10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2009; 5(2):192-200. doi:10.7150/ijbs.5.192 This issue Cite
Short Research Communication
BMP-13 Emerges as a Potential Inhibitor of Bone Formation
1. Orthopaedic Research Institute and Department of Orthopaedic Surgery, St George Hospital, University of New South Wales, Sydney, Australia
2. Blood Stem Cell and Cancer Research Unit, Department of Haematology, St. Vincent's Hospital Sydney, University of New South Wales, Sydney, Australia
Received 2008-12-17; Accepted 2009-2-9; Published 2009-2-13
Abstract
Bone morphogenetic protein-13 (BMP-13) plays an important role in skeletal development. In the light of a recent report that mutations in the BMP-13 gene are associated with spine vertebral fusion in Klippel-Feil syndrome, we hypothesized that BMP-13 signaling is crucial for regulating embryonic endochondral ossification. In this study, we found that BMP-13 inhibited the osteogenic differentiation of human bone marrow multipotent mesenchymal stromal cells (BM MSCs) in vitro. The endogenous BMP-13 gene expression in MSCs was examined under expansion conditions. The MSCs were then induced to differentiate into osteoblasts in osteo-inductive medium containing exogenous BMP-13. Gene expression was analysed by real-time PCR. Alkaline phosphatase (ALP) expression and activity, proteoglycan (PG) synthesis and matrix mineralization were assessed by cytological staining or ALP assay. Results showed that endogenous BMP-13 mRNA expression was higher than BMP-2 or -7 during MSC growth. BMP-13 supplementation strongly inhibited matrix mineralization and ALP activity of osteogenic differentiated MSCs, yet increased PG synthesis under the same conditions. In conclusion, BMP-13 inhibited osteogenic differentiation of MSCs, implying that functional mutations or deficiency of BMP-13 may allow excess bone formation. Our finding provides an insight into the molecular mechanisms and the therapeutic potential of BMP-13 in restricting pathological bone formation.
Keywords: BMP-13, GDF6, CDMP-2, osteogenic differentiation, mesenchymal stromal cells
1. Introduction
Bone morphogenetic proteins (BMPs) are members of the transforming growth factor-β (TGF-β) superfamily, which includes the families of TGF-βs, BMPs, activins and inhibins. BMPs were originally identified because of their ability to induce endochondral bone formation and are important regulators of key events in the process of bone formation during embryogenesis, postnatal growth, remodelling and regeneration of the skeleton [1, 2, 3]. Recombinant human BMP-2 and BMP-7 are used clinically to augment bone formation in spinal fusion and many other applications in which bone induction is desired [4].
BMP-13, also known as growth differentiation factor 6 (GDF6) and cartilage-derived morphogenetic protein-2 (CDMP-2), was first identified and isolated as a component of bovine cartilage [5]. Expression of BMP-13 has since been detected in a variety of mesenchymal derivatives such as tendon and cartilage, but its biological function is poorly defined [6]. The majority of studies focus on connective tissue healing where de-novo implantation of BMP-13 induces neo-tendon/ligament formation in rats [7][8], and the expression of chondrocyte markers such as proteoglycans [6]. Comparisons with other BMPs known for their ability to stimulate bone growth have suggested that BMP-13 is not strongly osteo-inductive [9, 10, 11, 12]. Indeed, BMP-13 expression was absent from osteoblasts or newly formed osteocytes, rather BMP-13 was strongly detected in chondrocytes in a human osteophyte (bone spur) study [13]. In vivo, mutational inactivation of the BMP-13 gene is associated with Klippel-Feil syndrome (KFS), characterised by congenital fusion of the cervical spine vertebrae [14], and caused defects in joint, ligament, and cartilage formation in a transgenic mouse model [15].
The function of BMPs in bone remodelling and fracture repair is associated with the recruitment of stem cells from surrounding tissues and with effects on the osteogenic lineage commitment of these cells. Bone marrow multipotent mesenchymal stromal cells (BM MSCs) are capable of differentiating into multiple cell types including osteoblasts and chondrocytes, which makes them an attractive cell source for tissue repair. We need to understand the complex differentiation pathways driven by multiple growth factors in order to select appropriate growth factors for clinical use.
