ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2015; 11(9):1088-1099. doi:10.7150/ijbs.10603 This issue Cite
Research Paper
The Small Molecule R-(-)-β-O-Methylsynephrine Binds to Nucleoporin 153 kDa and Inhibits Angiogenesis
1. Department of Biotechnology, Translational Research Center for Protein Function Control, College of Life Science & Biotechnology, Yonsei University, Seoul 120-749, Republic of Korea
2. Eskitis Institute, Griffith University, Brisbane QLD 4111, Australia
3. Faculty of Health Sciences, University of Macau, Av. Universidade, Taipa, Macau SAR, China
4. Department of Ophthalmology, Seoul National University College of Medicine, Seoul 110-799, Republic of Korea
5. Department of Microbiology and Immunology, Seoul National University College of Medicine, Seoul 110-799, Republic of Korea
6. Department of Internal Medicine, Yonsei University College of Medicine, Seoul 120-752, Republic of Korea
Abstract
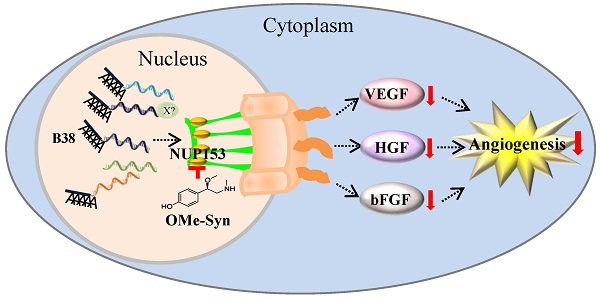
R-(-)-β-O-methylsynephrine (OMe-Syn) is a naturally occurring small molecule that was identified in a previous screen as an inhibitor of angiogenesis. In this study, we conducted two animal model experiments to investigate the in vivo antiangiogenic activity of OMe-Syn. OMe-Syn significantly inhibited angiogenesis in a transgenic zebrafish model as well as in a mouse retinopathy model. To elucidate the underlying mechanisms responsible for the antiangiogenic activity of OMe-Syn, we used phage display cloning to isolate potential OMe-Syn binding proteins from human cDNA libraries and identified nucleoporin 153 kDa (NUP153) as a primary binding partner of OMe-Syn. OMe-Syn competitively inhibited mRNA binding to the RNA-binding domain of NUP153. Furthermore, depletion of NUP153 in human cells or zebrafish embryos led to an inhibition of angiogenesis, in a manner similar to that seen in response to OMe-Syn treatment. These data suggest that OMe-Syn is a promising candidate for the development of a novel antiangiogenic agent and that inhibition of NUP153 is possibly responsible for the antiangiogenic activity of OMe-Syn.
Keywords: Angiogenesis, Natural product, Small molecule, NUP153, Target identification
