10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2011; 7(6):869-880. doi:10.7150/ijbs.7.869 This issue Cite
Research Paper
Dimerumic Acid Inhibits SW620 Cell Invasion by Attenuating H2O2-Mediated MMP-7 Expression via JNK/C-Jun and ERK/C-Fos Activation in an AP-1-Dependent Manner
1. Department of Surgery, National Taiwan University Hospital, Taipei, Taiwan.
2. Department of Biochemical Science & Technology, College of Life Science, National Taiwan University, Taipei, Taiwan.
3. Department of Surgery, Cheng-Ching General Hospital Chung-Kang Branch, Taichung, Taiwan.
Received 2011-4-11; Accepted 2011-7-12; Published 2011-7-19
Abstract
Reactive oxygen species (ROS) such as hydrogen peroxide (H2O2) in the tumor microenvironment play important roles in tumor invasion and metastasis. Recently, ROS have been reported to cause a significant increase in the production and expression of matrix metalloproteinase (MMP)-7, which is closely correlated with metastatic colorectal cancer. The present study was undertaken to evaluate the scavenging activity of dimerumic acid (DMA) for H2O2 isolated from Monascus-fermented rice to investigate the inhibitory effects of DMA on the invasive potential of SW620 human colon cancer cells, and to explore the mechanisms underlying both these phenomena. Our results showed that increased MMP-7 expression due to H2O2 exposure was mediated by activation of mitogen-activated protein kinases (MAPKs) such as Jun N-terminal kinase (JNK), extracellular-regulated kinase (ERK), and p38 kinase. DMA pretreatment suppressed activation of H2O2-mediated MAPK pathways and cell invasion. Moreover, H2O2-triggered MMP-7 production was demonstrated via JNK/c-Jun and ERK/c-Fos activation in an activating protein 1 (AP-1)-dependent manner. Taken together, these results suggest that DMA suppresses H2O2-induced cell invasion by inhibiting AP-1-mediated MMP-7 gene transcription via the JNK/c-Jun and ERK/c-Fos signaling pathways in SW620 human colon cancer cells. Our data suggest that DMA may be useful in minimizing the development of colorectal metastasis. In the future, DMA supplementation may be a beneficial antioxidant to enhance surgical outcomes.
Keywords: dimerumic acid, H2O2, MAPK, metastasis, MMP-7, ROS.
Introduction
The normal physiologic status of redox homeostasis consists of a balance between produced and scavenged reactive oxygen species (ROS). ROS and antioxidants are known to affect the expression of numerous genes and multiple signaling pathways. In fact, a variety of tumor cells constitutively produce large quantities of hydrogen peroxide (H2O2) compared to normal cells 1. Large quantities of ROS including the superoxide, H2O2, and the hydroxyl radical, surround the tumor microenvironment and play an essential role in tumor progression 2-3. Among the ROS, H2O2 acts as a second messenger for the expression of various genes, including those closely involved in tumor proliferation, angiogenesis, and metastasis 4-6.
The smallest matrix metalloproteinase (MMP), MMP-7 (matrilysin), is secreted in a 28-kDa latent form and is then activated through proteolytic removal of a 9-kDa prodomain from the N-terminus to the 19-kDa active form 7. The expression of MMP-7 correlated significantly with the presence of distant metastases in colorectal cancers (CRCs) and could be a predictive marker for patients after surgical treatment or radiotherapy 8-11. However, radiation exposure of tumor cells can also affect the neighboring bystander cells associated with H2O2 generation 12-13. Exogenous H2O2 has been shown to be an important mediator in the induction of several genes from distinct signal transduction pathways 2, 5, 14-15. H2O2-induced MMP-7 expression via JNK/AP-1 pathway activation has also been recently observed in cultured cells 16.
The hydroxamic acid derivative of dimerumic acid (DMA) isolated from Monascus-fermented rice has been conclusively proven to have radical scavenging activity 17-18. Monascus-fermented products are widely used for the preparation of fermented foods and are utilized as traditional Chinese medicine for the anti-cholesterol effects 19. In the present study, we showed that DMA was critical for H2O2-mediated upregulation of MMP-7 and cell invasion in an SW620 human colon cancer cell line that was established from a lymph node metastasis of a CRC patient 20. Moreover, DMA could effectively prevent cancer cell invasion by suppressing H2O2-stimulated MMP-7 gene transcription via complex AP-1-dependent mechanisms, which required the upstream signal transduction from both the JNK/c-Jun and the ERK/c-Fos signaling pathways. These results indicated for the first time a potential chemopreventive role for DMA in other aspects of CRC metastasis.
Materials and methods
Experimental reagents
DMA (C22H37N4O8) was isolated from Monascus purpureus NTU 568-fermented rice. Fetal bovine serum (FBS) and TRIzol were purchased from Invitrogen (Carlsbad, CA, USA). Leibovitz's L-15 medium, 0.05% trypsin-EDTA, and 1× antibiotic-antimycotic were obtained from Hyclone (Logan, UT, USA). Phosphorylated JNK1/2, ERK1/2, and p38 MAPK and nonphosphorylated JNK1/2, ERK1/2, and p38 antibodies and anti-β-actin were purchased from Cell Signaling Technology, Inc. (Beverly, MA, USA). MMP-7, c-Fos, c-Jun, and the anti-mouse IgG horseradish peroxidase (HRP)-linked and anti-rabbit IgG HRP-linked secondary antibodies were obtained from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). MMP-7 neutralizing antibody (MAB3322) was purchased from Millipore (Chemicon, Temecula, CA, USA). The MAPK inhibitors SP600125 (JNK1/2 inhibitor), PD98059 (ERK1/2 inhibitor), and SB202190 (p38 inhibitor) were purchased from Calbiochem (San Diego, CA, USA). All other chemicals employed in this study were of analytical grade and were purchased from Sigma Chemical Co. (St. Louis, MO, USA).