Given the phenotypic effects of joint fusions and joint aberrations caused by BMP-13 mutations [14], we hypothesized that BMP-13 plays an important regulatory role in the balance between osteogenesis and chondrogenesis during early embryonic development. The purpose of this study was to evaluate the role of BMP-13 in determining the lineage fate of BM MSCs. BMP-13 was found to inhibit osteogenic differentiation, demonstrated by the deceleration of calcium mineralisation of the extracellular matrix and reduced alkaline phosphatase induction in BM MSCs. BMP-7, known to be a potent osteo-inductive agent [16], was used as a positive control throughout the study. Our data suggest a potential role of BMP-13 in directing stem cell fates in the development of structural tissues.
2. Materials and Methods
Cell isolation and cultivation
Human bone marrow was collected from surgically discarded tissue during hip replacement procedures under written informed consent following local ethics committee approval. BM MSCs were isolated by the antibody mediated negative selection, Ficoll-Paque density gradient centrifugation and plastic adherence as previously described [17]. The cells were cultured in growth medium (GM) consisting of 60% Dulbecco's Modified Eagle's Medium - low glucose (DMEM-LG; Invitrogen, Carlsbad, CA, USA), 40% MCDB-201 medium, 1% insulin transferrin selenium (ITS), 1% linoleic acid / bovine serum albumin (BSA), 1 nM dexamethasone, 30 µg/ml ascorbic acid 2-phosphate (Sigma-Aldrich, St. Louis, MO, USA), 100 U/ml penicillin, 100 µg/ml streptomycin and 10% foetal bovine serum (FBS). The cells were incubated at 37ºC in a humidified atmosphere with 5% CO2. The medium was changed twice weekly.
Osteogenic differentiation
BM MSCs at Passage 2 - 3 were harvested using a standard method. For osteogenic differentiation, 1 x 105 cells per well of BM MSCs were seeded in 24-well culture plates. The MSCs were induced approximately 24 hours later using the published method [18] with the osteo-inductive medium (OM) consisting of DMEM-LG, 10% FBS, 10 mM β-glycerophosphate, 100 nM dexamethasone, 50 µg/ml ascorbic acid-2-phosphate, 100 U/ml penicillin and 100 µg/ml streptomycin in the absence or presence of recombinant human (rh) BMP-13 (PeproTech, Rocky Hill, NJ) at 100, 300 and 500 ng/ml. Undifferentiated MSCs were cultured in parallel in growth medium as negative control. As for the positive control, MSCs were induced in osteo-inductive medium supplemented with 100 ng/ml of rhBMP-7 (a gift from Stryker Biotech). Cells were kept at 37ºC, 5% CO2 for up to 21 days and the media were changed twice weekly.
Cytological staining
Alkaline phosphatase (ALP) expression as the early osteogenic marker was determined at day 14 of MSC differentiation using Fast blue staining. Calcium mineralization of extracellular matrix was visualized at days 14 and 21 as an advanced osteogenic marker using Alizarin red S staining. Briefly, the cell layers of differentiated MSCs were washed with phosphate-buffered saline (PBS), fixed with 4% paraformaldehyde for 5 min. The membrane-bound ALP was detected by incubation in 0.1% Fast blue RR salt and naphthol AS-BI phosphate solution at pH 9.4, 37ºC for 20 min. For Alizarin red S staining, the cells were washed and fixed as above and then immersed in the 2% Alizarin red S solution at pH 4.2 for 10 min followed by 3 washes in H2O. Proteoglycan (PG) deposition in the presence and absence of BMP-13 was detected by staining with 1% Alcian blue at day 14 of culture using a previously reported method [19].
Alkaline phosphatase assay
BM MSCs were induced to osteogenic differentiation under the above described osteogenic induction conditions with supplementation of 3 - 800 ng/ml of BMP-13 (Peprotech) at 37ºC, 5% CO2 for 14 days. Cells were washed with PBS and then lysed for 1 h with 100 µl of 1% NP40 in ALP buffer (0.1 M glycine, pH 9.6, 1 mM MgCl2, 1 mM ZnCl2). ALP activity was determined by incubating lysed cells with 100 µl of p-Nitrophenyl phosphate substrate solution (Sigma-Aldrich) for 10 min and measuring the absorbance at 405 nm [20].
RNA extraction, cDNA synthesis and real-time PCR
Total RNA was isolated from undifferentiated and differentiated MSCs using TRIzol reagent (Invitrogen) and RNeasy kit (Qiagen, Dusseldorf, Germany) following manufacturers' instructions. cDNA was generated by reverse transcription of 1 µg total RNA using SuperScript III first-strand synthesis kit (Invitrogen). The 1:40 diluted cDNA was used in 20 µl-reactions for real-time PCR analysis using a Rotor-Gene RG3000 system (Corbett Life Science, Sydney). The thermal profile for all reactions was as follows: 5 min at 95ºC, followed by 40 amplification cycles of 15 sec at 95ºC, 30 sec at 60ºC and 30 sec at 72ºC [19]. The GenBank access numbers, real-time PCR primer pairs and product sizes are listed in Table 1. Relative expression levels were calculated as a ratio to the average value of house-keeping genes, glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and hypoxanthine phosphoribosyltransferase 1 (HPRT1).
