transferred to tubes with anticoagulate solution and used to extract deoxyribonucleic acid (DNA). The levels of total cholesterol (TC), triglyceride (TG), high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C) in samples were determined by enzymatic methods. Serum apolipoprotein (Apo) A1 and ApoB levels were detected by the immunoturbidimetric immunoassay. All determinations were performed with an autoanalyzer (Type 7170A; Hitachi Ltd., Tokyo, Japan) in our Clinical Science Experiment Center [31,32].
Genotyping
Genomic DNA was extracted from the peripheral blood leukocytes by the phenol-chloroform method as our previous reports [33-40]. Genotyping of ATP-binding cassette transporter A1 (ABCA-1) V825I (rs2066715), acyl-CoA:cholesterol acyltransferase-1 (ACAT-1) rs1044925, low density lipoprotein receptor (LDL-R) AvaⅡ, hepatic lipase gene (LIPC) -250G>A (rs2070895), endothelial lipase gene (LIPG) 584C>T (rs2000813), methylenetetrahydrofolate reductase (MTHFR) 677C>T (rs1801133), proprotein convertase subtilisin-like kexin type 9 (PCSK9) E670G (rs505151), peroxisome proliferator-activated receptor delta (PPARD) +294T>C (rs2016520) and Scavenger receptor class B type 1 (SCARB1) rs5888 was performed using polymerase chain reaction and restriction fragment length polymorphism (PCR-RFLP). The sequences of the forward and backward primers and restriction ezyme used for the genotyping of nine SNPs, the thermocycling protocol, the approach to electrophoresis, and the procedures for quality control have been described previously [33-40]. Genotypes were scored by an experienced reader blinded to epidemiological data and blood pressure levels.
Diagnostic criteria
Hypertension was defined as an average SBP of 140 mmHg or greater and/or an average DBP of 90 mmHg or greater [2,3]. The normal values of serum TC, TG, HDL-C, LDL-C, ApoA1 and ApoB levels, and the ratio of ApoA1 to ApoB in our Clinical Science Experiment Center were 3.10-5.17, 0.56-1.70, 0.91-1.81, 2.70-3.20 mmol/L, 1.00-1.78, 0.63-1.14 g/L, and 1.00-2.50; respectively [31,32].
Statistical analysis
Quantitative variables were presented as mean ± SD or percentage. All statistical analyses were done with the statistical software package SPSS 13.0 (SPSS Inc., Chicago, Illinois, USA). Chi square tests were used to compare the differences in percentages and to assess Hardy-Weinberg expectations, whereas analysis of covariance (ANOVA) was used to compare the differences in various continuous variables between respective comparison groups. The sex, age, education level, physical activity, BMI, waist circumference, alcohol consumption, and hyperlipidemia were adjusted for the statistical analyses. All significant associations were corrected for multiple testing by applying a Bonferroni correction. The allelic and genotypic frequencies were calculated from the observed genotypic counts. The interactions of nine SNPs and cigarette smoking on blood pressure levels were assessed by using a factorial regression analysis after controlling for potential confounders including sex, age, education level, physical activity, BMI, waist circumference, alcohol consumption, and hyperlipidemia. Multiple linear regression was used to ascertain the correlation between genotypes (ABCA-1 V825I: GG = 1, GA = 2, AA = 3; ACAT-1 rs1044925: AA = 1, AC = 2, CC = 3; LDL-R AvaⅡ: A-A- = 1, A-A+ = 2, A+A+ = 3; LIPC -250G>A: GG = 1, GA = 2, AA = 3; LIPG 584C>T: CC = 1, CT = 2, TT = 3; MTHFR 677C>T: CC = 1, CT = 2, TT = 3; PCSK9 E670G: AA = 1, AG = 2; PPARD +294T>C: TT = 1, TC = 2, CC = 3; SCARB1 rs5888: CC = 1, CT = 2, TT = 3) or alleles (the minor allele noncarrier = 1, the minor allele carrier = 2) and blood pressure levels in the combined population of nonsmokers and smokers, nonsmokers, and smokers; respectively.
Results
General characteristics between nonsmokers and smokers
As shown in Table 1, the levels of average age, height, weight, waist circumference, SBP, DBP, hypertensive prevalence, TC, TG, HDL-C, ApoA1; the ratio of ApoA1 to ApoB; the percentages of subjects who consumed alcohol; and the ratio of males to females were higher in smokers than in nonsmokers (P < 0.05-0.001). There were no significant differences in the levels of education, BMI, PP, LDL-C and ApoB between smokers and nonsmokers (P > 0.05 for all).
The general characteristics and blood pressure levels between the nonsmokers and smokers.