Cell culture and viability assay
The human colorectal adenocarcinoma cell line SW620 was purchased from the Bioresource Collection and Research Center in Taiwan. Cells were cultured in Leibovitz's L-15 medium with 10% FBS and 1× antibiotic-antimycotic (HyClone), and incubated at 37°C in an atmosphere devoid of CO2. To measure cell survival after exposure to DMA and H2O2, MTT assays were carried out on these 2 groups. SW620 cells were incubated in 24-well plates in 1 mL of L-15 medium. Experimental details including concentrations and test incubation times are described in the text and the corresponding figure legend. Incubation was terminated by media aspiration and addition of 5 mg/mL of MTT working solution to each well. Formazane formation was terminated after 3 h by removal of the MTT solution. Subsequently, appropriate amounts of DMSO were added to each well to solubilize the formazane. The formazane-containing samples were transferred to a new 96-well plate and measured at 590 nm by an Opsys MR microplate spectrophotometer (Thermo LabSystems, Chantilly, VA, USA).
Matrigel invasion assay
Matrigel-coated transwell chambers with Millicell-HA filters (8-µm pore size; Millipore Co., Bedford, MA, USA) were used to assess the role of DMA and H2O2 on the invasive potential of SW620 cells. The frozen Matrigel was thawed overnight in a 4°C refrigerator and maintained on ice before use. The liquefied Matrigel was diluted in serum-free L-15 medium to a final concentration of 50 µg/mL, aliquoted into the inner chambers, and gelated at 37°C. The lower chambers contained medium with 10% FBS, and the upper chambers contained cells that were pretreated with DMA, H2O2, or a combination. Cells were incubated at 37°C for 24 h in the absence of CO2. Non-invading cells in the upper chamber were then wiped off with a cotton swab. Cells that had attached to the lower surface of the membrane were fixed with 4% formaldehyde, stained with 0.5% crystal violet, and counted microscopically. Microscopic fields were photographed with a digital camera and each photograph was measured using NIH ImageJ software (http://rsb.info.nih.gov/ij/). Three separate microscopic fields on the stained membranes from duplicate experiments were counted to determine the average number of cells/field (error bar = SD), and differences were considered significant at p < 0.05.
RNA isolation and reverse-transcription PCR analysis
Total RNA was extracted from cells using TRIzol (Invitrogen) according to the manufacturer's protocol. The concentrations of purified RNA were measured spectrophotometrically using a Picodrop (Picodrop Ltd., Walden, UK). The reverse transcription (RT) reaction was performed using a SuperScript III First-Strand Synthesis System (Invitrogen) according to the manufacturer's protocol. Amplification of the RT product by PCR was performed using Promega Taq DNA Polymerase (Promega Co., Madison, WI, USA). All reactions were performed in a thermal cycler (model 2400; Perkin-Elmer, Norwalk, CT, USA) with the following primers: MMP-7 sense, 5'- GGT CAC CTA CAG GAT CGT ATC ATA T-3', MMP-7 antisense, 5'-CAT CAC TGC ATT AGG ATC AGA GGA A-3', glyceraldehyde-3-phosphate dehydrogenase (GAPDH) sense, 5'-TGA TGA CAT CAA GAA GGT GGT GAA G-3', and GAPDH antisense, 5'-TCC TTG GAG GCC ATG TGG GCC AT-3'. The reaction sequence consisted of 1 cycle of initial denaturation at 94°C for 5 min, 30 cycles of denaturation at 94°C for 1 min, primer annealing at 62°C for 1 min, extension at 72°C for 1 min, and then 1 cycle of final extension at 72°C for 5 min. The PCR products of MMP-7 (373 bp) and GAPDH (240 bp) were run on 1.5% agarose gel, and then stained with ethidium bromide. The stained bands were visualized using UVP GDS-7900 digital imaging system (UVP AutoChemi System, Cambridge, UK).
Preparation of cell extracts and Western blot analysis
Cells were cultivated and treated as described in the text. The cells were washed twice with ice-cold PBS followed by the addition of 1 mL of radioimmunoprecipitation assay (RIPA) buffer (Sigma-Aldrich) with protease inhibitors. The cells were rapidly scraped off, and the crude lysates were transferred to microtubes and centrifuged at 15,000 × g for 30 min at 4°C. Cleared cell lysates containing equal amounts of proteins (50 µg) were denatured in sample buffer and separated by SDS-polyacrylamide gel electrophoresis (PAGE) and transferred to polyvinylidene difluoride membranes (PVDF; Pall Corporation, Ann Arbor, MI, USA). After blocking with 5% non-fat dry milk in PBS with 0.05% Tween 20 (PBST), the PVDF membranes were incubated overnight at 4°C with the chosen primary antibodies and then probed with the appropriate HRP-conjugated secondary antibodies to visualize specific bands using a Western Lightning Chemiluminescence Reagent kit (PerkinElmer, Waltham, MA, USA).
Casein zymography
Total proteins were loaded on precast 12% Novex zymogram blue casein gels (Invitrogen) to measure MMP-7 proteolytic activity. Following electrophoresis, the gels were renatured in Novex Zymogram Renaturing Buffer (Invitrogen) for 30 min at room temperature and then incubated at 37°C in Novex Zymogram Developing Buffer (Invitrogen) to allow degradation of the substrate in the gel matrix. Enzymatic activity was visualized as a clear band against a dark background.
Transient transfection and luciferase activity assay
Transient transfections were performed using Lipofectamine 2000 reagent (Invitrogen) according to the manufacturer's protocol. SW620 cells cultured in 6-well plates at a density of 5 × 105 cells/well were cotransfected with 1 µg of pAP1-luc and 0.05 µg of control pRL-TK reference vector (Promega). Cells were incubated with transfection reagent for 6 h and then changed with growth medium to recover for 24 h. After preincubation with SP600125, SB203580, PD98059, or DMA for 30 min, cells were treated with H2O2 in the presence or absence of MAPK inhibitors for 24 h. For the luciferase reporter assays, cells were harvested in 200 μL of Passive Lysis Buffer (PLB) and quantitation of relative light units was determined using a Dual Luciferase Stop & Glo reagent kit according the manufacturer's protocol (Promega).
Statistical analysis
Data are expressed as means ± standard deviation. Differences/correlations between groups were calculated using Student's t-test. Statistical significance was determined at p < 0.05.
Result
High dose concentration administration of H2O2 but not DMA inhibits SW620 cell viability
The Monascus secondary metabolite DMA (Fig. 1A) is a known antioxidant compound in Monascus-fermented products 21. However, the potential anticancer activity of DMA is completely unknown. In this study, we first evaluated the general cytotoxicity of DMA on SW620 colon cancer cells.