Primer pairs used for real-time PCR.
| Gene Symbol | GenBank Accession Number | Sequence (5' - 3') | Product Size (bp) |
|---|---|---|---|
| BMP2 | NM_001200 | F - TTTCCATGTGGACGCTCTTT | 118 |
| R - GGAAGCAGCAACGCTAGAAG | |||
| BMP7 | NM_001719 | F - GAGTGTGCCTTCCCTCTGAA | 140 |
| R - TAGAGGACGGAGATGGCATT | |||
| BMP13 | NM_001001557 | F - TGCCAGCTTTTTCCAGTCTT | 85 |
| R - AGGAGTGTGCGAGAGATCGT | |||
| ALPL | NM_000478 | F - GACAAGAAGCCCTTCACTGC | 120 |
| R - AGACTGCGCCTGGTAGTTGT | |||
| BGLAP | NM_199173 | F - GGCGCTACCTGTATCAATGG | 106 |
| R - TCAGCCAACTCGTCACAGTC | |||
| COL1A1 | NM_000088 | F - GAGAGCATGACCGATGGATT | 149 |
| R - ATGTAGGCCACGCTGTTCTT | |||
| SP7 | NM_152860 | F - ATGTCTTGCCCCAAGATGTC | 115 |
| R - TATCCACCACTACCCCCAGT | |||
| SPP1 | NM_000582 | F - GCCGAGGTGATAGTGTGGTT | 101 |
| R - TGAGGTGATGTCCTCGTCTG | |||
| RUNX2 | NM_004348 | F - GCCTAGGCGCATTTCAGA | 66 |
| R - CTGAGAGTGGAAGGCCAGAG | |||
| TAZ | NM_015472 | F - CAGCAATGTGGATGAGATGG | 66 |
| R - TGGGGATTGATGTTCATGG | |||
| NOG | NM_005450 | F - AGCGAGATCAAAGGGCTAGA | 86 |
| R - TGTAACTTCCTCCGCAGCTT | |||
| GAPDH | NM_002046 | F - AATCCCATCACCATCTTCCA | 82 |
| R - TGGACTCCACGACGTACTCA | |||
| HPRT1 | NM_000194 | F - GACCAGTCAACAGGGGACAT | 132 |
| R - CCTGACCAAGGAAAGCAAAG |
Statistics
Data is presented as the mean ± Standard Deviation (SD). Statistical analyses were performed using the Student's t test to analyse gene expression data. Differences between undifferentiated (CTL) and differentiated (OM, BMP7 or BMP13) BM MSCs were considered statistically significant at p < 0.05 (*), p < 0.01 (**) and p < 0.005 (***).
3. Results and Discussion
BMP-13 gene expression in BM MSCs
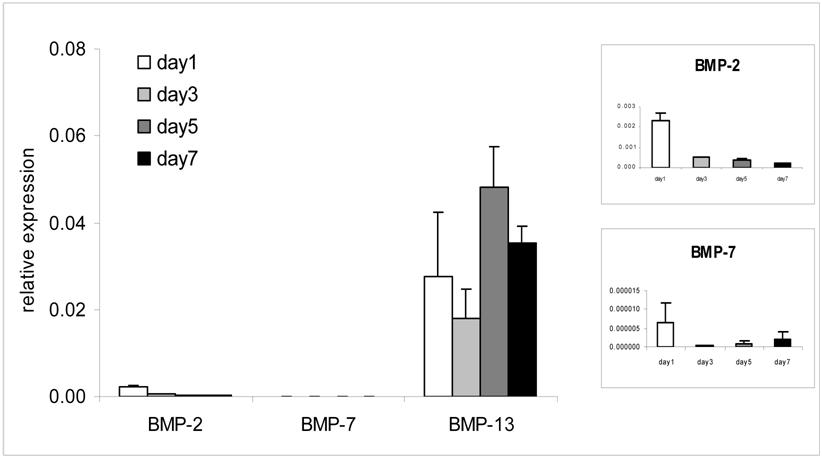
BM MSCs grown in monolayer culture in growth medium containing 10% FBS were harvested at days 1, 3, 5, and 7 for gene expression analysis (Fig. 1). The endogenous expression of BMP-13 was detected at all time points, showing considerably higher levels than either BMP-2 or BMP-7. Expression increased over time, peaking at day 5-7. Whilst literature reports indicate BMP-13 expression in a variety of tissues localised to sites of cell proliferation or tissue re-modelling [6], BMP-13 expression in BM MSCs has only previously been reported in mouse [21]. Our finding suggests an important regulatory role of BMP-13 in BM MSCs which might be different from the other more osteogenic BMPs.