| Characteristics | Nonsmoker (n = 935) | Smoker (n = 845) | t (χ2) | p |
|---|---|---|---|---|
| Age (years) | 39.75±16.31 | 43.64±16.32 | -5.023 | 0.000 |
| Male/female | 285/650 | 709/136 | 513.751 | 0.000 |
| Education level (years) | 6.20±3.26 | 6.31±3.16 | 0.721 | 0.471 |
| Height (cm) | 151.73±7.39 | 158.13±7.28 | -18.378 | 0.000 |
| Weight (kg) | 51.40±8.09 | 55.07±8.50 | -9.311 | 0.000 |
| Body mass index (kg/m2) | 22.30±2.90 | 22.10±2.53 | 1.559 | 0.119 |
| > 24 kg/m2 [n(%)] | 223 (23.9) | 134 (15.9) | 17.683 | 0.000 |
| Waist circumference (cm) | 71.84±8.84 | 74.23±8.12 | 5.920 | 0.000 |
| Alcohol consumption [n (%)] | 246 (26.3) | 602 (71.2) | 361.471 | 0.000 |
| Systolic blood pressure (SBP, mmHg) | 119.20±17.31 | 122.78±15.50 | -4.606 | 0.000 |
| SBP ≥ 140 mmHg [n (%)] | 110 (11.8) | 121 (14.3) | 2.565 | 0.109 |
| Diastolic blood pressure (DBP, mmHg) | 74.87±10.37 | 77.63±9.47 | -5.882 | 0.000 |
| DBP ≥ 90 mmHg [n (%)] | 111 (11.9) | 106 (12.5) | 0.188 | 0.665 |
| Pulse pressure (mmHg) | 44.34±11.96 | 45.16±11.77 | -1.463 | 0.144 |
| Hypertensive prevalence [n (%)] | 151 (16.1) | 169 (20.0) | 4.462 | 0.035 |
| Total cholesterol (mmol/L) | 4.48±0.93 | 4.60±1.06 | -2.653 | 0.008 |
| Triglycerides (mmol/L) | 1.23±1.08 | 1.38±1.24 | -2.686 | 0.007 |
| HDL-C (mmol/L) | 1.74±0.46 | 1.84±0.49 | -4.153 | 0.000 |
| LDL-C (mmol/L) | 2.59±0.69 | 2.59±0.87 | -0.101 | 0.920 |
| Apolipoprotein (Apo) A1 (g/L) | 1.33±0.29 | 1.41±0.33 | -5.694 | 0.000 |
| ApoB (g/L) | 0.86±0.22 | 0.88±0.23 | -1.646 | 0.101 |
| ApoA1/ApoB | 1.64±0.59 | 1.73±0.74 | -2.878 | 0.004 |
Genotypic and allelic frequencies between nonsmokers and smokers
The genotypic and allelic frequencies of the nine SNPs between nonsmokers and smokers are shown in Table 2. The genotypic distribution of nine SNPs was consistent with the Hardy-Weinberg equilibrium. The genotypic and allelic frequencies of LDL-R, LIPC, PPARD and SCARB1 were different between nonsmokers and smokers (P < 0.05-0.001). The genotypic frequencies of ACAT-1 and LIPG, and the allelic frequencies of ABCA-1 were also different between nonsmokers and smokers (P < 0.05 for all). There were no significant differences in the genotypic and allelic frequencies of MTHFR and PCSK9 between nonsmokers and smokers (P < 0.05 for all). The GG homozygous of the PCSK9 E670G was not detected in our study population.
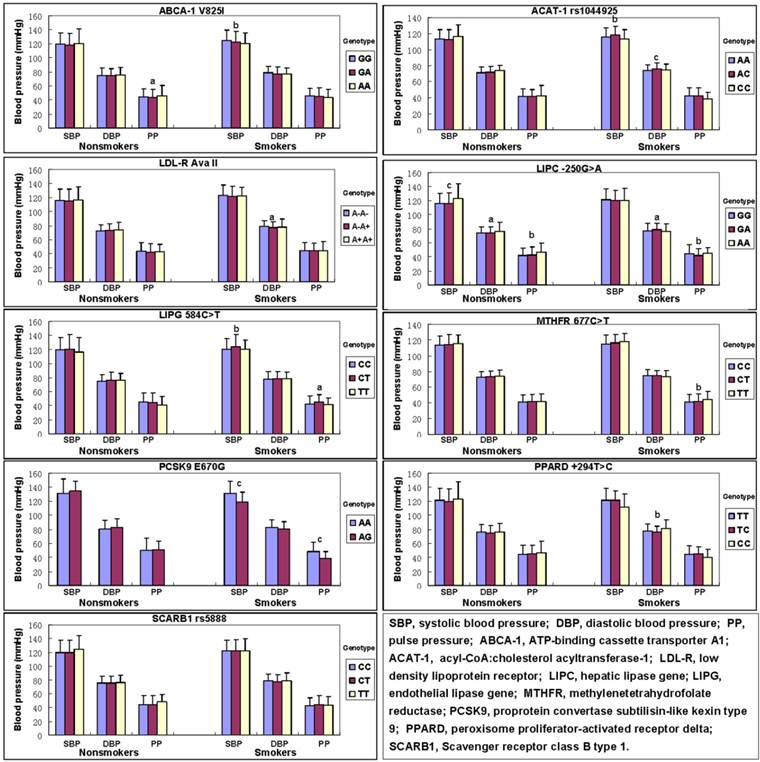
Genotypes and blood pressure levels between nonsmokers and smokers
The association of genotypes and blood pressure levels between nonsmokers and smokers is shown in Figure 1. The levels of PP in nonsmokers were different among the three genotypes of ABCA-1 (P < 0.01), and the levels of SBP, DBP and PP in nonsmokers were also different among the three genotypes of LIPC (P < 0.01-0.001).
For the smokers, the levels of SBP were different among the three genotypes of ABCA-1, ACAT-1, LIPG and PCSK9 (P < 0.01-0.001), the levels of DBP were different among the three genotypes of ACAT-1, LDL-R, LIPC, PCSK9 and PPARD (P < 0.01-0.001), and the levels of PP were different among the three genotypes of LIPC, LIPG, MTHFR and PCSK9 (P < 0.01-0.001).
Interactions of several SNPs and cigarette smoking on blood pressure levels
The interactions of nine SNPs and cigarette smoking on blood pressure levels are given in Table 3. The genotypes of ABCA-1 GG and GA and ACAT-1 AA and AC were shown interactions with cigarette smoking to increase SBP levels, whereas the genotype of PCSK9 AG was shown interactions with cigarette smoking to decrease SBP levels (P < 0.05-0.001). The genotypes of ACAT-1 AA and AC, LDL-R A-A-, A-A+ and A+A+, MTHFR CC and CT, and PCSK9 AA were shown consistent interactions with cigarette smoking to increase DBP levels (P < 0.05-0.01). The genotypes of ABCA-1 GA and LIPC GG were shown interactions with cigarette smoking to increase PP levels, whereas the genotypes of PCSK9 AA and AG, and PPARD CC were shown interactions with cigarette smoking to decrease PP levels (P < 0.05-0.001).
Correlation between genotypes or alleles and blood pressure levels
The results of multiple linear regression analysis between genotypes or alleles and blood pressure levels are shown in Table 4. For the nonsmokers, the levels of SBP were associated with the genotypes of LIPC (P < 0.01); the levels of DBP were associated with the genotypes and/or alleles of LIPG and PPARD (P < 0.05 for all); and the levels of PP were associated with the genotypes and/or alleles of LIPC (P < 0.05 for each).