Effects of DMA or H2O2 on SW620 cell viability. A, chemical structure of DMA isolated from M. purpureus NTU 568. B, SW620 cells were treated with DMA (●) or H2O2 (○) at various doses for 24 h. The results represent dose-dependent effects of H2O2 but not DMA on SW620 cells. C, time-dependent decline of cell viability after H2O2 (10 μM) treatment. In each experiment, cell viability in vehicle-treated control was designated as 100%. Data represent the mean ± SD of reduction in cell viability of three independent experiments.
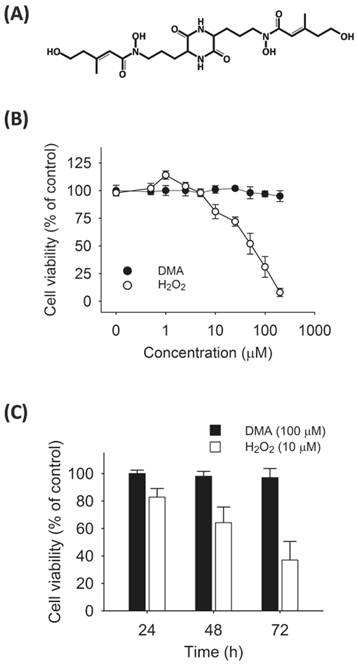
To examine DMA toxicity, SW620 cells were treated with DMA (10-200 μM) and incubated for 24 h. The percentage of viable cells was determined using an MTT assay and compared with control cells (vehicle). In our results, DMA concentration up to 200 μM did not influence cell viability. Nevertheless, high H2O2 accumulation (10-200 μM) resulted in poor viability in a dose-dependent manner (Fig. 1B). Cells were subsequently treated with 100 µM DMA or 10 µM H2O2 for the times indicated (24-72 h) to evaluate the duration effects. Time-dependent inhibition of SW620 cell viabilities were observed only under H2O2 treatment and not by DMA addition (Fig.1C).
DMA prevents H2O2-induced SW620 colon cancer cell invasion
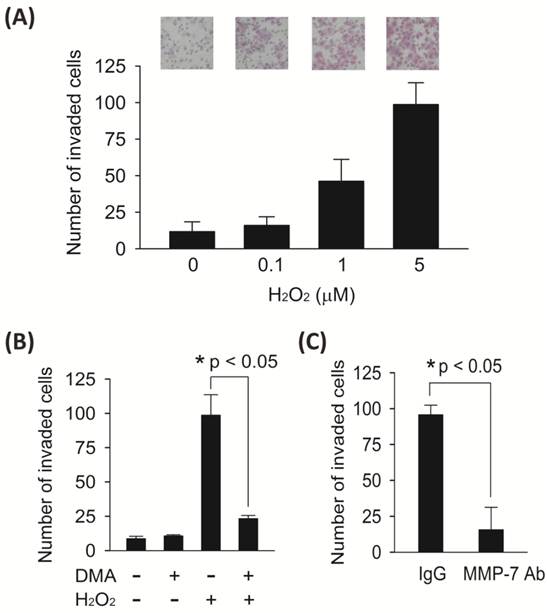
Because the preliminary result indicated that accumulation of 5 μM H2O2 had no lethal effect on SW620 cells, cell invasion assessment was done at 0.1, 1, and 5 μM of H2O2 treatment. The effect of H2O2 on the invasive activity of metastatic SW620 cells was measured using a Matrigel-coated Boyden chamber invasion assay. Representative images of filters containing invaded cells were taken with a microscope using a 10× objective lens and reveal that H2O2 dramatically increased invading cell numbers in a dose-dependent manner (Fig. 2A). To investigate whether H2O2-mediated cell invasion could be antagonized by DMA, SW620 cells were pretreated with DMA (100 μM) for 30 min and then incubated with non-toxic dose of H2O2 (5 μM) for 24 h. Results showed that 100 μM of DMA effectively disrupted Matrigel invasion in the presence of H2O2 (Fig. 2B). To confirm the role of MMP-7 in H2O2-modulated cell invasion, we further treated SW620 cells with MMP-7 neutralizing antibody (10 μg/mL) or control IgG. After 24 h, the invasive ability of SW620 cells was significantly decreased after MMP-7 neutralizing antibody treatment (Fig. 2C). These results indicate that DMA prevents H2O2-induced cell invasion and that MMP-7 activity may play an important role in the management of cancer invasion.
DMA suppresses H2O2-facilitated MMP-7 expression
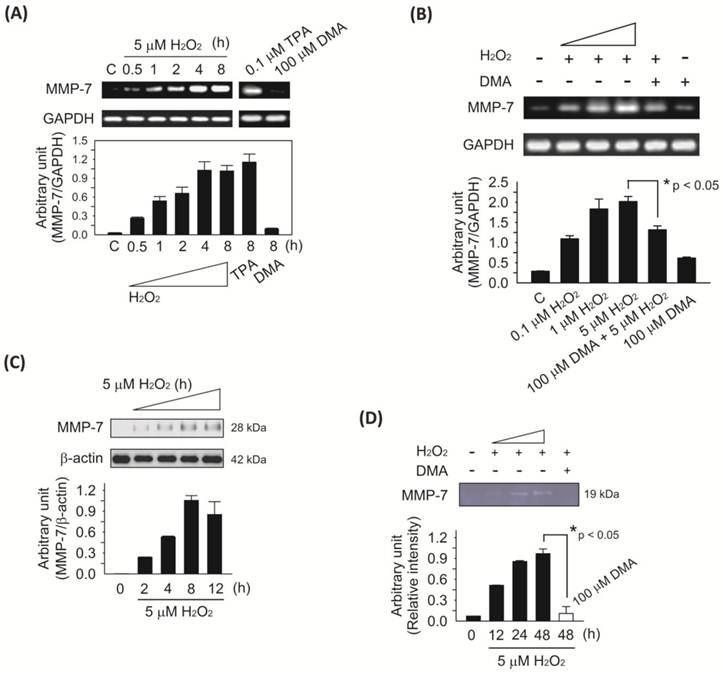
We investigated the role of MMP-7, which is believed to play an important part in the invasion process 22 and was found to be activated in a clinical study 11. Using semi-quantitative RT-PCR analysis, we found that the mRNA expression of MMP-7 in SW620 cells was up-regulated by H2O2 induction in a time-dependent increase and that it peaked at 4 h. MMP-7 mRNA transcription was not induced by DMA (100 μM), but by TPA (0.1 μM), in SW620 cells which were continuously incubated for 8 h (Fig. 3A). This finding suggests that DMA is unlikely to directly inhibit MMP-7 activity. Furthermore, SW620 cells were treated with H2O2 at the indicated doses for 4 h. MMP-7 mRNA expression increased in a dose-dependent manner after H2O2 treatment, but decreased after DMA pretreatment (Fig. 3B). Western blot analyses showed that H2O2 induced MMP-7 protein expression in a time-dependent manner (Fig. 3C). Finally, casein zymographic analysis showed that caseinolytic activities were upregulated by H2O2 (5 μM) treatment and suppressed by DMA pretreatment (Fig. 3D). All of the obtained bands were quantitated by densitometry using the ImageJ program (NIH). The data are representative of three separate experiments that yielded similar results.