BMP-13 inhibits osteogenic differentiation of BM MSCs
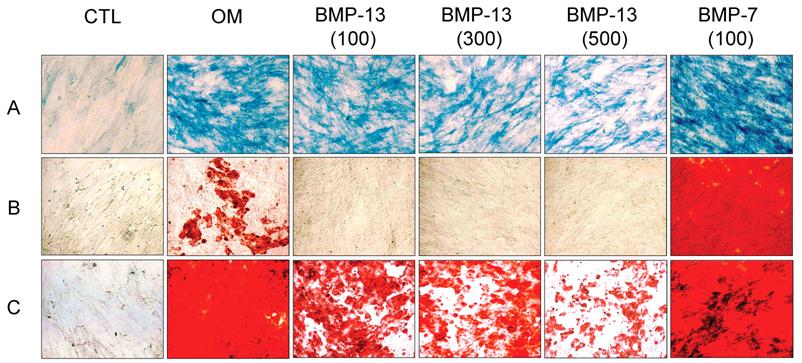
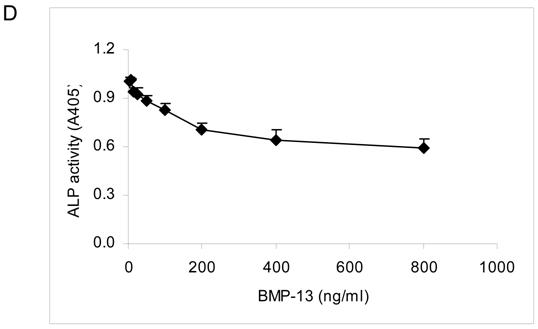
Cytological staining with Fast blue and Alizarin red S showed that BM MSCs cultured in osteo-inductive medium for 14-21 days were differentiated into osteoblasts, with elevated expression of cell surface alkaline phosphatase (ALP) and calcium mineralisation of the extracellular matrix compared to MSCs grown under expansion conditions where staining was negligible (Fig. 2). Addition of BMP-13 at 100, 300 and 500 ng/ml in osteo-inductive medium resulted in a dose-dependent decrease in detectable ALP (Fig. 2A). A parallel dose dependent inhibition of calcium mineralisation, a later marker of osteogenic differentiation, was observed using Alizarin red S staining over both 14 and 21 days (Fig. 2B and C). In contrast, 100 ng/ml of BMP-7 dramatically increased both ALP expression and matrix mineralization in osteo-differentiated MSCs at day 14 (Fig. 2B) and day 21 (Fig. 2C), which represents an enhancement or acceleration of osteogenic differentiation under these conditions. Whilst not shown here, BMP-7 enhancement of osteogenic differentiation is also dose-dependent (data not shown; Shen et al 2008, manuscript in preparation). The effect of BMP-13 on ALP in MSCs was further illustrated in measurements of ALP activity (Fig. 2D). BMP-13 dose dependently inhibited ALP activity to a maximum of 40%, confirming the results obtained from Fast blue staining (Fig 2A).
Endogenous BMP-13 gene expression in undifferentiated BM MSCs by real-time PCR analysis. BMP-13 gene expression was detected at days 1, 3, 5 and 7 in MSC expansion cultures and compared to that of BMP-2 and -7 in parallel. Relative expression was calculated as a ratio to the average value of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and hypoxanthine phosphoribosyltransferase 1 (HPRT1). The individual BMP-2 and -7 time-course expression levels are shown in insets.