For the smokers, the levels of SBP were correlated with the genotypes and/or alleles of ABCA-1, LIPG and PCSK9 (P < 0.01 for all), the levels of DBP were correlated with the genotypes and/or alleles of ABCA-1, ACAT-1, LDL-R and PCSK9 (P < 0.05-0.001), and the levels of PP were correlated with the genotypes and/or alleles of ABCA-1, LIPC, LIPG, MTHFR and PCSK9 (P < 0.05-0.001).
The genotypes of nine SNPs and blood pressure levels between the nonsmokers and smokers. aP < 0.01, bP < 0.005, and cP < 0.001.
The genotypic and allelic frequencies between nonsmokers and smokers.
| SNP | Genotype (Allele) | Nonsmoker (n = 935) | Smoker (n = 845) | P Value |
|---|---|---|---|---|
| ABCA-1 V825I | GG | 307(32.8) | 308(36.5) | |
| GA | 448(47.9) | 399(47.2) | ||
| AA | 180(19.3) | 138(16.3) | 0.146 | |
| G | 1062(56.8) | 1015(60.1) | ||
| A | 808(43.2) | 675(39.9) | 0.048 | |
| ACAT-1 rs1044925 | AA | 660(70.6) | 592(70.1) | |
| AC | 252(27.0) | 213(25.2) | ||
| CC | 23(2.4) | 40(4.7) | 0.030 | |
| A | 1572(84.1) | 1397(82.7) | ||
| C | 298(15.9) | 293(17.3) | 0.262 | |
| LDL-R AvaⅡ | A-A- | 468(50.1) | 378(44.7) | |
| A-A+ | 422(45.1) | 301(35.6) | ||
| A+A+ | 45(4.8) | 166(19.7) | 0.000 | |
| A- | 1358(72.6) | 1057(62.5) | ||
| A+ | 512(27.4) | 633(37.5) | 0.000 | |
| LIPC -250G>A | GG | 433(46.3) | 350(41.4) | |
| GA | 433(46.3) | 350(41.4) | ||
| AA | 69(7.4) | 145(17.2) | 0.000 | |
| G | 1299(69.5) | 1050(62.1) | ||
| A | 571(30.5) | 640(37.9) | 0.000 | |
| LIPG 584C>T | CC | 432(46.2) | 362(42.8) | |
| CT | 469(50.2) | 430(50.9) | ||
| TT | 34(3.6) | 53(6.3) | 0.024 | |
| C | 1333(71.3) | 1154(68.3) | ||
| T | 537(28.7) | 536(31.7) | 0.051 | |
| MTHFR 677C>T | CC | 431(46.1) | 416(49.2) | |
| CT | 437(46.7) | 367(43.5) | ||
| TT | 67(7.2) | 62(7.3) | 0.340 | |
| C | 1299(69.5) | 1199(70.9) | ||
| T | 571(30.5) | 491(29.1) | 0.335 | |
| PCSK9 E670G | AA | 892(95.4) | 793(93.8) | |
| AG | 43(4.6) | 52(6.2) | ||
| GG | 0(0) | 0(0) | 0.145 | |
| A | 1827(97.7) | 1638(96.9) | ||
| G | 43(2.3) | 52(3.1) | 0.151 | |
| PPARD +294T>C | TT | 517(55.3) | 496(58.7) | |
| TC | 347(37.1) | 313(37.0) | ||
| CC | 71(7.6) | 36(4.3) | 0.011 | |
| T | 1381(73.9) | 1305(77.2) | ||
| C | 489(26.1) | 385(22.8) | 0.020 | |
| SCARB1 rs5888 | CC | 534(57.1) | 408(48.3) | |
| CT | 367(39.3) | 371(43.9) | ||
| TT | 34(3.6) | 66(7.8) | 0.000 | |
| C | 1435(76.7) | 1187(70.2) | ||
| T | 435(23.3) | 503(29.8) | 0.000 |
SNP: single nucleotide polymorphism; ABCA-1: ATP-binding cassette transporter A1; ACAT-1: acyl-CoA:cholesterol acyltransferase-1; LDL-R: low density lipoprotein receptor; LIPC: hepatic lipase gene; LIPG: endothelial lipase gene; MTHFR: methylenetetrahydrofolate reductase; PCSK9: proprotein convertase subtilisin-like kexin type 9; PPARD: peroxisome proliferator-activated receptor delta; SCARB1: Scavenger receptor class B type 1.
Interactions of several single nucleotide polymorphisms and cigarette smoking on blood pressure levels.