Preventive effect of DMA on exogenous H2O2-stimulated invasion promotion of SW620 cells. A, visualization of dose-dependent increases SW620 cell invasion by H2O2 treatment. Equal numbers of cells were added to the upper wells of Matrigel-coated chambers in serum-free L-15 medium with various doses of H2O2. After incubation for 24 h, remaining cells in the upper chambers were removed by scrubbing with cotton swabs, and cells that invaded the lower chambers were fixed and stained with 0.5% crystal violet. B, DMA suppresses H2O2-induced Matrigel invasion of metastatic SW620 cancer cells. Cells were seeded atop chamber membranes in the presence or absence of 5 μM of H2O2 (- or + 100 μM DMA, respectively) with 10% FBS in the bottom well as the chemoattractant. After 24 h, the invaded cells were fixed, stained, and counted microscopically. The present data use of 100 μM DMA combination with H2O2 are significantly different (*p < 0.05, Student's t test) from date of H2O2-treatment alone. C, MMP-7 neutralizing antibody suppresses H2O2-induced Matrigel invasion of metastatic SW620 cancer cells. Cells were pretreated with H2O2 (5 μM), MMP-7 neutralizing antibody (10 μg/mL), or IgG and placed in the upper chamber coated with Matrigel. After 24-h culture, the invasive cells were fixed and counted. Each pretreatment was plated in triplicate in each experiment. *p < 0.05 was considered statistically significant.
Effect of H2O2 on MMP-7 activation and expression in SW620 cells. A, H2O2 induced MMP-7 mRNA expression in a time-dependent manner. RT-PCR analysis demonstrated that non-lethal H2O2 levels (5 μM) immediately induced MMP-7 mRNA expression that plateaued at 4 h. TPA was used as a positive control for DMA. B, H2O2-induced dose-dependent increase of MMP-7 mRNA expression. SW620 cells were treated with H2O2 at the indicated doses for 4 h. RT-PCR products of MMP-7 after H2O2 treatment increased in a dose-dependent manner. Cells were pretreated with DMA (100 μM) for 30 min followed by incubation for additional 4 h in the absence or presence of H2O2 (5 μM). The densitometry graph shows that DMA (100 μM) pretreatment induced a significant reduction in MMP-7 mRNA expression compared to that of H2O2 (5 μM) treatment only (*p < 0.05). C, Western blot analyses showed that H2O2 (5 μM) induced MMP-7 secretion in a time-dependent manner. Whole cell lysates were immunoblotted with β-actin antibody for the normalization of cell numbers. D, casein zymogram showing the presence of MMP-7 in SW620 cells after H2O2 (5 μM) treatment for the indicated time. Areas of casein substrate cleared by MMP-7 were seen in H2O2 (5 μM)-treated samples but declined in the presence of DMA (100 μM). The casein zymographic analysis plots were performed using lytic bands normalized for protein concentration. DMA pretreatment (open bar) in the presence of H2O2 (5 μM) for 48 h significantly suppressed the caseinolytic activity compared to H2O2 (5 μM) treatment only (*p < 0.05). All experiments from three independent experiments are plotted. Columns represent the mean ± SD.
Inhibitory effects of DMA on H2O2-induced MAPK activation
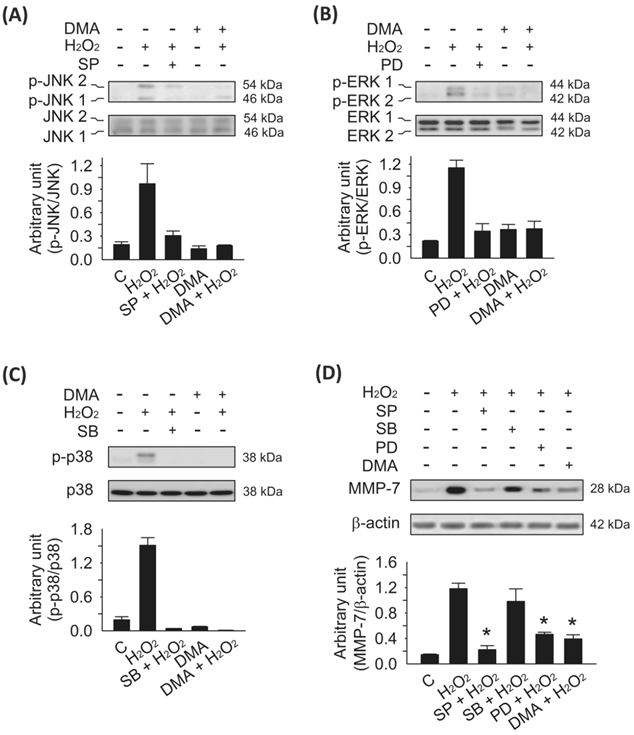
To address the question of what kind of signaling might be involved in MMP-7 induction by H2O2 and negative DMA regulation, we analyzed the effects of inhibiting protein kinases that have been shown to participate in free radical stress signaling 23. When the tumor microenvironment is insufficient for redox homeostasis, the ROS level, including H2O2 in tumors, becomes elevated compared to that in surrounding normal tissues. Cancer cells encountering high ROS levels are known to activate MAPK pathways such as ERK1/2, p38 MAPK, and JNK 6. We examined the role of DMA and the influence of these MAPK pathways on H2O2-mediated activation of MMP-7 activity. Using Western blotting analyses, we observed marked activation of p-JNK1/2 (Fig. 4A), p-ERK1/2 (Fig. 4B), and p-p38 MAPK (Fig. 4C) within 1 h after H2O2 (5 μM) treatment in SW620 cells. Interestingly, DMA treatment alone failed to affect JNK1/2, ERK1/2, or p38 MAPK phosphorylation, but DMA pretreatment could effectively suppress H2O2-induced MAPKs activation. In addition, MMP-7 protein was expressed soon after H2O2 stimulation, resulting in an 8-fold increase in MMP-7 protein levels over the control (Fig. 4D). Specific MAPK inhibitors were used to determine which MAPK pathway activation mediates H2O2-induced MMP-7 activities. Pretreatment with SP600125 (50 μM) or PD98059 (50 μM), the specific JNK1/2 and ERK1/2 inhibitors, respectively, significantly reduced MMP-7 protein expression to near basal control levels. In contrast, inhibition of H2O2-stimulated p38 MAPK activation by SB203580 (10 μM) only slightly decreased MMP-7 protein levels (no significant difference) (Fig. 4D). These results indicate a critical role of both JNK1/2 and ERK1/2, but not p38 MAPK, in H2O2-induced MMP-7 expression in SW620 cells.