Effects of BMP-13 on alkaline phosphatase (ALP) expression and matrix mineralization of osteogenic differentiated BM MSCs by cytological staining. MSC differentiation was induced in osteo-inductive medium and supplemented with 100, 300, and 500 ng/ml of BMP-13 or 100 ng/ml of BMP-7 respectively for 14-21 days. Active ALP was detected by a blue cleavage product using a Fast blue RR salt dye (A). Matrix mineralization was visualised by Alizarin red S staining at days 14 (B) and 21 (C). Undifferentiated MSCs were used as the negative control. CTL = undifferentiated negative control; OM = osteo-inductive medium; BMP-7 = OM+BMP-7; BMP-13 = OM+BMP-13 for all figures. The BMP concentrations at ng/ml are listed in brackets. Original magnification x 100 for all images. ALP activity was measured in MSCs after 14 days differentiation in osteo-inductive medium with or without BMP-13 (3-800 ng/ml). ALP activity is represented as A405 mean values from quadruplicate cultures (D).
Our results demonstrate that the presence of BMP-13 in osteo-inductive media can, despite an environment promoting osteogenic differentiation, prevent MSCs from differentiating into a mature osteogenic phenotype. This represents a dramatically different role for BMP-13 to the better characterised BMP-2 and BMP-7. BMP-13 was shown to enhance BMP-7 promoted ALP expression in mouse myoblast cell line C2C12 [9], but BMP-13 stimulation of ALP activity was generally at negligible to low levels in mouse BM MSCs or cell lines cultured in basal growth media [9, 10, 11, 21, 22]. Unlike previous studies, we have utilised primary human BM MSCs and the effect of BMP-13 was examined during the osteogenic differentiation of BM MSCs. Such a model system has not been previously studied and may provide deeper insights into BMP-13 function. Actually, recent reports have suggested that BMP-13 was capable of inducing the chondrogenic differentiation of mouse mesenchymal stromal or progenitor cells, based on increased chondrocytic marker expression [23][22]. Instead of promoting bone formation, BMP-13 is more likely to be a potential factor for maintaining cartilage development. Thus, whilst previous reports have suggested BMP-13 is not a potent osteogenic differentiation inducer (9, 10), our data goes one step further in showing that BMP-13 can inhibit osteoblast formation. This implies that functional mutations or deficiency of BMP-13 may result in excessive endochondral ossification, disturbing the development of skeletal tissues.
The regulatory role of BMP-13 in osteogenic gene expression of BM MSCs
In order to evaluate more specifically the potential mechanism of BMP-13 in the inhibition of osteogenic differentiation of BM MSCs, we performed gene expression analysis of markers for osteogenesis using real-time PCR assay (Fig. 3).
Gene expression analysis of osteogenic markers and BMP antagonists in differentiated BM MSCs by real-time PCR analysis. The osteogenic differentiation of BM MSCs was induced by osteo-inductive media in the presence and absence of 100 ng/ml BMP-7 or 300 ng/ml BMP-13 for 14 days. Undifferentiated MSCs were used as negative control. The gene expression level was calculated as a relative ratio to the average value of house-keeping genes, GAPDH and HPRT1.
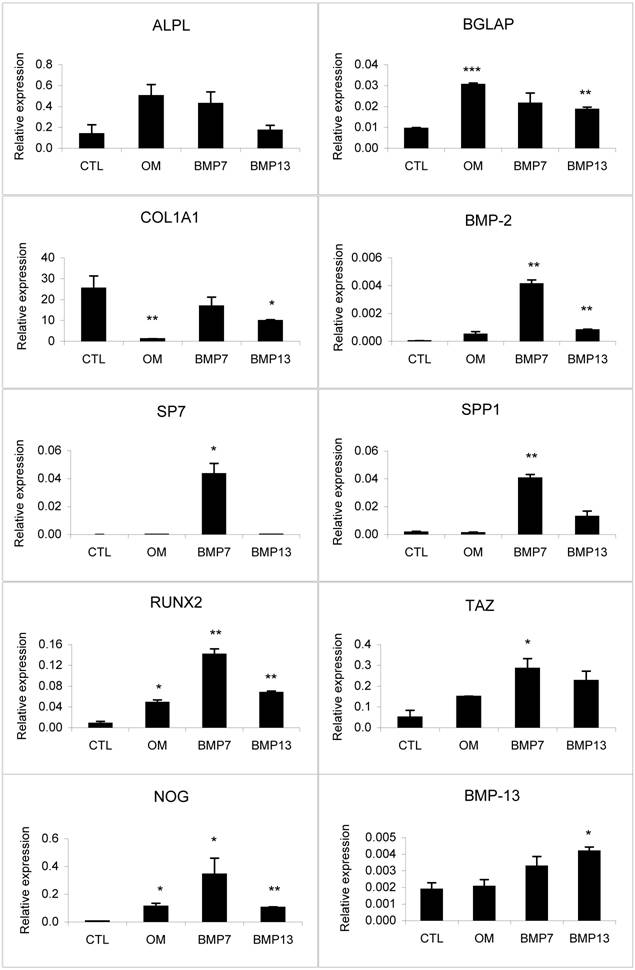
