ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2007; 3(5):292-302. doi:10.7150/ijbs.3.292 This issue Cite
Research Paper
Human Maf1 negatively regulates RNA Polymerase III transcription via the TFIIB family members Brf1 and Brf2
1. Department of Biological Sciences, St. John's University, Queens NY, USA.
2. Department of Biochemistry, Albert Einstein College of Medicine, Bronx NY, USA.
* These authors contributed equally to this work.
Abstract
RNA polymerase III (RNA pol III) transcribes many of the small structural RNA molecules involved in processing and translation, thereby regulating the growth rate of a cell. Initiation of pol III transcription requires the evolutionarily conserved pol III initiation factor TFIIIB. TFIIIB is the molecular target of regulation by tumor suppressors, including p53, RB and the RB-related pocket proteins. However, our understanding of negative regulation of human TFIIIB-mediated transcription by other proteins is limited. In this study we characterize a RNA pol III luciferase assay and further demonstrate in vivo that a human homolog of yeast Maf1 represses RNA pol III transcription. Additionally, we show that Maf1 repression of RNA pol III transcription occurs via TFIIIB, specifically through the TFIIB family members Brf1 and Brf2.
Keywords: RNA polymerase III, TFIIIB, Maf1, Brf2
1. Introduction
Eukaryotic cells divide the responsibility of transcribing cellular genes among three DNA dependent RNA polymerases [1]. RNA pol I transcribes rRNA, RNA pol II transcribes mRNA, as well as a few of the small nuclear RNAs (snRNAs) involved in splicing, and RNA pol III transcribes the small structural RNAs involved in processing and translation [2]. RNA pol III is the largest of the eukaryotic polymerases and has the greatest number of subunits, yet its transcriptional products are untranslated RNAs that are usually smaller than 300 nucleotides in length [2, 3]. The genes transcribed by human RNA pol III may be further subdivided according to promoter structure, which may be entirely gene internal (type 1 and 2) or external (type 3), and basal transcription factor requirements [4]. Recruitment of RNA pol III is a consequence of specific transcription factor complex assembly on the different classes of promoters [2]. Ultimately, proper initiation by RNA pol III requires TFIIIB [2, 5-8].
S. cerevisiae TFIIIB is well defined, comprised of three polypeptides, the TATA box binding protein TBP, the TFIIB related factor Brf1, and a third polypeptide Bdp1[7-9]. All three polypeptides are necessary and sufficient to reconstitute TFIIIB activity in yeast [6, 10, 11]. In humans, TFIIIB activity is more complex, as at least two forms of TFIIIB activity have been identified [12-15]. Transcription from gene internal promoters, such as tRNA genes, requires a TFIIIB complex comprised of TBP, Brf1 and Bdp1, whereas expression from gene external promoters, such as the U6 snRNA gene, require a TFIIIB complex containing TBP, Brf2 and Bdp1 [2, 13, 14].
RNA pol III activity is intimately linked to growth conditions and is tightly regulated throughout the cell cycle; RNA pol III activity is low during mitosis, increases slowly through G1 and reaches its maximal activity during the S and G2 phases [16, 17]. As such, RNA pol III transcription is a target of regulation by tumor suppressors including p53, RB, and the RB-related pocket proteins [18-20]. Specifically, the tumor suppressors p53, RB, p130, and p107 have been demonstrated to inactivate RNA pol III transcription through physical associations with TFIIIB subunits at different stages of the cell cycle [19, 21, 22].
Maf1 has been identified as a global repressor of pol III transcription in yeast [5, 23-25], see [26, 27] for a review. The repressing activity of Maf1 was originally proposed based, in part, on the observation that maf1 mutant yeast cells have higher levels of mature tRNAs, as compared to wild-type [28]. Subsequently, yeast Maf1 was shown to be required for down-regulation of RNA pol III transcription during the growth cycle and in response to nutrient limitation, DNA damage, oxidative stress and a variety of drug treatments [5, 24]. Hence, conditions that cause inhibition of cell proliferation, at least in yeast, appear to activate different signaling pathways that converge on Maf1 and regulate its repressing activity [24, 25]. It has been determined that two important targets of yeast Maf1 repression are the TFIIIB subunit Brf1 and RNA pol III [5, 24]. Yeast Maf1 co-precipitates with Pol III and Brf1 [5, 24] and recombinant Maf1 has been shown to inhibit tRNA transcription in vitro [5]. Recently, it has been demonstrated that phosphorylation and dephosphorylation of Maf1 by protein kinase A and protein phosphatase 2A respectively, regulate the cellular localization of Maf1 and hence its ability to repress transcription [24, 29-31]. Putative Maf1 homologs have been identified in a variety of organisms, including humans, and it has been speculated that the function of Maf1 will be conserved from yeast to man [28]. The Maf1 orthologs are highly conserved; containing three regions (termed A, B, C) with a high degree of sequence similarity, but with unknown function [28]. Only recently has a role for human Maf1 in RNA pol III transcription been described, whereby Reina et al. [32] demonstrate that human Maf1 represses RNA pol III transcription from type 1, 2, and 3 promoters in vitro and that endogenous Maf1 associates with Brf1 and the largest subunit of RNA pol III via co-immunoprecipitation and pull-down assays.
In contrast to yeast, there are at least two forms of human TFIIIB, each requiring different TFIIB family members for transcription from the different classes of RNA pol III promoters [2, 13, 14]. Thus, we sought to determine if a putative human homolog of yeast Maf1 functions as an in vivo repressor of gene internal RNA pol III transcription through the TFIIB family member Brf1, and whether human Maf1 would be able to also repress gene external RNA pol III transcription through the TFIIB family member Brf2, which does not have a functional homolog in yeast.
Here we report that a human homolog of yeast Maf1 represses RNA pol III transcription from both gene -internal and -external promoters through TFIIIB using an in vivo RNA pol III luciferase assay. We further demonstrate that human Maf1 is capable of repressing two forms of human TFIIIB activity via the TFIIB family members Brf1 and Brf2. Specifically, we have identified a mechanism by which Maf1 inhibits U6 transcription through direct interactions with Brf2.