| SNP | Genotype (F/P) | SBP | DBP | PP |
|---|---|---|---|---|
| ABCA-1 V825I | GG | ↑ | - | - |
| GA | ↑ | - | ↑ | |
| AA | - | - | - | |
| F | 3.918 | 1.168 | 3.739 | |
| P | 0.020 | 0.311 | 0.024 | |
| ACAT-1 rs1044925 | AA | ↑ | ↑ | - |
| AC | ↑ | ↑ | - | |
| CC | - | - | - | |
| F | 3.677 | 4.293 | 1.043 | |
| P | 0.026 | 0.014 | 0.353 | |
| LDL-R AvaⅡ | A-A- | - | ↑ | - |
| A-A+ | - | ↑ | - | |
| A+A+ | - | ↑ | - | |
| F | 0.458 | 4.717 | 0.950 | |
| P | 0.633 | 0.009 | 0.387 | |
| LIPC -250G>A | GG | - | - | ↑ |
| GA | - | - | - | |
| AA | - | - | - | |
| F | 0.761 | 1.424 | 4.514 | |
| P | 0.467 | 0.241 | 0.011 | |
| LIPG 584C>T | CC | - | - | - |
| CT | - | - | - | |
| TT | - | - | - | |
| 0.582 | 0.035 | 1.446 | ||
| 0.559 | 0.965 | 0.236 | ||
| MTHFR 677C>T | CC | - | ↑ | - |
| CT | - | ↑ | - | |
| TT | - | - | - | |
| F | 0.097 | 3.266 | 2.068 | |
| P | 0.908 | 0.038 | 0.127 | |
| PCSK9 E670G | AA | - | ↑ | ↓ |
| AG | ↓ | - | ↓ | |
| F | 18.610 | 3.847 | 15.401 | |
| P | 0.000 | 0.050 | 0.000 | |
| PPARD +294T>C | TT | - | - | - |
| TC | - | - | - | |
| CC | - | - | ↓ | |
| F | 0.919 | 1.322 | 4.738 | |
| P | 0.399 | 0.267 | 0.009 | |
| SCARB1 rs5888 | CC | - | - | - |
| CT | - | - | - | |
| TT | - | - | - | |
| F | 1.058 | 1.055 | 1.607 | |
| P | 0.347 | 0.349 | 0.201 |
SNP: single nucleotide polymorphism; ABCA-1: ATP-binding cassette transporter A1; ACAT-1: acyl-CoA:cholesterol acyltransferase-1; LDL-R: low density lipoprotein receptor; LIPC: hepatic lipase gene; LIPG: endothelial lipase gene; MTHFR: methylenetetrahydrofolate reductase; PCSK9: proprotein convertase subtilisin-like kexin type 9; PPARD: peroxisome proliferator-activated receptor delta; SCARB1: Scavenger receptor class B type 1; ↑: genotype and cigarette smoking interactions to increase blood pressure; ↓: genotype and cigarette smoking interactions to decrease blood pressure; -: no interaction of genotype and cigarette smoking on blood pressure.
Correlation between genotypes or alleles and blood pressure levels in the nonsmokers and smokers.
| BP | Genotype/allele | Unstandardized coefficient | Std. error | Standardized coefficient | t | P |
|---|---|---|---|---|---|---|
| Both | ||||||
| SBP | ABCA-1 V825I allele | -1.584 | 0.717 | -0.045 | -2.209 | 0.027 |
| LIPC -250G>A genotype | 2.137 | 0.488 | 0.090 | 4.376 | 0.000 | |
| LIPC -250G>A allele | 1.337 | 0.661 | 0.042 | 2.021 | 0.043 | |
| LIPG 584C>T allele | 1.790 | 0.730 | 0.051 | 2.453 | 0.014 | |
| MTHFR 677C>T genotype | 1.045 | 0.428 | 0.055 | 2.440 | 0.015 | |
| MTHFR 677C>T allele | 1.167 | 0.532 | 0.050 | 2.194 | 0.028 | |
| PCSK9 E670G genotype | -5.389 | 1.767 | -0.063 | -3.050 | 0.002 | |
| DBP | ACAT-1 rs1044925 genotype | 1.025 | 0.314 | 0.074 | 3.265 | 0.001 |
| ACAT-1 rs1044925 allele | 1.265 | 0.373 | 0.077 | 3.392 | 0.001 | |
| LIPC -250G>A genotype | 1.308 | 0.321 | 0.089 | 4.070 | 0.000 | |
| LIPC -250G>A allele | 1.118 | 0.435 | 0.056 | 2.570 | 0.010 | |
| LIPG 584C>T genotype | 1.034 | 0.390 | 0.057 | 2.655 | 0.008 | |
| LIPG 584C>T allele | 1.323 | 0.454 | 0.063 | 2.914 | 0.004 | |
| PPARD +294T>C allele | -1.025 | 0.459 | -0.050 | -2.231 | 0.026 | |
| PP | MTHFR 677C>T genotype | 0.739 | 0.360 | 0.470 | 2.053 | 0.040 |
| PCSK9 E670G genotype | -4.655 | 1.389 | -0.069 | 3.350 | 0.001 | |
| Nonsmoker | ||||||
| SBP | LIPC -250G>A genotype | 2.192 | 0.751 | 0.085 | 2.920 | 0.004 |
| DBP | LIPG 584C>T genotype | 1.195 | 0.562 | 0.063 | 2.214 | 0.034 |
| LIPG 584C>T allele | 1.466 | 0.637 | 0.069 | 2.303 | 0.022 | |
| PPARD +294T>C allele | -1.361 | 0.652 | -0.065 | -2.089 | 0.037 | |
| PP | LIPC -250G>A genotype | 1.838 | 0.572 | 0.099 | 3.211 | 0.001 |
| LIPC -250G>A allele | 1.544 | 0.716 | 0.067 | 2.158 | 0.031 | |
| Smoker | ||||||
| SBP | ABCA-1 V825I genotype | -2.077 | 0.671 | -0.094 | -3.098 | 0.002 |
| ABCA-1 V825I allele | -2.669 | 0.976 | -0.083 | -2.735 | 0.006 | |
| LIPG 584C>T allele | 2.560 | 0.963 | 0.081 | 2.659 | 0.008 | |
| PCSK9 E670G genotype | -11.804 | 2.139 | -0.164 | -5.519 | 0.000 | |
| DBP | ABCA-1 V825I genotype | -0.934 | 0.441 | -0.069 | -2.118 | 0.034 |
| ABCA-1 V825I allele | -1.431 | 0.641 | -0.073 | -2.231 | 0.026 | |
| ACAT-1 rs1044925 genotype | 1.533 | 0.433 | 0.119 | 3.543 | 0.000 | |
| ACAT-1 rs1044925 allele | 2.336 | 0.535 | 0.146 | 4.363 | 0.000 | |
| LDL-R AvaⅡallele | -1.184 | 0.581 | -0.066 | -2.039 | 0.042 | |
| PCSK9 E670G genotype | -2.923 | 1.469 | -0.064 | -1.989 | 0.047 | |
| PP | ABCA-1 V825I genotype | -1.179 | 0.527 | -0.070 | -2.237 | 0.026 |
| LIPC -250G>A allele | -1.731 | 0.715 | -0.077 | -2.421 | 0.016 | |
| LIPG 584C>T allele | 1.534 | 0.726 | 0.069 | 2.113 | 0.035 | |
| MTHFR 677C>T genotype | 1.226 | 0.545 | 0.075 | 2.249 | 0.025 | |
| PCSK9 E670G genotype | -9.643 | 1.670 | -0.178 | -5.775 | 0.000 |
BP: blood pressure; SBP: systolic blood pressure; DBP: diastolic blood pressure; PP: pulse pressure; ABCA-1: ATP-binding cassette transporter A1; ACAT-1: acyl-CoA:cholesterol acyltransferase-1; LDL-R: low density lipoprotein receptor; LIPC: hepatic lipase gene; LIPG: endothelial lipase gene; MTHFR: methylenetetrahydrofolate reductase; PCSK9: proprotein convertase subtilisin-like kexin type 9; PPARD: peroxisome proliferator-activated receptor delta; SCARB1: Scavenger receptor class B type 1.