DMA suppresses H2O2-induced MMP-7 expression by inhibiting AP-1 mediated gene expression via the JNK/c-Jun and ERK/c-Fos signaling pathway in SW620 cells
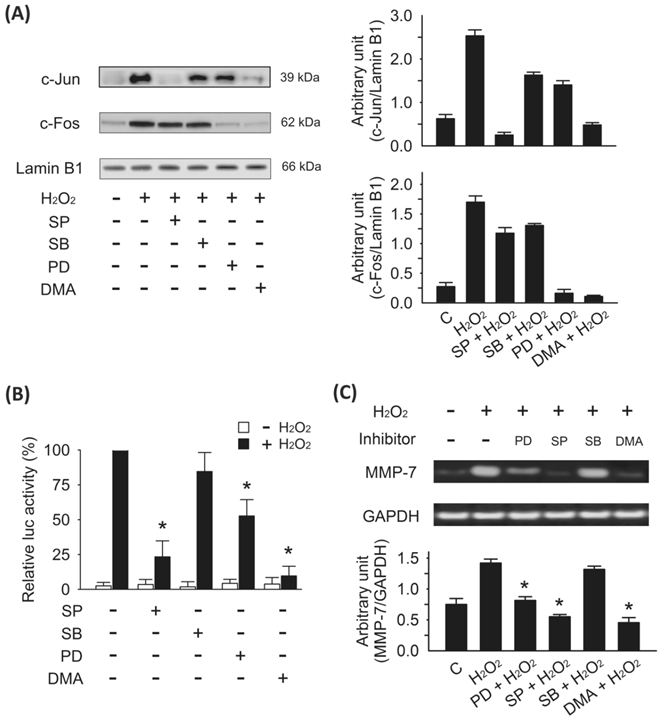
The results described above indicated that both JNK and ERK phosphorylation are potent stimulators of MMP-7 expression and cell invasion. In addition, the MMP-7 promoter has been shown to contain an AP-1 motif between positions -67 to -61 24 that is recognized by the transcription factors and is composed of members of the c-Jun and c-Fos families 25. To address the signaling cascades by which H2O2 stimulates MAPK activation, we next examined the effect of H2O2 on the transactivating function of c-Jun and c-Fos of the AP-1 family of transcription factors. In this study, H2O2 appeared to stimulate the translocation of both c-Jun and c-Fos, the major downstream components of MAPKs and was upregulated by JNK and ERK phosphorylation (Fig. 5A). To further explore the possible inhibitory mechanisms of DMA on H2O2-mediated MMP-7 transcriptional regulation, we examined the effects of DMA and MAPK inhibitors on AP-1 activation using the luciferase reporter gene assay. DMA, SP600125 (JNK1/2 inhibitor), or PD98059 (ERK1/2 inhibitor) interfered with the DNA binding of AP-1 accompanied with down regulation of JNK/c-Jun and ERK/c-Fos expression (Fig. 5B). To further clarify the involvement of JNK and ERK signaling in H2O2-induced MMP-7 expression, SW620 cells were cultured with H2O2 in the absence or presence of DMA (100 μM), SP600125 (50 μM), PD98059 (50 μM), or SB203580 (10 μM) respectively. DMA, SP600125 (JNK1/2 inhibitor), and PD98059 (ERK1/2 inhibitor) but not SB203580 (p38 MAPK inhibitor) significantly downregulated MMP-7 mRNA expression (Fig. 5C). These data indicate that stimulation of MMP-7 expression by H2O2 requires activation of JNK/c-Jun and ERK/c-Fos, but not p38 MAPK via the AP-1 signaling pathway. Based on these findings, we deduced that DMA suppressed H2O2-induced MMP-7 expression and cell invasion by inhibiting AP-1-mediated gene transcription via the JNK/c-Jun and ERK/c-Fos pathways (Fig. 6).
Discussion
Surgery of primary tumors provides the best prognosis for patients with CRC. Unfortunately, a significant portion of CRC patients will develop metastases, even after successful resection of the primary tumor. The effect of surgery on metastasis may be attributed to a number of risk factors, suggesting that surgical treatment induces inflammation associated with severe oxidative stress. ROS have contributed to various aspects of malignant tumors, including carcinogenesis, aberrant proliferation, angiogenesis, and metastasis, all of which serve as second messengers in the gene regulatory and signal transduction pathways within cells 23.
In relation to the stimulating effects of low H2O2 concentration, early studies have demonstrated that low-dose H2O2 induced cell proliferation possibly via various elevated factors 2, 4, 26-29. Here, we show that exposure of SW620 cells to lower levels of H2O2 (1 μM) actually stimulated statistically significant proliferation. Nevertheless, there were no significant differences among 2.5-5 μM H2O2 concentrations for 24-h treatment (Fig. 1B).