2. Materials and Methods
Luciferase reporter constructs
The human U6 promoter was cloned by PCR from human genomic DNA (Clontech) using primers previously described [33] with the restriction sites for Kpn I and Bgl II added to the forward and reverse primers, respectively, allowing directional cloning into the pGL3 basic luciferase vector, which lacks eukaryotic promoter and enhancer sequences (Promega). The VAI promoter region was amplified by PCR from the pADVantage vector (nucleotides 780-939) (Promega) and subsequently directionally cloned into pGL3 using the Kpn I and Bgl II restriction sites. A mutant U6 construct, (pGL3-mtU6) containing mutations in the proximal sequence element (PSE) and the TATA box was generated by PCR, using a plasmid with a mutant human PSE and TATA box as a template, which has been described [34-36]. A deletion pGL3-VAI construct for the VAI promoter was generated as previously described [37]. pGL3mtTATAU6 was generated using previously characterized mutations in the TATA box of pGL3-U6 [38]. A mutant pGL3-U6TTT was generated by PCR to add a previously characterized tandem run of 6T residues (5'-AGCTTTTTTGGAATTTTTTGGAAAGCCAC-3') [39] downstream of the U6 promoter, but upstream of the Kozak consensus translation initiation site of the luciferase coding sequence of pGL3.
Mammalian expression constructs
3XFlagMaf1 was generated by PCR amplification of the coding sequence of Maf1 (Genbank accession number NM032272) from HeLa cDNA and ligated into the Xba I and BamH I sites of p3XFlag-CMV7.1 (Sigma). Mammalian expression vectors for Brf1 and Brf2 were generated by PCR amplification of coding sequences of pSBETFlag/HTBrf1[13]and pSBETFlag/HT-Brf2 and ligated into the Xba I and BamH I sites of p3XFlag-CMV-7.1 (Sigma). GFP-Brf2 was generated by PCR amplification of Brf2 coding sequence and subsequent ligation into the Kpn I and BamH I sites of pEGFP-N3 (BD Biosciences Clontech). For GSTMaf1 used in vitro GST pull-down assay, we PCR amplified Maf1 sequence from HeLa cDNA and ligated the coding sequence in frame of GST tag using the BamH I and EcoR I sites of pGEX-5X-1(Pharmacia Biotech) and expressed and purified as previously described [13].
Cell culture and luciferase assays
HeLa cells (ATCC) were maintained in 10% FBS DMEM (Biowhittaker). Transient transfections were accomplished using TransIT-LT1 (Mirus), as per the manufacturer's protocol. Dual-luciferase reporter assay system (Promega) was used to monitor luciferase activity in HeLa cells as per the manufacturer's recommendations, using a Sirius single tube luminometer (Berthold). A Renilla luciferase vector (Promega) was co-transfected in all transfections described to monitor transfection efficiency. Luciferase experiments were performed in triplicate or quadruplicate, repeated three independent times, and the data presented are representative experiments. All luciferase results are reported as relative light units (RLU): the average of the Photinus pyralis firefly activity observed divided by the average of the activity recorded from Renilla luciferase vector. Results were analyzed using Graphpad PRISM 3.03.
Immunofluorescence
Transiently transfected HeLa cells were fixed in either 4% paraformaldehyde for 20 minutes or 0.5% paraformaldehyde overnight. Cells were permeabilized with 0.2%Triton-X for 20 minutes, blocked in 10% goat serum for 45 minutes at room temperature or overnight at 4oC. All 1o antibodies, rabbit and mouse anti-Flag (Sigma) and rabbit anti-GFP (Molecular Probes), were diluted 1:100 in 2% goat serum and cells were incubated for 1 hour. FITC-conjugated goat anti rabbit IgG (Amersham Biosciences) and Alexa-546 conjugated goat anti mouse IgG (Molecular Probes) secondary antibodies were diluted 1:200 with 2% goat serum. Cells that were stained with propidium iodide (5ug/ml) were incubated for 5 min. after pretreatment with RNase (Roche) or stained with DAPI (Sigma) to determine nuclear localization. All images were captured using a laser scanning confocal microscope (Leica TCS SL)
Co-immunoprecipitation and GST pull-down assays
Nuclear extract was prepared from HeLa cells [13] transfected with FlagMaf1 or empty Flag vectors and incubated with NET-2 (150 mM NaCl/40 mM Tris•HCl, pH 7.5/0.05% TritonX-100) equilibrated Flag M2 agarose (Sigma) overnight at 4 degrees, then extensively washed with NET-2 and eluted with Flag peptide (Sigma). Eluants were resolved by SDS-PAGE, wet transferred to nitrocellulose and subsequently immuno-blotted with anti-Brf2 antibody (CSH 1228), a generous gift from N. Hernandez, Cold Spring Harbor Laboratory. Flag/HTBrf2, GSTMaf1 and the empty GST vector were expressed and purified as previously described [13, 40] and mixed with TBS (100 mM Tris-HCl, 0.9% NaCl, pH 7.4) equilibrated GST agarose (Sigma) overnight at 4 degrees, then extensively washed with TBS, containing 300mM NaCl, and eluted in 50mM Tris containing 10mM reduced glutathione. Eluants were resolved by SDS-PAGE, wet transferred to nitrocellulose and subsequently immunoblotted with anti-Flag antibody (Sigma).
Western blot analysis
Transiently transfected cells were harvested and nuclear extract was prepared as previously described [13]. Nuclear extract was separated on 10% SDS-PAGE gels, wet transferred to nitrocellulose, blocked 1 hour room temperature using nonfat milk in Tris Buffered Saline (TBS), pH 7.5. The blot(s) were incubated in primary antibodies: anti-actin (Santa Cruz), anti-Flag polyclonal (Sigma), overnight at 4 degrees. The blot(s) were washed in 1X TBS and then incubated in secondary antibody at room temperature, then developed using ECF (for anti-rabbit-AP or anti-mouse-AP antibodies, Amersham) or TMB substrate (Promega) (for anti-goat-HRP, Amersham) and photodocumented using a BioDocit system (UVP).
3. Results
Transcription from gene internal (VAI) and gene external (U6) promoters can be monitored in HeLa cells using a dual luciferase assay
RNA pol III transcribes both gene- internal and external promoters as exemplified by the VAI (a prototypical tRNA-like promoter) and human U6 promoters, respectively. Previously, most of our understanding of the mechanisms of RNA pol III transcription has come from in vitro transcription assays utilizing nuclear or whole cell extracts prepared from mammalian cells grown in culture. Although this methodology has been invaluable for understanding RNA polymerase III transcription and in the subsequent identification of transcription factors, the major limitation is our inability to investigate the regulation of RNA pol III transcription in intact mammalian cells. Therefore, we sought to develop and characterize a reporter assay that could be used to study RNA pol III transcription from gene –internal and –external promoters in intact mammalian cells.