Discussion
In the present study, we showed that the genotypic frequencies of ACAT-1 and LIPG, the allelic frequencies of ABCA-1, and the genotypic and allelic frequencies of LDL-R, LIPC, PPARD and SCARB1 were different between nonsmokers and smokers. These results indicate that several lipid-related genes may also involve in the regulation of blood pressure. In the current study, our study population, Bai Ku Yao, is an isolated subgroup of the Yao minority in China. Strict intra-ethnic marriages have been performed in this population from time immemorial. But they can not intermarry within the direct descendant blood kin and the collateral branch blood kin in seven generations [2,3,31,32]. Thus, this population is thought to share the same ethnic ancestry and to possess a homogeneous genetic background. Although genetic variants in some genes have been associated with risk of hypertension in some studies, the results of these association studies conducted with blood pressure traits are inconsistent [8-16].
The association of these SNPs and blood pressure levels between nonsmokers and smokers has not yet been thoroughly studied. In the present study, we showed that the levels of PP in nonsmokers were different among the three genotypes of ABCA-1, and the levels of SBP, DBP and PP in nonsmokers were also different among the three genotypes of LIPC. The levels of SBP in smokers were different among the three genotypes of ABCA-1, ACAT-1, LIPG and PCSK9, the levels of DBP were different among the three genotypes of ACAT-1, LDL-R, LIPC, PCSK9 and PPARD, and the levels of PP were different among the three genotypes of LIPC, LIPG, MTHFR and PCSK9. To the best of our knowledge, the association between ABCA-1 V825I, ACAT-1 rs1044925, LIPC -250G>A, LIPG 584C>T, PCSK9 E670G, and SCARB1 rs5888 SNPs and blood pressure variation has not been previously explored.
The association of LDL-R AvaII, MTHFR C677T and PPARD +294T>C SNPs and blood pressure variation has been evaluated in several previous studies [41-55]. However, the findings are inconsistent. Liu et al. [41] found that there were no differences in the genotypic and allelic frequencies of LDL-R AvaII between normotensives and essential hypertensives in Shanghai. Rodríguez-Esparragón et al. [42] reported that TT genotype of MTHFR C677T was associated with an increased risk of hypertension in males. A previous meta-analysis showed that the MTHFR C677T was consistently associated with severe diastolic hypertension during pregnancy [43]. The MTHFR C667T modulated baseline DBP and DBP responsiveness by short-term treatment of benazepril [44,45]. A significant association between MTHFR C677T and hypertension/hypertension-in-pregnancy in both Caucasian and Asian populations was also observed in a recent meta-analysis [46]. The patients carrying MTHFR 677T allele were at increased risk of hypertension. The frequency of co-occurrence of MTHFR 677CT/1298CC genotypes was significantly higher in the patients compared to controls (P < 0.05) and was associated with increased risk of hypertension [47]. Marinho et al. [48] showed that genotype distribution of MTHFR differed significantly between control and hypertensive patients with a greater prevalence of CT genotypes. The MTHFR 677C allele was significantly more frequent in controls compared with patients with essential hypertension (P < 0.05), and CC genotype was more frequent in controls compared to patients with essential hypertension [49]. However, several studies showed that there were no associations of MTHFR C677T and the prevalence of hypertension or blood pressure levels [50-52]. Gao et al. [53] reported that PPAR-gamma2 SNP is associated with hypertension in the Han population of Inner Mongolia. The frequency of Ala allele was lower in patients with hypertension (1.3%) than in controls (3.6%, P = 0.018). The Pro12Ala polymorphism in PPAR-gamma was associated with blood pressure in subjects with type 2 diabetes. The subjects with Pro/Ala (24%) or Ala/Ala (2%) had lower DBP when adjusted for age and gender compared with Pro/Pro subjects (74%). This association was restricted to men, who also had a borderline significant difference in SBP [54]. However, Yan et al. [55] reported that the frequencies of the PPARD +294T>C genotypes were not different among the groups of metabolic syndrome, essential hypertension, and type 2 diabetes.
The interactions of these SNPs and cigarette smoking on blood pressure levels are not fully known. In the present study, the genotypes of ABCA-1 GG and GA and ACAT-1 AA and AC were shown interactions with cigarette smoking to increase SBP levels, whereas the genotype of PCSK9 AG was shown interactions with cigarette smoking to decrease SBP levels. The genotypes of ACAT-1 AA and AC, LDL-R A-A-, A-A+ and A+A+, MTHFR CC and CT, and PCSK9 AA were shown consistent interactions with cigarette smoking to increase DBP levels. The genotypes of ABCA-1 GA and LIPC GG were shown interactions with cigarette smoking to increase PP levels, whereas the genotypes of PCSK9 AA and AG, and PPARD CC were shown interactions with cigarette smoking to decrease PP levels. These findings suggest that some blood pressure parameters in our study subjects were partly influenced by the interactions of several SNPs and cigarette smoking. Smoking cessation and other healthy lifestyles are necessary for maintaining of normal blood pressure. The effect of different types of cigarettes on blood pressure levels is not well known. In our current study, 95% of the cigarettes smoked by the subjects were natural tobacco leaves, which the toxin content may be high. Tobacco smoke is a complex mixture of over 4,000 chemical constituents. There are many toxins in cigarette smoke such as nicotine, cadmium, carbon monoxide, and reactive oxygen species that might contribute to the cardiovascular toxicity [56]. Thus, the interactions of SNPs and different kinds of cigarettes on blood pressure levels still need to be determined.