Analysis of the different MAPK pathways activated by H2O2 in SW620 cells. SW620 cells were cultured in serum-free L-15 medium for 24 h to synchronize the cells and to make them quiescent prior to treatment with SP600125 (50 μM; JNK1/2 phosphorylation inhibitor), PD98059 (50 μM; ERK1/2 phosphorylation inhibitor), or SB203580 (10 μM; p38 MAPK phosphorylation inhibitor) for 30 min prior to the addition of H2O2 (5 μM) for 1 h. Cell lysates were prepared and immunoblotted with the antibodies indicated in the panel. A, the levels of phospho-JNK1/2 were evaluated by H2O2 stimulation and suppressed by SP600125 or DMA (100 μM) pretreatment. B, the levels of phospho-ERK1/2 were evaluated by H2O2 stimulation and suppressed by PD98059 or DMA (100 μM) pretreatment. C, the level of phospho-p38 MAPK was evaluated by H2O2 stimulation and suppressed by SB203580 or DMA (100 μM) pretreatment. Ratios of bands with phosphospecific versus non-phosphospecific MAPK antibodies were determined, and quantitated by densitometry using the ImageJ program (NIH). Means ± SD are given. D, H2O2-enhanced MMP-7 protein expression was associated with increased JNK and ERK signaling in SW620 cells. Pretreatment with DMA (100 μM), SP600125 (50 μM), or PD98059 (50 μM) for 30 min prior to H2O2 (5 μM) induction for 8 h in SW620 cells significantly declined the expression of MMP-7 compared with H2O2-only treatment (*p < 0.05). However, the level of MMP-7 protein was only slightly decreased with no significant difference effect in the presence of SB203580 (10 μM) after H2O2-induced MAPK stimulation. The bars in the lower panel denote means ± S.D. of three experiments for each condition determined from densitometry relative to β-actin.
Regulation of MMP-7 transcription is dependent on JNK/c-Jun and ERK/c-Fos activities in H2O2-treated SW620 cells. A, effects of DMA and MAPK inhibitors on H2O2-induced c-Jun and c-Fos translocation. Nucleoprotein was isolated from SW620 cells treated with DMA (100 μM), SP600125 (50 μM), or PD98059 (50 μM) for 30 min prior to H2O2 (5 μM) induction for 4 h. Afterward, Western blotting analysis was performed using specific antibodies against c-Jun and c-Fos. Expression of lamin B1 was examined in parallel to confirm that equal amounts of nucleoprotein were being analyzed for each condition. The right upper panel showed that the JNK inhibitor SP600125 and DMA effectively inhibited H2O2-induced c-Jun translocation. The right lower panel showed that the ERK inhibitor PD98059 and DMA effectively inhibited H2O2-induced c-Fos translocation. B, DMA suppressed H2O2-induced AP-1 transcriptional activity. AP-1 activation was induced by H2O2 treatment via the JNK/cJun and ERK/c-Fos signaling pathways. SW620 cells were transiently transfected with AP-1 luciferase reporter (pAP1-luc) and pRL-TK reference vector for 24 h and were co-cultured with DMA or MAPK inhibitors in the absence (□) or presence (■) of H2O2 (5 μM). Relative luciferase activities were measured by calculating the light emitted and were normalized by coexpression of pRL-TK Renilla luciferase (ratio multiplied by an arbitrary factor to set the control of H2O2 induction only to 100). The relative luciferase activities are presented as means ± SD. C, H2O2-stimulated MMP-7 transcription depends on both JNK and ERK signaling activation. Pretreatment with DMA (100 μM), SP600125 (50 μM), or PD98059 (50 μM) for 30 min prior to H2O2 (5 μM) induction for 4 h in SW620 cells significantly reduced the expression of MMP-7 mRNA compared with H2O2-only treatment (*p < 0.05). The bars in the lower panel denote means ± SD of three experiments for each condition determined from densitometry relative to GAPDH mRNA.
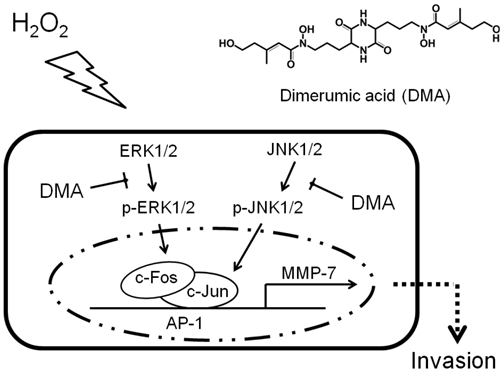
Schematic model of the proposed signal pathways inhibition of cancer cell invasion by DMA. Exogenous H2O2 triggers JNK1/2 and ERK1/2 activation, thereby inducing the expression of c-Jun and c-Fos, respectively. The c-Jun and c-Fos proteins cooperatively bind to the AP-1 binding site and promote MMP-7 gene transcription. DMA acts as an inhibitor of the JNK1/2 and ERK1/2 signaling pathways, suppressing H2O2-induced MAPK activation and MMP-7 expression.
Interestingly, DMA pretreatment effectively attenuated H2O2-induced cell invasion through Matrigel-coated Boyden chambers (Fig. 2). To examine whether MMP-7 was involved in response to exogenous H2O2-induced oxidative stress in SW620 cells, the expression profiles of MMP-7 mRNA were confirmed by RT-PCR. Although time-dependent elevation of MMP-7 mRNA following H2O2 administration had been observed, it could be effectively suppressed by DMA pretreatment (Figs. 3A & 3B). In clinical reports, MMP-7 expression has been considered one of the major mechanisms leading to the poor survival rate of CRC patients 9-10. Moreover, evidence is emerging that members of the MMP family including MMP-2, -7, -9, and -13 can serve not only as potential prognostic biomarkers, but also as indicators of tumor recurrence, metastatic spread, and response to primary and adjuvant therapy for CRC 30.
Expression of MMPs in tumors has been shown to be involved in activation of numerous MAPKs that are mediated in an autocrine or paracrine manner by growth factors, cytokines, or endogenous oxidative stress factors secreted by tumor-infiltrating inflammatory cells, stromal cells, or the tumor itself 31-32. Our results provide evidence that exogenous H2O2 exposure could remarkably contribute to increased phosphorylation of all 3 MAPKs (JNK, ERK, and p38 MAPK), but have failed to find during DMA intervention (Figs. 4A-C). Furthermore, DMA significantly inhibited H2O2-mediated MMP-7 expression that might be associated with attenuation of JNK and ERK signaling axes (Fig. 4D). In addition to the exogenous H2O2 in our results, induction of MMP-7 activation has been observed in fibroblast growth factor-1 (FGF-1) 33, hepatocyte growth factor/scatter factor (HGF/SF) 34, vascular endothelial growth factor (VEGF) and interlukin-8 (IL-8) 35. In addition, environmental insults such as ultraviolet B (UVB) radiation 36, cigarette smoke exposure 37, dietary components 38, and microorganism infection 39 contribute to the generation of free radicals and ROS that stimulate the inflammatory process and trigger MMP-7 expression.