To determine if we could monitor gene external RNA pol III promoter activity in vivo we PCR amplified the human U6 promoter from human genomic DNA as previously described [33]. This human U6 promoter contains both a proximal sequence element (PSE) and TATA box located upstream of the transcription start site and was ligated into pGL3-basic, lacking both promoter and enhancer sequences, resulting in pGL3-U6. pGL3-U6 was transiently transfected into HeLa cells yielding higher luciferase activity, as compared to the empty pGL3 vector (Figure 1A). Addition of increasing amounts of pGL3-U6 resulted in a dose-dependent increase in luciferase activity. These results are in agreement with a recent report demonstrating that both the human and mouse U6 promoters can drive luciferase expression in a promoterless pGL4.10 plasmid [39]. To examine whether expression of the luciferase reporter in HeLa cells could be driven by a gene internal RNA polymerase III promoter in vivo, we utilized the well studied VAI gene, a prototypical tRNA-like promoter. The VAI gene has canonical A and B box promoter elements located downstream of the transcription start site. The VAI gene was PCR amplified from pADVantage and ligated into pGL3-Basic, yielding pGL3-VAI which was transiently transfected into HeLa cells and demonstrated a dose response increase in luciferase activity, as compared to the empty pGL3 vector (Figure 1A). Taken together these data suggest that gene-internal and -external RNA pol III promoter activity may be monitored in HeLa cells using this luciferase assay.
Previously, the issue has been raised as to whether RNA pol II can transcribe RNA pol III promoters in short hairpin RNA (shRNA) expression vectors [39]. Hence, we sought to determine if the observed luciferase activities from luciferase vectors driven by RNA pol III promoters (Figures 1A) were specific to RNA pol III transcription. We generated a mutant pGL3-U6 expression construct (pGL3-mtU6) with previously characterized mutations in both the PSE and TATA box [35, 36] and tested its ability to express luciferase in transiently transfected HeLa cells. As depicted in Figure 1B, mutations in the PSE and TATA box of the human U6 promoter abolishes luciferase activity in HeLa cells, as compared to luciferase activity driven by the wild type human U6 promoter. We also determined the specificity of pol III transcription from the human pGL3-VAI construct by generating a previously characterized deletion mutant [37] lacking both the canonical A and B box promoter elements of VAI. This construct was subsequently tested in our luciferase assay, and as expected, deletion of the A and B boxes severely inhibited VAI transcription (Figure 1C).
To ensure that our RNA pol III luciferase assay was indeed measuring RNA pol III and not cryptic RNA pol II activity, we tested whether this RNA pol III luciferase assay followed the classical alpha-amanitin sensitivity profile established for eukaryotic DNA dependent RNA polymerases [41]. We observed that pGL3-U6 luciferase activity was not affected at low doses of alpha amanitin (3ug/ml), but transcription from a RNA pol II-like promoter [38](pGL3-mtTATAU6) was decreased at 3 ug/ml (Figure 1D). Also, transcription from pGL3-VAI was not inhibited at 2 ug/ml alpha amanitin, whereas transcription from pGL3-mtTATAU6 was severely inhibited 2 ug/ml alpha amanitin (Figure 1E).
To determine if we could artificially terminate RNA pol III transcription in our system, we inserted a prototypical RNA pol III termination sequence, a tandem oligo containing 6T residues downstream of the human U6 promoter, but upstream of the Kozak consensus translation initiation site of the luciferase coding sequence of pGL3 [39, 42]. The insertion of a run of six T residues severely inhibited luciferase activity from the pGL3-U6 promoter (Figure 1F), as compared to wild-type pGL3-U6. These results differ from Rumi et al [39] who were unable to demonstrate that the insertion of a RNA pol III termination signal downstream of the human U6 promoter and upstream of the luciferase start ATG significantly inhibited RNA pol III transcription. A potentially important difference is vector construction. Rumi et al inserted the EGFP shRNA sequence between the human H1 promoter and the RNA pol III terminator sequence [39]. Using TESS: Transcription Element Search Software [43], we determined that the EGFP shRNA sequence contains putative RNA pol II transcription factor binding sites which may serve to enhance cryptic RNA pol II transcription from the human H1 promoter in the pGL4.10 utilized by Rumi et al [39].
RNA polymerase III transcription can be measured in vivo using a RNA pol III dual luciferase assay. (A) Transient transfection of HeLa cells with increasing concentrations of pGL3-U6 (100ng, 200ng, 300ng), pGL3-VAI (100ng, 200ng, 300ng) or empty pGL3 vector (300ng). (B) Luciferase activity comparison of the wild-type human U6 promoter (100ng) with a mutant human U6 promoter, lacking a functional TATA box and PSE (100ng). Cells were also transfected with empty pGL3 (100ng) as negative control. (C) Comparison of wild-type VAI (100ng) promoter luciferase activity with a mutant VAI construct lacking both the A and B box promoter elements of the VAI promoter (100ng). Cells were also transfected with empty pGL3 (100ng) as negative control. (D) HeLa cells transiently transfected with either empty pGL3 (50ng), wild-type pGL3-U6 (50ng), or a previously characterized mutant U6 lacking a functional TATA box and transcribed by RNA pol II, pGL3-mtTATAU6 (25ng) and treated with 0 μg/ml or 3 μg/ml α-amanitin. (E) HeLa cells transiently transfected with either empty pGL3 (100ng), wild-type pGL3-VAI (100ng), or a previously characterized mutant U6 lacking a functional TATA box and transcribed by RNA pol II, pGL3-mtTATAU6 (50ng) and treated with 0 μg/ml, 1 μg/ml or 2 μg/ml α-amanitin. (F) Transient transfection of pGL3 (100ng), pGL3-U6 (100ng), or pGL3-U6TTT (100ng) with a run of T residues inserted downstream of the human U6 promoter, but upstream of the the Kozak consensus translation initiation site of the luciferase gene in pGL3. All luciferase assay results expressed as relative light units (RLU): the average of the Photinus pyralis firefly activity observed divided by the average of the activity recorded from Renilla luciferase vector. Experiments were done in triplicate, repeated three times, representative experiments are depicted.