Study Limitations
The present study has several potential limitations. First, we did not match smokers and nonsmokers in terms of sex; therefore, the smokers had a much higher proportion of males than did the nonsmokers. Among young and middle-aged adults, population surveys have shown hypertension to be more frequent among males than females, with sex differences of 4% in the United States, 8% in Canada, and 11% in Western Europe [57]. Second, the levels of average age, height, weight, waist circumference, and the percentages of subjects who consumed alcohol were higher in smokers than in nonsmokers. Although sex, age, education level, physical activity, BMI, waist circumference, alcohol consumption, and hyperlipidemia have been adjusted for the statistical analysis, we could not completely eliminate the potential effects of these factors on blood pressure levels among different genotypes in both groups. Third, the diet was not adjusted for the statistical analysis. In the present study, however, the diet in this population is consistent throughout the year and among individuals because of the Bai Ku Yao's reliance on a limited number of locally available food items. Their staple food is corn gruel or corn tortillas. On ordinary days, they are vegetarians [2,3]. Finally, it is clearly established that blood pressure levels are regulated by multiple environmental and genetic factors, and their interactions. Although we have detected the interactions of nine SNPs and cigarette smoking on blood pressure levels in this study, there are still many unmeasured environmental and genetic factors and their interactions. Thus, the interactions of gene-gene, gene-environment, and environment-environment on blood pressure levels remain to be determined.
Conclusion
Several lipid-related gene polymorphisms in nonsmokers and smokers were found to be associated with blood pressure levels in the Guangxi Bai Ku Yao population. The interactions of ABCA-1 AA and AC and ACAT-1 AA and AC genotypes and cigarette smoking to increase SBP, and PCSK9 AG genotype and cigarette smoking to decrease SBP; ACAT-1 AA and AC, LDL-R A-A-, A-A+ and A+A+, MTHFR CC and CT, and PCSK9 AA genotypes and cigarette smoking to increase DBP levels; and ABCA-1 GA and LIPC GG genotypes and cigarette smoking to increase PP, and PCSK9 AA and AG, and PPARD CC genotypes and cigarette smoking to decrease PP levels were also detected. The observed associations and interactions between these SNPs and blood pressure variation in this isolated ethnic subgroup may also be the major characteristics of this condition in the other ethnic groups, especially in the minorities. However, large studies of populations with different ethnic origins are required to confirm these observations.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 30660061).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Hui P, Nakayama T, Morita A. et al. Common single nucleotide polymorphisms in Japanese patients with essential hypertension: aldehyde dehydrogenase 2 gene as a risk factor independent of alcohol consumption. Hypertens Res. 2007;30:585-92
2. Ruixing Y, Weixiong L, Hanjun Y. et al. Diet, lifestyle, and blood pressure of the middle-aged and elderly in the Guangxi Bai Ku Yao and Han populations. Am J Hypertens. 2008;21:382-7
3. Ruixing Y, Shangling P, Shuquan L. et al. Comparison of hypertension and its risk factors between the Guangxi Bai Ku Yao and Han populations. Blood Press. 2008;17:306-16
4. Powell JT, MacSweeney ST, Greenhalgh RM. et al. Interaction between fibrillin genotype and blood pressure and the development of aneurysmal disease. Ann N Y Acad Sci. 1996;800:198-207
5. Schut AF, Sayed-Tabatabaei FA, Witteman JC. et al. Smoking-dependent effects of the angiotensin-converting enzyme gene insertion/deletion polymorphism on blood pressure. J Hypertens. 2004;22:313-9
6. Xu Q, Wang YH, Tong WJ. et al. Interaction and relationship between angiotensin converting enzyme gene and environmental factors predisposing to essential hypertension in Mongolian population of China. Biomed Environ Sci. 2004;17:177-86
7. Gambier N, Marteau JB, Batt AM. et al. Interaction between CYP1A1 T3801C and AHR G1661A polymorphisms according to smoking status on blood pressure in the Stanislas cohort. J Hypertens. 2006;24:2199-205
8. Montasser ME, Shimmin LC, Hanis CL. et al. Gene by smoking interaction in hypertension: identification of a major quantitative trait locus on chromosome 15q for systolic blood pressure in Mexican-Americans. J Hypertens. 2009;27:491-501
9. Mayer O Jr, Filipovský J, Pesta M. et al. The interaction of endothelial nitric oxide synthase polymorphism and current smoking in terms of increased arterial stiffness. Physiol Res. 2010;59:529-36
10. Levy D, Ehret GB, Rice K. et al. Genome-wide association study of blood pressure and hypertension. Nat Genet. 2009;41:677-87
11. Newton-Cheh C, Johnson T, Gateva V. et al. Genome-wide association study identifies eight loci associated with blood pressure. Nat Genet. 2009;41:666-76
12. Sõber S, Org E, Kepp K. et al. Targeting 160 candidate genes for blood pressure regulation with a genome-wide genotyping array. PLoS One. 2009;4:e6034
13. Adeyemo A, Gerry N, Chen G. et al. A genome-wide association study of hypertension and blood pressure in African Americans. PLoS Genet. 2009;5:e1000564
14. Takeuchi F, Isono M, Katsuya T. et al. Blood pressure and hypertension are associated with 7 loci in the Japanese population. Circulation. 2010;121:2302-9
15. Padmanabhan S, Melander O, Johnson T. et al. Genome-wide association study of blood pressure extremes identifies variant near UMOD associated with hypertension. PLoS Genet. 2010;6:e1001177