Osteogenic medium alone induced the up-regulation of specific lineage marker expression [14, 15] such as alkaline phosphatase (ALPL), bone gamma-carboxyglutamate (gla) protein (BGLAP), runt-related transcription factor 2 (RUNX2), and TAZ (WW-domain-containing transcription regulator 1, WWTR1) (Fig. 3). The addition of BMP-13 inhibited ALPL mRNA expression, consistent with the ALP protein staining data discussed above. BMP-13 had no effect on the expression of crucial osteogenic transcription factor, osterix (SP7), whereas BMP-7 dramatically increased its expression by 1300-fold (Fig. 3). BMP-13 also showed considerably less induction of osteogenic markers, type I collagen (COL1A1), osteopontin (secreted phosphoprotein 1, SPP1) and RUNX2 compared to BMP-7 in the same experiment. BMP-13 somewhat surprisingly mediated up-regulation of SPP1 mRNA, although once again this was also to a much lower level than stimulated by BMP-7, where SPP1 was dramatically up-regulated. BMP-7 also induced up-regulation of BMP-2 expression, suggesting a potential mechanism for BMP-7 action via augmentation of BMP-2. BMP-13 gene expression was similar whether BM MSCs were cultured in growth medium or osteo-inductive medium. BMP-13 supplementation appeared to up-regulate its own gene expression under the osteogenic conditions. Additionally, the gene expression of noggin (NOG), a BMP antagonist, seems less responsive to BMP-13 than to BMP-7 stimulation (Fig. 3).
In summary, the gene expression analysis showed that BMP-13 down-regulated or failed to up-regulate crucial osteogenic marker gene expression, when compared to the gene expression pattern obtained with BMP-7 stimulation. BMP-13 was substantially less inductive of osteogenic marker genes, and the resulting phenotypic effect detected in cytological staining experiments suggests that such level of gene expression did not enhance the osteogenic differentiation process.
BMP-13 promotes proteoglycan synthesis of BM MSCs
Proteoglycan (PG) synthesis or accumulation in the extracellular matrix has been used as a marker of chondrogenesis of BM MSCs and BMP-13 has been shown to stimulate PG production [6, 11, 24]. In this study, we evaluated PG levels in the osteogenic differentiation of BM MSCs supplemented with BMP-13 using Alcian blue staining. As shown in Fig. 4, the presence of BMP-13 resulted in increased PG deposition or accumulation in the extracellular matrix of not only undifferentiated MSCs in growth medium, but even differentiated MSCs under specific osteo-inductive conditions.
Effect of BMP-13 on proteoglycan accumulation in the extracellular matrix of BM MSCs detected by a cytological staining. MSCs were cultured in growth medium (GM) and osteogenic differentiation medium (OM) with or without 300 ng/ml of BMP-13 for 14 days. At the termination of cultures, the cells were stained with Alcian blue to visualise the proteoglycans. Original magnification x 100 for all images.
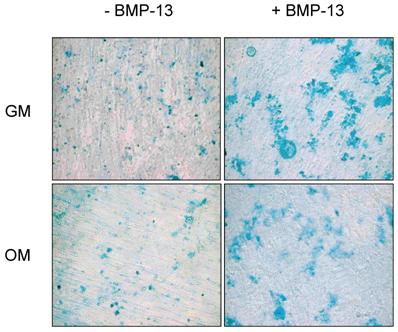
The data presented here suggest that in the presence of BMP-13, osteo-inductive signaling is inhibited or retarded, whilst expression of more chondrogenic gene products is promoted. By inference, the absence of BMP-13 signaling at crucial stages of differentiation could be expected to cause an over-induction of osteogenic cell phenotypes, contributing to abnormal endochondral ossification of cartilaginous tissue. Strong expression of BMP-13 in the BM MSC population may play an important role in keeping osteogenic differentiation in check. Several studies have shown that BMP-13 is capable of inducing chondrogenic phenotypes, characterised by increased proteoglycan production in human foetal chondrocytes [11] and a mouse chondrocytic cell line [24]. BMP-13 is known to be expressed in the joints of mice in a particular striped pattern [15, 25] and mutations in BMP-13 gene are implicated in sporadic and familial cases of KFS, characterised by the spinal vertebral fusion [14].