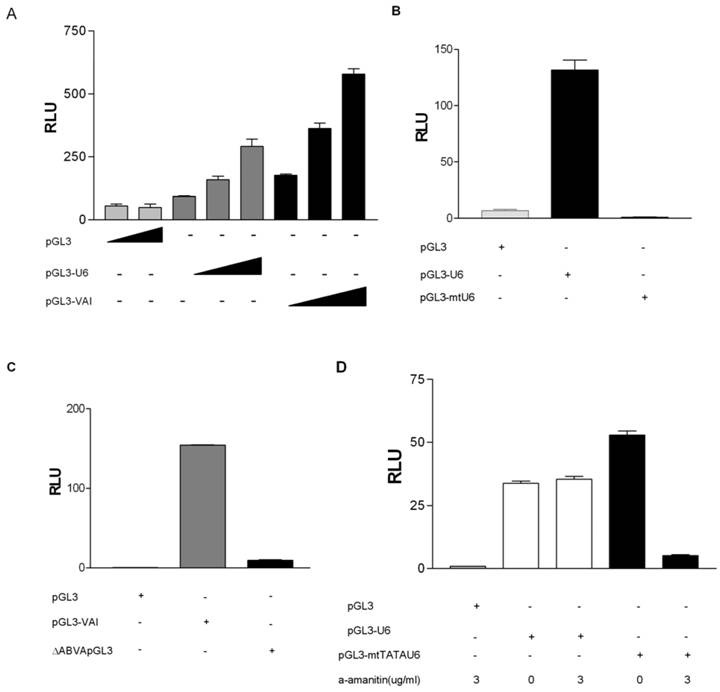
Taken together (Figures 1A-1F) these data suggest that our RNA pol III luciferase assay can specifically and effectively monitor transcription from prototypical gene- internal and -external RNA pol III promoters.
Human Maf1 represses RNA pol III transcription in HeLa cells
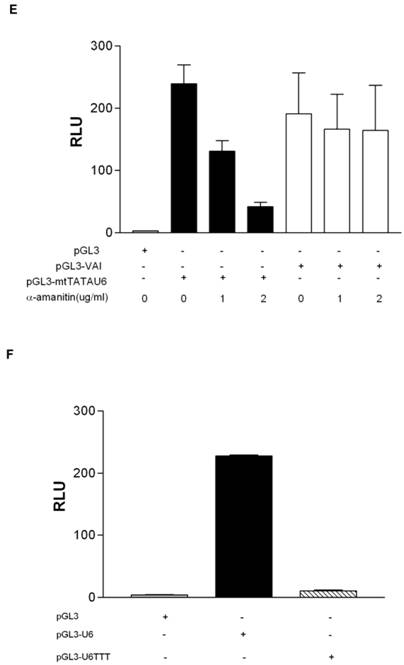
To determine if the putative human homolog of yeast Maf1 is indeed a repressor of human RNA pol III transcription, we sub-cloned human Maf1 (Genbank accession number NM032272) from HeLa cDNA into p3XFlag-CMV-7.1 and transiently co-transfected the construct with pGL3-U6 into HeLa cells. By western blot (Figure 2A), the 3XFlag-tagged Maf1 construct expresses at detectable levels in HeLa cells, as compared to HeLa cells transfected with the empty Flag vector. Interestingly, we routinely observed a doublet, indicated by asterisks, when expressing Flag-Maf1 in HeLa cells (lane 2 Figure 2A). We speculate that the observed upper band is a phosphorylated form of Maf1, consistent with the properties of Maf1 in yeast [29, 30] and a recent report regarding mammalian Maf1[32]. To determine if Maf1 is indeed phosphorylated, we treated the FlagMaf1 expressing HeLa nuclear extract with calf intestine phosphatase for various times and analyzed the samples by anti-Flag western blot. We demonstrated a loss in the upper band and a proportional increase in the lower faster migrating band (Figure 2C). As of yet, it is unclear if this observed dephosphorylation of human Maf1 plays the same important role in the regulation of RNA pol III transcription recently observed in yeast [30, 31].
To determine if human Maf1 is a repressor of RNA pol III transcription, like its yeast counterpart, we performed transient transfections and demonstrated that increasing concentrations of Maf1 inhibited both U6- and VAI-driven luciferase activity in a dose- dependent manner (Figure 2D). Taken together these luciferase data suggest that human Maf1 is an inhibitor of both gene internal and gene external RNA pol III transcription in vivo and these data are in agreement with a recent report demonstrating Maf1 regulation of human RNA pol III transcription using nuclear run on (VAI) and RNA protection (U6) assays in vitro [32].
Human Maf1 inhibits RNA polymerase III transcription from both gene internal and gene external promoters. (A) Anti-flag immunoblot of nuclear extract prepared from HeLa cells transiently transfected with: empty flag vector (lane 1), FlagMaf1 (lane 2), FlagBrf1 (lane 3), FlagBrf2 (lane 4). Arrows depict location of each protein as determined by anti-Flag antibody. Asterisks denote polypeptide doublet routinely observed in HeLa cells over-expressing FlagMaf1. (B) Same immunoblot from 2A reprobed using anti-actin antibody as loading control for transfections with FlagBrf1, FlagBrf2 and empty Flag vector. (C) Nuclear extract prepared from HeLa cells over-expressing FlagMaf1 treated with calf intestine phosphatase (CIP) for increasing amounts of time and immunoblotted with an anti-Flag antibody. (D) Transient transfection of HeLa cells with pGL3-VAI (100ng) (left panel) or pGL3-U6 (100ng) (right panel) with increasing amounts of FlagMaf1(50ng, 100ng, 200ng) and empty vectors Flag (200ng) and pGL3(100ng) as indicated by + sign.