Tumor cells, stromal cells, and infiltrating immune cells all contribute to benefit the tumor microenvironment via direct or indirect mediation. In the tumor milieu, infiltrating inflammatory cells receive ample activation signals and release abundant ROS to promote tumor progression. Among the ROS, H2O2 has a relatively long biological half-life and is able to diffuse across the cell membrane to modulate many downstream signaling molecules, such as transcription factors and protein kinases 23. It has been proposed that invasion-associated MMPs are regulated by AP-1 to regulate a program of gene expression that induces invasion in tumor cells 25. In our results, we attempted to clarify the potential molecular mechanisms responsible for the H2O2-induced activation of MMP-7. We suggested that H2O2-induced MMP-7 activation was AP-1-dependent through parallel signaling pathways that may involve the JNK/c-Jun and ERK/c-Fos signaling cascades (Fig. 5). Based on previous evidence of the radical scavenging activity of DMA 17, we described a hypothetical schematic model to demonstrate the potential role of DMA in H2O2-induced activation of MMP-7 and cancer invasion (Fig. 6). Further studies should be conducted in the future to demonstrate whether the effects of DMA on MMP-7 expression and cell invasion were mediated by its inhibitory role in the JNK and ERK signaling pathways. JNK/c-Jun and ERK/c-Fos activation should also be addressed to explain whether the AP-1 binding site of c-Jun and c-Fos in pAP1-luc is identical to that in the MMP-7 promoter.
Various adjuvant therapeutic strategies to prevent tumor recurrence after surgery have been explored and offer promising means of improving patients' outcome 40-43. Dietary chemoprevention has recently received attention for CRC prevention. Use of natural dietary compounds has received extensive thought for cancer prevention 44. Moreover, functional Monascus-fermented products containing several essential secondary metabolites with various biological activities have been described in our previous studies 45-50. In this study, we found that exogenous H2O2 can stimulate cell invasion activity and that DMA pretreatment can reverse the malignant phenotype of cancer cells. These results suggest that DMA plays not only a free radical scavenger but also an essential role in preventing cancer metastasis.
Abbreviations
AP-1: activator protein-1; CRC: colorectal cancer; DMA: dimerumic acid; ERK: extracellular-regulated kinase; JNK: Jun N-terminal kinase; MAPK: mitogen-activated protein kinase; MMP: matrix metalloproteinase; ROS: reactive oxygen species.
Acknowledgements
We would like to thank Drs. Hsu YW and Kuo YH, National Research Institute of Chinese Medicine, for providing DMA. This work was supported financially by the National Science Council, Taiwan, Republic of China (NSC 95-2313-B-002-096-MY2 and NSC 97-2313-B002-032-MY3) and the Technology Development Program for Academia (TDPA) of the Ministry of Economic Affairs (MOEA) of Taiwan, Republic of China (98-EC-17-A-17-S2-0136).
Conflict of Interests
The authors have declared that no conflict of interest exists.
References
1. Szatrowski TP, Nathan CF. Production of large amounts of hydrogen peroxide by human tumor cells. Cancer Res. 1991;51:794-8
2. Park IJ, Hwang JT, Kim YM, Ha J, Park OJ. Differential modulation of AMPK signaling pathways by low or high levels of exogenous reactive oxygen species in colon cancer cells. Ann N Y Acad Sci. 2006;1091:102-9
3. Polytarchou C, Hatziapostolou M, Papadimitriou E. Hydrogen peroxide stimulates proliferation and migration of human prostate cancer cells through activation of activator protein-1 and up-regulation of the heparin affin regulatory peptide gene. J Biol Chem. 2005;280:40428-35
4. Xia C, Meng Q, Liu LZ, Rojanasakul Y, Wang XR, Jiang BH. Reactive oxygen species regulate angiogenesis and tumor growth through vascular endothelial growth factor. Cancer Res. 2007;67:10823-30
5. Wu WS, Wu JR, Hu CT. Signal cross talks for sustained MAPK activation and cell migration: the potential role of reactive oxygen species. Cancer Metastasis Rev. 2008;27:303-14
6. Nishikawa M. Reactive oxygen species in tumor metastasis. Cancer Lett. 2008;266:53-9
7. Woessner JJr. Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J. 1991;5:2145-54
8. Masaki T, Matsuoka H, Sugiyama M, Abe N, Goto A, Sakamoto A, Atomi Y. Matrilysin (MMP-7) as a significant determinant of malignant potential of early invasive colorectal carcinomas. Br J Cancer. 2001;84:1317-21
9. Adachi Y, Yamamoto H, Itoh F, Hinoda Y, Okada Y, Imai K. Contribution of matrilysin (MMP-7) to the metastatic pathway of human colorectal cancers. Gut. 1999;45:252-8
10. Adachi Y, Yamamoto H, Itoh F, Arimura Y, Nishi M, Endo T, Imai K. Clinicopathologic and prognostic significance of matrilysin expression at the invasive front in human colorectal cancers. Int J Cancer. 2001;95:290-4
11. Zeng ZS, Shu WP, Cohen AM, Guillem JG. Matrix metalloproteinase-7 expression in colorectal cancer liver metastases: evidence for involvement of MMP-7 activation in human cancer metastases. Clin Cancer Res. 2002;8:144-8
12. Prise KM, O'Sullivan JM. Radiation-induced bystander signalling in cancer therapy. Nat Rev Cancer. 2009;9:351-60