16. Puppala S, Coletta DK, Schneider J. et al. Genome-wide linkage screen for systolic blood pressure in the Veterans Administration Genetic Epidemiology Study (VAGES) of Mexican-Americans and confirmation of a major susceptibility locus on chromosome 6q14.1. Hum Hered. 2011;71:1-10
17. Fox ER, Young JH, Li Y. et al. Association of genetic variation with systolic and diastolic blood pressure among African Americans: the Candidate Gene Association Resource study. Hum Mol Genet. 2011;20:2273-84
18. International Consortium for Blood Pressure Genome-Wide Association Studies, Ehret GB, Munroe PB. et al. Genetic variants in novel pathways influence blood pressure and cardiovascular disease risk. Nature. 2011;478:103-9
19. Ehret GB, Morrison AC, O'Connor AA. et al. Replication of the Wellcome Trust genome-wide association study of essential hypertension: the Family Blood Pressure Program. Eur J Hum Genet. 2008;16:1507-11
20. Halimi JM, Giraudeau B, Vol S. et al. The risk of hypertension in men: direct and indirect effects of chronic smoking. J Hypertens. 2002;20:187-93
21. Bang LE, Buttenschøn L, Kristensen KS. et al. Do we undertreat hypertensive smokers? A comparison between smoking and non-smoking hypertensives. Blood Press Monit. 2000;5:271-4
22. Niskanen L, Laaksonen DE, Nyyssönen K. et al. Inflammation, abdominal obesity, and smoking as predictors of hypertension. Hypertension. 2004;44:859-65
23. Green MS, Jucha E, Luz Y. Blood pressure in smokers and nonsmokers: epidemiologic findings. Am Heart J. 1986;111:932-40
24. Okubo Y, Suwazono Y, Kobayashi E. et al. An association between smoking habits and blood pressure in normotensive Japanese men: a 5-year follow-up study. Drug Alcohol Depend. 2004;73:167-74
25. Lee DH, Ha MH, Kim JR. et al. Effects of smoking cessation on changes in blood pressure and incidence of hypertension: a 4-year follow-up study. Hypertension. 2001;37:194-98
26. Fogari R, Zoppi A, Lusardi P. et al. Cigarette smoking and blood pressure in a worker population: a cross-sectional study. J Cardiovasc Risk. 1996;3:55-9
27. Primatesta P, Falaschetti E, Gupta S. et al. Association between smoking and blood pressure: evidence from the health survey for England. Hypertension. 2001;37:187-93
28. Halperin RO, Sesso HD, Ma J. et al. Dyslipidemia and the risk of incident hypertension in men. Hypertension. 2006;47:45-50
29. Centers for Disease Control, Prevention (CDC). Cigarette smoking among adults—United States, 1992, and changes in the definition of current cigarette smoking. MMWR Morb Mortal Wkly Rep. 1994;43:342-6
30. Caban-Martinez AJ, Davila EP, Zhao W. et al. Disparities in hypertension control advice according to smoking status. Prev Med. 2010;51:302-6
31. Ruixing Y, Qiming F, Dezhai Y. et al. Comparison of demography, diet, lifestyle, and serum lipid levels between the Guangxi Bai Ku Yao and Han populations. J Lipid Res. 2007;48:2673-81
32. Ruixing Y, Dezhai Y, Shuquan L. et al. Hyperlipidaemia and its risk factors in the Guangxi Bai Ku Yao and Han populations. Public Health Nutr. 2009;12:816-24
33. Cao XL, Yin RX, Wu DF. et al. Genetic variant of V825I in the ATP-binding cassette transporter A1 gene and serum lipid levels in the Guangxi Bai Ku Yao and Han populations. Lipids Health Dis. 2011;10:14
34. Wu DF, Yin RX, Aung LH. et al. Polymorphism of rs1044925 in the acyl-CoA:cholesterol acyltransferase-1 gene and serum lipid levels in the Guangxi Bai Ku Yao and Han populations. Lipids Health Dis. 2010;9:139
35. Long XJ, Yin RX, Li KL. et al. Low density lipoprotein receptor gene Ava II polymorphism and serum lipid levels in the Guangxi Bai Ku Yao and Han populations. Lipids Health Dis. 2011;10:34
36. Meng L, Ruixing Y, Yiyang L. et al. Association of LIPC -250G>A polymorphism and several environmental factors with serum lipid levels in the Guangxi Bai Ku Yao and Han populations. Lipids Health Dis. 2010;9:28
37. Liu WY, Yin RX, Zhang L. et al. Association of the LIPG 584C>T polymorphism and serum lipid levels in the Guangxi Bai Ku Yao and Han populations. Lipids Health Dis. 2010;9:110
38. Zhang L, Yin RX, Liu WY. et al. Association of methylenetetrahydrofolate reductase C677T polymorphism and serum lipid levels in the Guangxi Bai Ku Yao and Han populations. Lipids Health Dis. 2010;9:123
39. Aung LH, Yin RX, Miao L. et al. The proprotein convertase subtilisin/kexin type 9 gene E670G polymorphism and serum lipid levels in the Guangxi Bai Ku Yao and Han populations. Lipids Health Dis. 2011;10:5
40. Miao L, Yin RX, Wu DF. et al. Peroxisome proliferator-activated receptor delta +294T>C polymorphism and serum lipid levels in the Guangxi Bai Ku Yao and Han populations. Lipids Health Dis. 2010;9:145
41. Liu AP, Zhan SY, Li LM. et al. Association between AvaII exon 13 polymorphism at the LDL receptor gene different and serum lipid levels in normotensives and essential hypertensives in Shanghai. Zhonghua Liu Xing Bing Xue Za Zhi. 2003;24:542-6
42. Rodríguez-Esparragón F, Hernández-Perera O, Rodríguez-Pérez JC. et al. The effect of methylenetetrahydrofolate reductase C677T common variant on hypertensive risk is not solely explained by increased plasma homocysteine values. Clin Exp Hypertens. 2003;25:209-20
43. Kosmas IP, Tatsioni A, Ioannidis JP. Association of C677T polymorphism in the methylenetetrahydrofolate reductase gene with hypertension in pregnancy and pre-eclampsia: a meta-analysis. J Hypertens. 2004;22:1655-62
44. Jiang S, Hsu YH, Xu X. et al. The C677T polymorphism of the methylenetetrahydrofolate reductase gene is associated with the level of decrease on diastolic blood pressure in essential hypertension patients treated by angiotensin-converting enzyme inhibitor. Thromb Res. 2004;113:361-9