Repression of RNA Pol III transcription by Maf1 occurs through the TFIIB family members Brf1 and Brf2
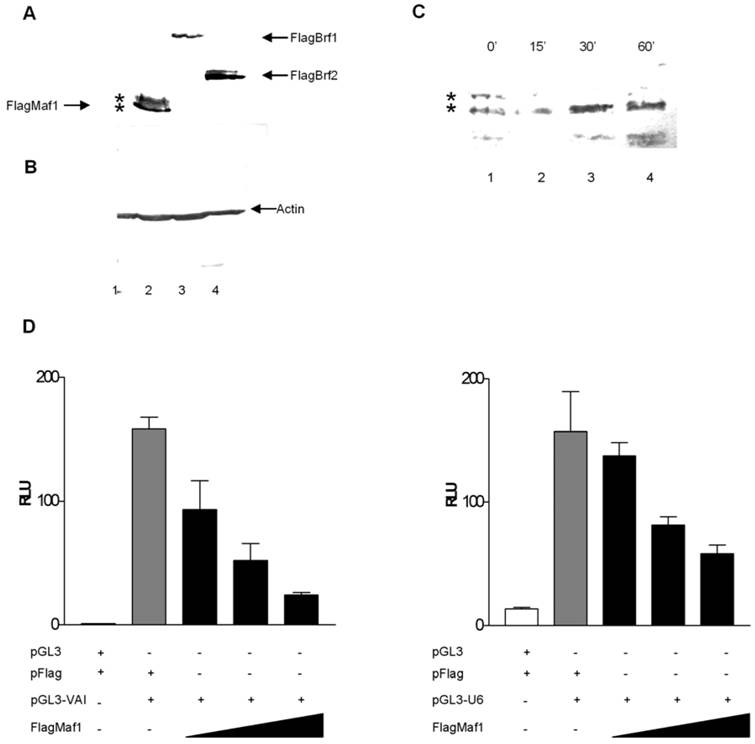
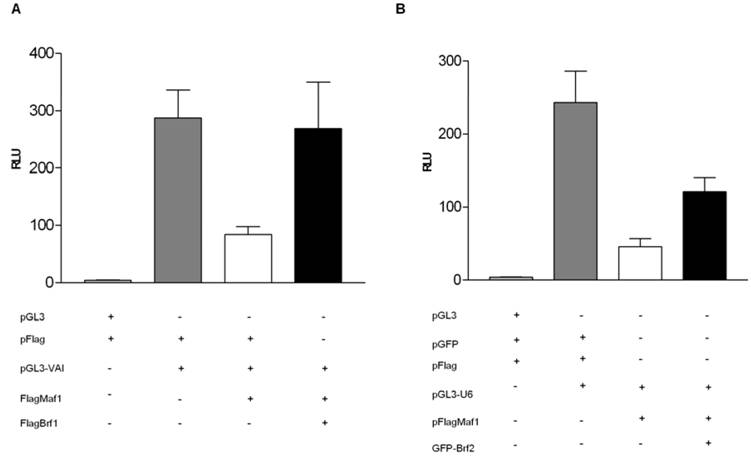
Desai et al [5] identified a two step mechanism by which yeast Maf1 represses RNA pol III transcription: (1) inhibition of TFIIIB assembly at the promoter and (2) inhibition of RNA pol III recruitment to TFIIIB already assembled on promoters under conditions which favor cellular growth repression. To elucidate whether human Maf1 represses RNA pol III transcription by a similar mechanism, we attempted to overexpress the TFIIIB subunits Brf1 and Brf2 to determine if this would alleviate repression of RNA pol III transcription by human Maf1. Overexpression of human Brf1 in HeLa cells co-expressing human Maf1 alleviates the observed repression of RNA pol III transcription from the VAI promoter (Figure 3A). We are able to restore RNA pol III activity in the Maf1 repressed HeLa cells to activity levels similar to that observed in the control HeLa cells by the overexpression of Brf1 (Figure 3A). These results suggest that inhibition of VAI transcription by human Maf1 occurs via the TFIIIB subunit Brf1, suggesting a similar mechanism to that observed in yeast [5, 24]. Furthermore, Reina et al [32] demonstrated Maf1 physically associates with Brf1 and the RNA pol III subunit RPC1 by co-immunoprecipitation assays.
Yeast cells do not utilize gene-external RNA pol III promoters of the type that require Brf2 for transcription in human cells [2, 13]. Thus, it is not known whether Brf2, like Brf1, is a molecular target of Maf1 repression. To address this question, we tested whether transient overexpression of Brf2 can rescue U6 promoter-driven luciferase activity in HeLa cells co-transfected with Maf1. Co-transfection of human Brf2 with Maf1 could only restore activity to approximately 50% of the control luciferase activity observed in the control HeLa cells not transfected with the Maf1 construct (Figure 3B). Although we were able to alleviate Maf1 repression of U6 transcription nearly 50%, by the co-expression of GFP-Brf2, we were never able to restore U6 transcription levels to those seen in the control HeLa cells transfected with the U6 promoter and the vector expressing only the Flag epitope. Currently, we cannot rule out the possibility that overexpression of a GFP-Brf2 fusion is not as efficient at rescuing Maf1 repression. Alternatively or in addition, the U6 promoter may be more sensitive than the VA1 promoter to other effects of Maf1-dependent repression. To further investigate the mechanism by which Maf1 represses U6 transcription we sought to determine if Brf2 and Maf1 could co-localize in HeLa cells.
Overexpression of the TFIIIB subunits Brf1 or Brf2 alleviates Maf1 induced repression of transcription from both gene internal and external promoters RNA polymerase III. (A) HeLa cells were transiently transfected with: pGL3 and Flag vector, or pGL3-VAI and Flag vector, pGL3-VAI and FlagMaf1, pGL3-VAI, FlagMaf1 and Brf1. (B) HeLa cells were transiently transfected with: pGL3 and Flag vector, or pGL3-U6 and Flag vector, pGL3-U6 and FlagMaf1, pGL3-U6, FlagMaf1 and Brf2.
Human Maf1 interacts with Brf2 in HeLa cells
To date there have been few reports depicting the dynamics of TFIIIB by immunofluorescence in living mammalian cells [44, 45]. Recently, Chen et al investigated the physical association of the TFIIIB subunit TBP with chromosomes during the cell cycle, and determined that TBP stably associates with the condensed mitotic chromosomes, despite the known inactivation of TFIIIB and the inhibition of RNA pol III transcription that occurs during mitosis [44].
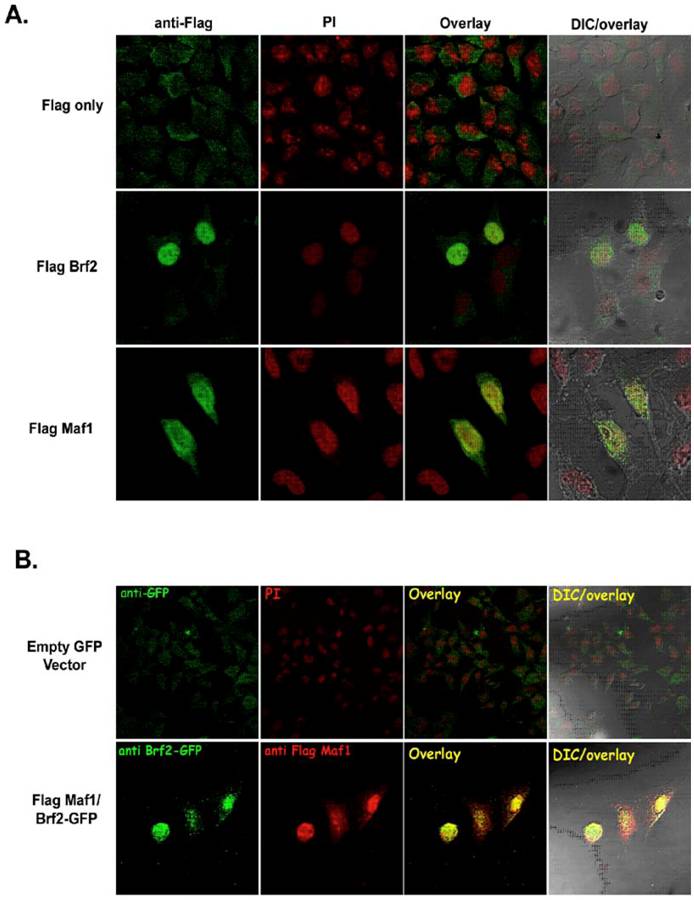
There is little experimental evidence depicting the physical localization of human TFIIIB and known repressors of RNA pol III transcription. Hence, we sought to determine if our epitope tagged TFIIIB subunit Brf2 and the RNA pol III repressor Maf1 constructs were able to localize to the nucleus. FlagBrf2 was transiently transfected into HeLa cells and localized to the nucleus, as judged by propidium iodide staining (Figure 4A). In contrast to the localization patterns of FlagMaf1 and FlagBrf2, the vector expressing only the Flag epitope showed primarily cytoplasmic staining with little or no signal in the nucleus (Figure 4A). In Figure 4B, FlagMaf1 and GFP-Brf2 were co-transfected into HeLa cells where they co-localized. Both the Brf2 and Maf1 constructs localized to the nucleus as judged by DAPI staining (data not shown).