In conclusion, this study demonstrates for the first time that BMP-13 potentially plays a crucial role in the delineation of BM MSC differentiation by inhibiting osteogenic lineage development, suggesting that functional mutations or deficiency of BMP-13 may result in excessive endochondral ossification. Our finding provides a clue into the possible molecular mechanisms of BMP-13 associated diseases and the therapeutic potential of BMP-13 in restricting pathological bone formation.
Acknowledgements
The authors would like to thank Dr Allen Turnbull for enabling his patients undergoing hip surgery to donate tissues.
Conflict of Interests
The authors have declared that no conflict of interest exists.
References
1. Urist MR. Bone: formation by autoinduction. Science. 1965;150:893-9
2. Wang EA, Rosen V, Cordes P, Hewick RM, Kriz MJ, Luxenberg DP, Sibley BS & Wozney JM. Purification and characterization of other distinct bone-inducing factors. Proc Natl Acad Sci U S A. 1988;85:9484-8
3. Sampath TK & Reddi AH. Dissociative extraction and reconstitution of extracellular matrix components involved in local bone differentiation. Proc Natl Acad Sci U S A. 1981;78:7599-603
4. Wozney JM. Overview of bone morphogenetic proteins. Spine. 2002;27:S2-8
5. Chang SC, Hoang B, Thomas JT, Vukicevic S, Luyten FP, Ryba NJ, Kozak CA, Reddi AH & Moos MJr. Cartilage-derived morphogenetic proteins. New members of the transforming growth factor-beta superfamily predominantly expressed in long bones during human embryonic development. J Biol Chem. 1994;269:28227-34
6. Williams LA, Bhargav D & Diwan AD. Unveiling the bmp13 enigma: redundant morphogen or crucial regulator? Int J Biol Sci. 2008;4:318-29
7. Forslund C, Rueger D & Aspenberg P. A comparative dose-response study of cartilage-derived morphogenetic protein (CDMP)-1, -2 and -3 for tendon healing in rats. J Orthop Res. 2003;21:617-21
8. Wolfman NM, Hattersley G, Cox K, Celeste AJ, Nelson R, Yamaji N, Dube JL, DiBlasio-Smith E, Nove J, Song JJ, Wozney JM & Rosen V. Ectopic induction of tendon and ligament in rats by growth and differentiation factors 5, 6, and 7, members of the TGF-beta gene family. J Clin Invest. 1997;100:321-30
9. Yeh LC, Tsai AD, Zavala MC & Lee JC. Cartilage-derived morphogenetic proteins enhance the osteogenic protein-1-induced osteoblastic cell differentiation of C2C12 cells. J Cell Physiol. 2004;201:401-8
10. Yeh LC, Tsai AD & Lee JC. Cartilage-derived morphogenetic proteins induce osteogenic gene expression in the C2C12 mesenchymal cell line. J Cell Biochem. 2005;95:173-88
11. Erlacher L, McCartney J, Piek E, ten Dijke P, Yanagishita M, Oppermann H & Luyten FP. Cartilage-derived morphogenetic proteins and osteogenic protein-1 differentially regulate osteogenesis. J Bone Miner Res. 1998;13:383-92
12. Bobacz K, Ullrich R, Amoyo L, Erlacher L, Smolen JS & Graninger WB. Stimulatory effects of distinct members of the bone morphogenetic protein family on ligament fibroblasts. Ann Rheum Dis. 2006;65:169-77
13. Zoricic S, Maric I, Bobinac D & Vukicevic S. Expression of bone morphogenetic proteins and cartilage-derived morphogenetic proteins during osteophyte formation in humans. J Anat. 2003;202:269-77
14. Tassabehji M, Fang ZM, Hilton EN, McGaughran J, Zhao Z, de Bock CE, Howard E, Malass M, Donnai D, Diwan A, Manson FD, Murrell D & Clarke RA. Mutations in GDF6 are associated with vertebral segmentation defects in Klippel-Feil syndrome. Hum Mutat. 2008;29:1017-27
15. Settle SHJr, Rountree RB, Sinha A, Thacker A, Higgins K & Kingsley DM. Multiple joint and skeletal patterning defects caused by single and double mutations in the mouse Gdf6 and Gdf5 genes. Dev Biol. 2003;254:116-30
16. Giannoudis PV, Tzioupis C. Clinical applications of BMP-7: the UK perspective. Injury. 2005;36(Suppl 3):S47-50
17. Khoo ML, Shen B, Tao H & Ma DD. Long-term serial passage and neuronal differentiation capability of human bone marrow mesenchymal stem cells. Stem Cells Dev. 2008;17:883-96