45. Jiang S, Hsu YH, Niu T. et al. A common haplotype on methylenetetrahydrofolate reductase gene modifies the effect of angiotensin-converting enzyme inhibitor on blood pressure in essential hypertension patients--a family-based association study. Clin Exp Hypertens. 2005;27:509-21
46. Qian X, Lu Z, Tan M. et al. A meta-analysis of association between C677T polymorphism in the methylenetetrahydrofolate reductase gene and hypertension. Eur J Hum Genet. 2007;15:1239-45
47. Markan S, Sachdeva M, Sehrawat BS. et al. MTHFR 677 CT/MTHFR 1298 CC genotypes are associated with increased risk of hypertension in Indians. Mol Cell Biochem. 2007;302:125-31
48. Marinho C, Alho I, Arduíno D. et al. GST M1/T1 and MTHFR polymorphisms as risk factors for hypertension. Biochem Biophys Res Commun. 2007;353:344-50
49. Ilhan N, Kucuksu M, Kaman D. et al. The 677 C/T MTHFR polymorphism is associated with essential hypertension, coronary artery disease, and higher homocysteine levels. Arch Med Res. 2008;39:125-30
50. Ravera M, Viazzi F, Berruti V. et al. 5,10-Methylenetetrahydrofolate reductase polymorphism and early organ damage in primary hypertension. Am J Hypertens. 2001;14:371-6
51. Lwin H, Yokoyama T, Yoshiike N. et al. Polymorphism of methylenetetrahydrofolate reductase gene (C677T MTHFR) is not a confounding factor of the relationship between serum uric acid level and the prevalence of hypertension in Japanese men. Circ J. 2006;70:83-7
52. Stiefel P, Miranda ML, Bellido LM. et al. Genotype of the CYBA promoter -930A/G, polymorphism C677T of the MTHFR and APOE genotype in patients with hypertensive disorders of pregnancy: an observational study. Med Clin (Barc). 2009;133:657-61
53. Gao L, Wang L, Yun H. et al. Association of the PPARgamma2 gene Pro12Ala variant with primary hypertension and metabolic lipid disorders in Han Chinese of Inner Mongolia. Genet Mol Res. 2010;9:1312-20
54. Ostgren CJ, Lindblad U, Melander O. et al. Peroxisome proliferator-activated receptor-gammaPro12Ala polymorphism and the association with blood pressure in type 2 diabetes: skaraborg hypertension and diabetes project. J Hypertens. 2003;21:1657-62
55. Yan ZC, Shen CY, Zhong J. et al. PPARdelta + 294T/C gene polymorphism related to plasma lipid, obesity and left ventricular hypertrophy in subjects with metabolic syndrome. Zhonghua Xin Xue Guan Bing Za Zhi. 2005;33:529-33
56. Ruixing Y, Qi B, Tangwei L. et al. Effects of Nicotine on angiogenesis and restenosis in a rabbit model. Cardiology. 2007;107:122-31
57. Wolf-Maier K, Cooper RS, Banegas JR. et al. Hypertension prevalence and blood pressure levels in 6 European countries, Canada, and the United States. JAMA. 2003;289:2363-9
AUTHOR BIOGRAPHY
Dr Rui-Xing Yin chief physician of Institute of Cardiovascular Diseases, Guangxi Medical University; professor of internal medicine, Guangxi Medical University; and associate Editor-in-Chief of the Journal of Guangxi Medical University, Nanning, People's Republic of China. He finished his medical courses at Guangxi Medical University (1978-1983), and then trained for a physician at the Affiliated Hospital of Nanning District Health School. He trained in Cardiology and completed his MSc. at Guangxi Medical University again (1987-1990). He then worked as a visiting and vice chief physician in the Department of Cardiology, Institute of Cardiovascular Diseases, Guangxi Medical University (1990-1996). He obtained his M.D. at Guangdong Provincial Cardiovascular Institute (1996-1999). Since then he has worked at the Institute of Cardiovascular Diseases, Guangxi Medical University. He was a visiting scholar of Nutrition and Genomics Laboratory, Jean Mayer USDA HNRCA (2007-2008). Dr Yin was also a representative, the Tenth People's Congress of Guangxi (2003-2007). He attended the 4th HUGO Pacific Meeting & 5th Asia- Pacific Conference on Human Genetics in Pattaya, Thailand (2002), the 4th European Congress of Biogerontology in Newcastle upon Tyne, England (2004), and the 7th SCICC (2005). He visited Hue Medical College (2003), University of London (2004), and University of Newcastle Upon Tyne (2004). He was the Membership of the Society for Experimental Biology and Medicine (SEBM), Chinese Atherosclerosis Society (CAS), Society of Internal Medicine, Chinese Medical Association (CMA), and Committeeman of Cardiovascular Disease Committee of Chinese Association of Rehabilitation Medicine (CARM). He was the editorial board of Open Heart Failure Journal, Journal of Medical Genetics and Genomics, and Journal of Clinical Medicine and Research. Four research programs were supported by the Chinese National Natural Science Foundation (No: 81160111, No: 30960130, No: 30660061, and No: 30360038). He has published more than 290 articles in Chinese or in English. Fifty-six articles were collected in the Web of Science.
![]() Corresponding author: Rui-Xing Yin, MD, Department of Cardiology, Institute of Cardiovascular Diseases, the First Affiliated Hospital, Guangxi Medical University, 22 Shuangyong Road, Nanning 530021, Guangxi, People's Republic of China; Tel: +86-771-5326125; Fax: +86-771-5353342; Email: yinruixingcom.cn.
Corresponding author: Rui-Xing Yin, MD, Department of Cardiology, Institute of Cardiovascular Diseases, the First Affiliated Hospital, Guangxi Medical University, 22 Shuangyong Road, Nanning 530021, Guangxi, People's Republic of China; Tel: +86-771-5326125; Fax: +86-771-5353342; Email: yinruixingcom.cn.
Received 2012-3-26
Accepted 2012-5-8
Published 2012-5-10