Human Maf1 and Brf2 co-localize in HeLa cells. (A) HeLa cells transfected with empty Flag vector (left upper panel), FlagBrf2 (center left panel), or FlagMaf1 (lower left panel) and localization of proteins determined using an anti-Flag antibody denoted in green. Propidium iodide (PI), center left panels, identify the nucleus of HeLa cells. Overlay of cells stained with anti-Flag and PI demonstrate the nuclear localization of FlagMaf1 and FlagBrf2, right center panels. Right panels show the DIC and overlay of the transfected HeLa cells. (B) HeLa cells transfected with either empty Flag and GFP vectors or FlagMaf1 and Brf2GFP. Right panels demonstrate location of GFP signal (green), left center panels the Flag signal (red), overlay shows co-localization of FlagMaf1 and Brf2-GFP (yellow).
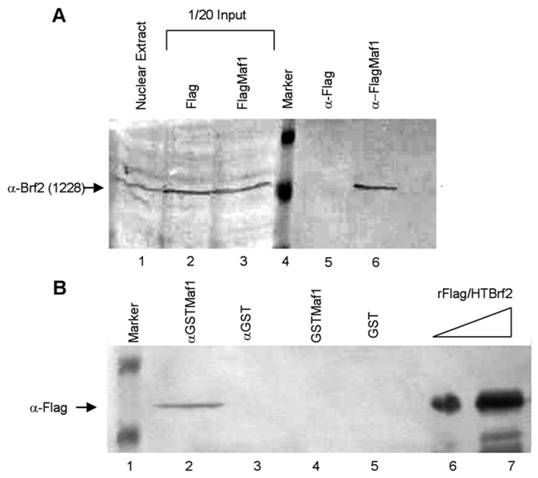
To further elucidate whether Maf1 physically associates with Brf2 we performed co-immunoprecipitation (co-IP) experiments. HeLa cells were transiently transfected with either the empty Flag vector or FlagMaf1 and nuclear extract was prepared. After incubation with M2 Flag agarose, the immunoprecipitates (IP) were extensively washed and the precipitated complexes were eluted using Flag peptide. The complexes were separated by SDS-PAGE and we immunoblotted using an anti-Brf2 antibody (CSH 1228), as previously described [13]. We find that FlagMaf1 specifically interacts with endogenous Brf2, (Figure 5A, lane 6), as no Brf2 was detected in the anti-Flag alone IP (Figure 5A, lane 5). This suggests that human Maf1 interacts with the TFIIIB subunit Brf2, but these experiments do not provide insight whether this is a direct or indirect interaction. To discriminate between these two possibilities we expressed and purified recombinant Flag/HTBrf2 and GSTMaf1 in E. coli, as previously described [13]. Using a GST pull-down assay, we demonstrate that Maf1 interacts directly with the TFIIIB subunit Brf2 (Figure 5B, lane 2). Taken together co-localization, co-immunoprecipitation, and GST pulldown assays strongly suggest that human Maf1 exerts its ability to inhibit RNA pol III U6 transcription through direct interaction with the TFIIIB subunit Brf2.
Maf1 directly associates with the TFIIB family member Brf2. (A) Nuclear extracts from HeLa cells transiently transfected with the empty Flag vector or FlagMaf1 were mixed with Flag agarose, subsequently immunoblotted with the gene specific anti-Brf2 antibody CSH 1228. Nuclear extract inputs: untransfected nuclear extract (lane 1) empty Flag vector (lane 2), and FlagMaf1 (lane 3). Immunoprecipitates: Flag vector (lane 5), and FlagMaf1 (lane 6). Arrow denotes migration location of endogenous Brf2 protein. (B) Recombinant Flag/HTBrf2 and GSTMaf1 were mixed with anti-glutathione agarose and immunoprecipitates were immunoblotted with an anti-Flag antibody. Increasing amounts of recombinant Flag/HTBrf2 (lanes 6 and 7), recombinant GST (lane 5), and recombinant GSTMaf1 (lane 4), immune complexes from anti-GST (lane 3) and anti-GSTMaf1 (lane 2) immunoprecipitations. Arrow denotes location of Flag/HT Brf2, as determined by anti-Flag antibody.
4. Discussion
In this study we have constructed RNA pol III type 2 and 3 promoter fusions to the Photinus pyralis luciferase gene that enable RNA polymerase III transcription to be monitored in vivo. We demonstrate specific monitoring of transcriptional activity from both gene internal and gene external RNA pol III promoters (Figures 1A through 1F). Although the use of the luciferase gene fused to prototypical RNA pol III promoters to monitor both type 2 and type 3 RNA pol III transciption in vivo is novel, the ability of RNA pol III to generate functional mRNAs has been documented in other studies [46]. It has been demonstrated that the human VAI promoter can drive expression of the Tat gene in vivo and that this VA-Tat RNA synthesized by RNA pol III is scanned for translational initiation, despite being uncapped [46, 47]. Examination of the luciferase coding sequence in pGL3 Basic reveals several regions with a run of four or more T residues which could potentially be used as termination signals by RNA pol III. However, it has been demonstrated that some RNA pol III transcripts such as rabbit tRNAlys, Xenopus laevis tRNAlys and the yeast tRNAser transcripts contain internal stretches of T residues indicating that a cluster of T residues are not always sufficient for efficient termination by RNA pol III [48]. This was clearly demonstrated in Xenopus laevis 5S RNA which requires a G + C rich sequence, surrounding a cluster of T residues for efficient termination by RNA pol III [49]. To ascertain if we were indeed measuring RNA pol III transcription we inserted a putative termination signal for RNA pol III, a run T residues downstream of the U6 promoter, but upstream of the start ATG of the luciferase gene of pGL3-U6, which inhibited luciferase expression (Figure 1F).