18. Caterson EJ, Nesti LJ, Danielson KG & Tuan RS. Human marrow-derived mesenchymal progenitor cells: isolation, culture expansion, and analysis of differentiation. Mol Biotechnol. 2002;20:245-56
19. Shen B, Wei AQ, Tao H, Ma DDF & Diwan AD. BMP-2 Enhances TGF-beta3-Mediated Chondrogenic Differentiation of Human Bone Marrow Multipotent Mesenchymal Stromal Cells in Alginate Bead Culture. Tissue Eng Part A. 2008 [Epub ahead of print]
20. Kirsch T, Nickel J & Sebald W. BMP-2 antagonists emerge from alterations in the low-affinity binding epitope for receptor BMPR-II. Embo J. 2000;19:3314-24
21. Gruber R, Mayer C, Schulz W, Graninger W, Peterlik M, Watzek G, Luyten FP & Erlacher L. Stimulatory effects of cartilage-derived morphogenetic proteins 1 and 2 on osteogenic differentiation of bone marrow stromal cells. Cytokine. 2000;12:1630-8
22. Nochi H, Sung JH, Lou J, Adkisson HD, Maloney WJ & Hruska KA. Adenovirus mediated BMP-13 gene transfer induces chondrogenic differentiation of murine mesenchymal progenitor cells. J Bone Miner Res. 2004;19:111-22
23. Tian H, Yang S, Xu L, Zhang Y & Xu W. Chondrogenic differentiation of mouse bone marrow mesenchymal stem cells induced by cartilage-derived morphogenetic protein-2 in vitro. J Huazhong Univ Sci Technolog Med Sci. 2007;27:429-32
24. Li J, Kim KS, Park JS, Elmer WA, Hutton WC & Yoon ST. BMP-2 and CDMP-2: stimulation of chondrocyte production of proteoglycan. J Orthop Sci. 2003;8:829-35
25. Storm EE & Kingsley DM. Joint patterning defects caused by single and double mutations in members of the bone morphogenetic protein (BMP) family. Development. 1996;122:3969-79
Author biography
Dr Ashish Diwan heads the Disc Biology Group, which focuses on understanding the molecular mechanisms of intervertebral disc degeneration. The group is housed in the Orthopaedic Research Institute, St George Clinical School which is part of the University of New South Wales in Australia. Dr Diwan is a leading and internationally respected orthopaedic surgeon and research scientist with awards for outstanding and innovative research, including the prestigious Merrill Lynch Global Innovation Distinguished Award and grant in 2000 for pioneering works applying molecular biology to fracture healing. His team has received many awards recognising the excellence and innovation of the work: 1) Best Poster Presentation award, ISSLS, Geneva 2008; 2) Lyman Smith Award for Best Scientific Work - the Intradiscal Therapy Society Annual Scientific Meeting, June 2007, Albi (Toulouse) France; 3) Best Presentation award - International Society for the Study of Lumbar Spine- Bergen Norway June 2006; 4) Finalist Medtronic Best Paper Award - Spine Society of Australia, April 2006; 5) New Investigator Research Award - Finalist - The Orthopaedic Research Society - Chicago 2006; 6) Orthotec Best Poster Award - Spine Society of Australia April 2005. The group formed in 2000 and established an extremely fruitful collaboration in 2003 with Prof. David Ma's Stem Cell Biology Group in St Vincent's Hospital Sydney in order to better pursue aspects of stem cell therapy for intervertebral disc degeneration.
Dr Bojiang Shen is a research scientist with 10 years of research experience in the orthopedic area. Dr Shen has a special interest in elucidating the molecular mechanisms of disc degeneration and developing regeneration strategies, with a best paper award from European Spine Journal 2002/2003. Currently she focuses on the action of BMPs in stem cell differentiation and their potential application in disc repair.
![]() Correspondence to: Dr Bojiang Shen, Orthopaedic Research Institute, St George Clinical School, The University of New South Wales, 4-10 South Street, Kogarah, NSW 2217. Tel: 61-2-9113 2248; Fax: 61-2-9350 3967; E-mail: b.shenedu.au
Correspondence to: Dr Bojiang Shen, Orthopaedic Research Institute, St George Clinical School, The University of New South Wales, 4-10 South Street, Kogarah, NSW 2217. Tel: 61-2-9113 2248; Fax: 61-2-9350 3967; E-mail: b.shenedu.au

 Global reach, higher impact
Global reach, higher impact