We also provide experimental evidence that a human homolog of yeast Maf1 is a repressor of type 2 and 3 RNA pol III transcription (Figure 2D) using an in vivo luciferase assay. These results are in agreement with Reina et al's [32] report that, human Maf1 inhibits RNA pol III transcription from both gene internal and gene external promoters using in vitro nuclear run on and RNA protection assays. We have confirmed that over expression of the TFIIB family members Brf1 and Brf2 alleviate Maf1 repression from gene internal and gene external RNA pol III promoters, respectively, suggesting that human Maf1 exerts its repressional activity, at least in part, through the TFIIIB complex (Figure 3). We have further identified a mechanism by which Maf1 represses U6 transcription by identifying a physical association, using co-localization, co-immunoprecipitation and GST pull-down assays, with the TFIIIB subunit Brf2 (Figure 5). Interestingly, Reina et al [32] were not able to detect a physical association between Maf1 and Brf2, using in vitro transcribed/translated Brf2 and there was no mention if the authors had attempted to detect a Maf1 and Brf2 association using the co-immunoprecipitation assay utilized to discern the Brf1 and RPC1 interaction with Maf1.
Although a role for Maf1 repressing human RNA pol III transcription in vitro has been recently demonstrated [32], our work presents further insight into the mechanism of repression in vivo. We show that interactions with the TFIIIB components Brf1 and Brf2 also play a role. Potentially, the interaction of Maf1 with Brf2 may be weaker than that with Brf1.
It is currently unclear whether phosphorylation and dephosphorylation of human Maf1 plays a critical role in the regulation of RNA pol III in a manner similar to that observed in yeast [24, 29-31]. We also provide in this report evidence that human Maf1 is a phosphoprotein (Figure 2C). It has yet to be discerned if the human homologs of the PP2A, PKA and Pkc1 will play similar roles in regulating human Maf1 [29-31]. Using the NetPhos prediction software [50], we have identified putative phosphorylation sites in Maf1 for PKC, PKA, Akt, Clk2, CK1, CK2, GSK3, CDC2, and Erk1, potentially placing Maf1 in a wide variety of signal transduction pathways, allowing Maf1 to transduce signals and thereby regulating RNA pol III transcription under a variety of growth conditions. As there are at least two forms of human TFIIIB, each requiring a different TFIIB family member for initiation from gene internal (Brf1) and gene external (Brf2) promoters; it is plausible that these different kinase(s) and phosphatase(s) may exert differential effects on these different TFIIB family members.
The generation of the in vivo FlagMaf1 construct and the characterization of an in vivo RNA pol III luciferase assay will be invaluable tools to help elucidate the signal transduction pathways Maf1 interacts with to exert its regulation on RNA pol III transcription.
Finally, the characterization of an in vivo RNA polymerase III luciferase assay provides an advantage to study the mechanisms of both gene –internal and –external RNA pol III transcription in intact mammalian cells. This assay will allow us to monitor RNA pol III activity under a variety of conditions, including: DNA damage, oxidative stress, mitogenic stimulus, and environmental toxins. This methodology will become an invaluable tool to rapidly screen compounds for their ability to inhibit or stimulate RNA pol III transcription. Thus, this RNA pol III luciferase assay could be used as a high through put screening method to identify and characterize pharmacological compounds capable of inhibiting deregulated cellular growth, a hallmark trait in variety of cancers.
Acknowledgements
We are grateful to Dr. Chris Bazinet, St. John's University for reagents. We are indebted to Dr. Ales Vancura, St. John's University for reagents and for critical reading of this manuscript. We thank Dr. Nouria Hernandez, Cold Spring Harbor Laboratory, for antibodies. This work was supported in part by the Henry Luce foundation (L. Schramm), a St. John's University faculty growth grant (L. Schramm) and by Department of Education's Graduate Assistance in Areas of National Need (GAANN) Grant P200A010130 (I. Veras and S. Cabarcas).
Conflict of Interests
The authors have declared that no conflicts of interests exist.
References
1. Roeder R.G. Nuclear RNA polymerases: role of general initiation factors and cofactors in eukaryotic transcription. Methods Enzymol. 1996;273:165-71
2. Schramm L, Hernandez N. Recruitment of RNA polymerase III to its target promoters. Genes Dev. 2002;16(20):2593-620
3. White R.J. RNA polymerases I and III, growth control and cancer. Nat Rev Mol Cell Biol. 2005;6(1):69-78
4. Gabrielsen O.S, Sentenac A. RNA polymerase III (C) and its transcription factors. Trends Biochem Sci. 1991;16(11):412-6
5. Desai N. et al. Two steps in Maf1-dependent repression of transcription by RNA polymerase III. J Biol Chem. 2005;280(8):6455-62
6. Willis I.M. RNA polymerase III. Genes, factors and transcriptional specificity. Eur J Biochem. 1993;212(1):1-11
7. Paule M.R, White R.J. Survey and summary: transcription by RNA polymerases I and III. Nucleic Acids Res. 2000;28(6):1283-98
8. Huang Y, Maraia R.J. Comparison of the RNA polymerase III transcription machinery in Schizosaccharomyces pombe, Saccharomyces cerevisiae and human. Nucleic Acids Res. 2001;29(13):2675-90
9. Paule M.R. et al. Initiation and regulation mechanisms of ribosomal RNA transcription in the eukaryote Acanthamoeba castellanii. Mol Cell Biochem. 1991;104(1-2):119-26
10. Geiduschek E.P, Kassavetis G.A. The RNA polymerase III transcription apparatus. J Mol Biol. 2001;310(1):1-26
11. Kumar A. et al. Transcription factor IIIB: the architecture of its DNA complex, and its roles in initiation of transcription by RNA polymerase III. Cold Spring Harb Symp Quant Biol. 1998;63:121-9
12. Kelter A.R, Herchenbach J., Wirth B. The transcription factor-like nuclear regulator (TFNR) contains a novel 55-amino-acid motif repeated nine times and maps closely to SMN1. Genomics. 2000;70(3):315-26
13. Schramm L. et al. Different human TFIIIB activities direct RNA polymerase III transcription from TATA-containing and TATA-less promoters. Genes Dev. 2000;14(20):2650-63
14. Teichmann M, Wang Z, Roeder R.G. A stable complex of a novel transcription factor IIB- related factor, human TFIIIB50, and associated proteins mediate selective transcription by RNA polymerase III of genes with upstream promoter elements. Proc Natl Acad Sci U S A. 2000;97(26):14200-5
15. McCulloch V. et al. Alternatively spliced hBRF variants function at different RNA polymerase III promoters. Embo J. 2000;19(15):4134-43
16. White R.J. et al. Cell cycle regulation of RNA polymerase III transcription. Mol Cell Biol. 1995;15(12):6653-62
17. Leresche A, Wolf V.J, Gottesfeld J.M. Repression of RNA polymerase II and III transcription during M phase of the cell cycle. Exp Cell Res. 1996;229(2):282-8
18. Brown T.R. et al. RNA polymerase III transcription: its control by tumor suppressors and its deregulation by transforming agents. Gene Expr. 2000;9(1-2):15-28
19. Felton-Edkins Z.A. et al. Direct regulation of RNA polymerase III transcription by RB, p53 and c-Myc. Cell Cycle. 2003;2(3):181-4
20. White R.J. RNA polymerase III transcription--a battleground for tumour suppressors and oncogenes. Eur J Cancer. 2004;40(1):21-7
21. White R.J. RNA polymerase III transcription and cancer. Oncogene. 2004;23(18):3208-16
22. White R.J. Transcription factor IIIB: An important determinant of biosynthetic capacity that is targeted by tumour suppressors and transforming proteins. Int J Oncol. 1998;12(4):741-8
23. Roberts D.N. et al. The RNA polymerase III transcriptome revealed by genome-wide localization and activity-occupancy relationships. Proc Natl Acad Sci U S A. 2003;100(25):14695-700
24. Upadhya R, Lee J, Willis I.M. Maf1 is an essential mediator of diverse signals that repress RNA polymerase III transcription. Mol Cell. 2002;10(6):1489-94
25. Willis I.M, Desai N, Upadhya R. Signaling repression of transcription by RNA polymerase III in yeast. Prog Nucleic Acid Res Mol Biol. 2004;77:323-53
26. Geiduschek E.P, Kassavetis G.A. Transcription: adjusting to adversity by regulating RNA polymerase. Curr Biol. 2006;16(19):R849-51
27. Willis I.M, Moir R.D. Integration of nutritional and stress signaling pathways by Maf1. Trends Biochem Sci. 2007;32(2):51-53
28. Pluta K. et al. Maf1p, a negative effector of RNA polymerase III in Saccharomyces cerevisiae. Mol Cell Biol. 2001;21(15):5031-40
29. Moir R.D. et al. Protein kinase A regulates RNA polymerase III transcription through the nuclear localization of Maf1. Proc Natl Acad Sci U S A. 2006;103(41):15044-9
30. Oficjalska-Pham D. et al. General Repression of RNA Polymerase III Transcription Is Triggered by Protein Phosphatase Type 2A-Mediated Dephosphorylation of Maf1. Mol Cell. 2006;22(5):623-632
31. Roberts D.N. et al. Dephosphorylation and Genome-Wide Association of Maf1 with Pol III-Transcribed Genes during Repression. Mol Cell. 2006;22(5):633-44
32. Reina J.H, Azzouz T.N, Hernandez N. Maf1, a New Player in the Regulation of Human RNA Polymerase III Transcription. PLoS ONE. 2006;1:e134
33. Ohkawa J, Taira K. Control of the functional activity of an antisense RNA by a tetracycline-responsive derivative of the human U6 snRNA promoter. Hum Gene Ther. 2000;11(4):577-85
34. Ford E, Strubin M, Hernandez N. The Oct-1 POU domain activates snRNA gene transcription by contacting a region in the SNAPc largest subunit that bears sequence similarities to the Oct-1 coactivator OBF-1. Genes Dev. 1998;12(22):3528-40
35. Sadowski C.L. et al. Targeting TBP to a non-TATA box cis-regulatory element: a TBP-containing complex activates transcription from snRNA promoters through the PSE. Genes Dev. 1993;7(8):1535-48
36. Mittal V, Hernandez N. Role for the amino-terminal region of human TBP in U6 snRNA transcription. Science. 1997;275(5303):1136-40
37. Rohan R.M, Ketner G. Point mutations in the regulatory region of the human adenoviral VAI gene. J Biol Chem. 1983;258(19):11576-81
38. Lobo S.M, Hernandez N. A 7 bp mutation converts a human RNA polymerase II snRNA promoter into an RNA polymerase III promoter. Cell. 1989;58(1):55-67
39. Rumi M. et al. RNA polymerase II mediated transcription from the polymerase III promoters in short hairpin RNA expression vector. Biochem Biophys Res Commun. 2006;339:540-547
40. Saxena A. et al. Structure-Function Analysis of the Human TFIIB-Related Factor II Protein Reveals an Essential Role for the C-Terminal Domain in RNA Polymerase III Transcription. Mol Cell Biol. 2005;25(21):9406-18
41. Kedinger C. et al. Alpha-amanitin: a specific inhibitor of one of two DNA-pendent RNA polymerase activities from calf thymus. Biochem Biophys Res Commun. 1970;38(1):165-71
42. Chu W.M, Ballard R.E, Schmid C.W. Palindromic sequences preceding the terminator increase polymerase III template activity. Nucleic Acids Res. 1997;25(11):2077-82
43. TESS: Transcription Element Search Software on the WWW. 2007 Schug J and Overton GC. http://www.cbil.upenn.edu/tess/
44. Chen D. et al. TBP dynamics in living human cells: constitutive association of TBP with mitotic chromosomes. Mol Biol Cell. 2002;13(1):276-84
45. Schoenen F, Wirth B. The zinc finger protein ZNF297B interacts with BDP1, a subunit of TFIIIB. Biol Chem. 2006;387(3):277-84
46. Gunnery S, Mathews M.B. Functional mRNA can be generated by RNA polymerase III. Mol Cell Biol. 1995;15(7):3597-607
47. Gunnery S, Maivali U, Mathews M.B. Translation of an uncapped mRNA involves scanning. J Biol Chem. 1997;272(34):21642-6
48. Gunnery S, Ma Y, Mathews M.B. Termination sequence requirements vary among genes transcribed by RNA polymerase III. J Mol Biol. 1999;286(3):745-57
49. Bogenhagen D.F, Brown D.D. Nucleotide sequences in Xenopus 5S DNA required for transcription termination. Cell. 1981;24(1):261-70
50. Blom N, Gammeltoft S, Brunak S. Sequence- and structure-based prediction of eukaryotic protein phosphorylation sites. Journal of Molecular Biology. 1999;294(5):1351-1362
Author contact
![]() Correspondence to: Laura Schramm, Department of Biological Sciences, 8000 Utopia Parkway, Queens, NY 11439; Email: schrammLedu; Fax: 718-990-5958
Correspondence to: Laura Schramm, Department of Biological Sciences, 8000 Utopia Parkway, Queens, NY 11439; Email: schrammLedu; Fax: 718-990-5958
Received 2007-3-1
Accepted 2007-4-24
Published 2007-5-1