13. Narayanan PK, Goodwin EH, Lehnert BE. α particles initiate biological production of superoxide anions and hydrogen peroxide in human cells. Cancer Res. 1997;57:3963-71
14. Hao Q, Rutherford SA, Low B, Tang H. Selective regulation of hydrogen peroxide signaling by receptor tyrosine phosphatase-α. Free Radic Biol Med. 2006;41:302-10
15. Brenneisen P, Briviba K, Wlaschek M, Wenk J, Scharffetter-Kochanek K. Hydrogen peroxide (H2O2) increases the steady-state mRNA levels of collagenase/MMP-1 in human dermal fibroblasts. Free Radic Biol Med. 1997;22:515-24
16. Kim M, Murakami A, Kawabata K, Ohigashi H. (-)-Epigallocatechin-3-gallate promotes pro-matrix metalloproteinase-7 production via activation of the JNK1/2 pathway in HT-29 human colorectal cancer cells. Carcinogenesis. 2005;26:1553-62
17. Aniya Y, Ohtani II, Higa T, Miyagi C, Gibo H, Shimabukuro M, Nakanishi H, Taira J. Dimerumic acid as an antioxidant of the mold, Monascus anka. Free Radic Biol Med. 2000;28:999-1004
18. Yamashiro JI, Shiraishi S, Fuwa T, Horie T. Dimerumic acid protected oxidative stress-induced cytotoxicity in isolated rat hepatocytes. Cell Biol Toxicol. 2008;24:283-90
19. Heber D, Yip I, Ashley JM, Elashoff DA, Elashoff RM, Go VL. Cholesterol-lowering effects of a proprietary Chinese red-yeast-rice dietary supplement. Am J Clin Nutr. 1999;69:231-6
20. Leibovitz A, Stinson JC, McCombs WB III, McCoy CE, Mazur KC, Mabry ND. Classification of human colorectal adenocarcinoma cell lines. Cancer Res. 1976;36:4562-9
21. Taira J, Miyagi C, Aniya Y. Dimerumic acid as an antioxidant from the mold, Monascus anka: the inhibition mechanisms against lipid peroxidation and hemeprotein-mediated oxidation. Biochem Pharmacol. 2002;63:1019-26
22. Yamamoto H, Itoh F, Hinod Y, Imai K. Suppression of matrilysin inhibits colon cancer cell invasion in vitro. Int J Cancer. 1995;61:218-22
23. Allen RG, Tresini M. Oxidative stress and gene regulation. Free Radic Biol Med. 2000;28:463-99
24. Gaire M, Magbanua Z, McDonnell S, McNeil L, Lovett DH, Matrisian LM. Structure and expression of the human gene for the matrix metalloproteinase matrilysin. J Biol Chem. 1994;269:2032-40
25. Ozanne BW, Spence HJ, McGarry LC, Hennigan RF. Transcription factors control invasion: AP-1 the first among equals. Oncogene. 2006;26:1-10
26. Sigaud S, Evelson P, Gonzalez-Flecha B. H2O2-induced proliferation of primary alveolar epithelial cells is mediated by MAP kinases. Antioxid Redox Signal. 2005;7:6-13
27. Kikuta K, Masamune A, Satoh M, Suzuki N, Satoh K, Shimosegawa T. Hydrogen peroxide activates activator protein-1 and mitogen-activated protein kinases in pancreatic stellate cells. Mol Cell Biochem. 2006;291:11-20
28. Lee SH, Na SI, Heo JS, Kim MH, Kim YH, Lee MY, Kim SH, Lee YJ, Han HJ. Arachidonic acid release by H2O2 mediated proliferation of mouse embryonic stem cells: involvement of Ca2+/PKC and MAPKs-induced EGFR transactivation. J Cell Biochem. 2009;106:787-97
29. Han Y, Huang C, Sun X, Xiang B, Wang M, Yeh ET, Chen Y, Li H, Shi G, Cang H, Sun Y, Wang J, Wang W, Gao F, Yi J. SENP3-mediated de-conjugation of SUMO2/3 from PML is correlated with accelerated cell proliferation under mild oxidative stress. J Biol Chem. 2010;285:12906-15
30. Roy R, Yang J, Moses MA. Matrix metalloproteinases as novel biomarkers and potential therapeutic targets in human cancer. J Clin Oncol. 2009;27:5287-97
31. Westermarck J, Kahari VM. Regulation of matrix metalloproteinase expression in tumor invasion. FASEB J. 1999;13:781-92
32. Chakraborti S, Mandal M, Das S, Mandal A, Chakraborti T. Regulation of matrix metalloproteinases: an overview. Mol Cell Biochem. 2003;253:269-85
33. Udayakumar TS, Stratton MS, Nagle RB, Bowden GT. Fibroblast growth factor-1 induced promatrilysin expression through the activation of extracellular-regulated kinases and STAT3. Neoplasia. 2002;4:60-7
34. Davies G, Jiang WG, Mason MD. Matrilysin mediates extracellular cleavage of E-cadherin from prostate cancer cells: a key mechanism in hepatocyte growth factor/scatter factor-induced cell-cell dissociation and in vitro invasion. Clin Cancer Res. 2001;7:3289-97
35. Wang FQ, So J, Reierstad S, Fishman DA. Matrilysin (MMP-7) promotes invasion of ovarian cancer cells by activation of progelatinase. Int J Cancer. 2005;114:19-31
36. Saarialho-Kere U, Kerkela E, Jeskanen L, Hasan T, Pierce R, Starcher B, Raudasoja R, Ranki A, Oikarinen A, Vaalamo M. Accumulation of matrilysin (MMP-7) and macrophage metalloelastase (MMP-12) in actinic damage. J Invest Dermatol. 1999;113:664-72
37. Shin VY, Jin HC, Ng EK, Sung JJ, Chu KM, Cho CH. Activation of 5-lipoxygenase is required for nicotine mediated epithelial-mesenchymal transition and tumor cell growth. Cancer Lett. 2010;292:237-45
38. Kim M, Murakami A, Ohigashi H. Modifying effects of dietary factors on (-)-epigallocatechin-3-gallate-induced pro-matrix metalloproteinase-7 production in HT-29 human colorectal cancer cells. Biosci Biotechnol Biochem. 2007;71:2442-50
39. Ogden SR, Noto JM, Allen SS, Patel DA, Romero-Gallo J, Washington MK, Fingleton B, Israel DA, Lewis ND, Wilson KT, Chaturvedi R, Zhao Z, Shyr Y, Peek RMJr. Matrix metalloproteinase-7 and premalignant host responses in Helicobacter pylori-infected mice. Cancer Res. 2010;70:30-5
40. Vermorken JB, Claessen AM, van Tinteren H, Gall HE, Ezinga R, Meijer S, Scheper RJ, Meijer CJ, Bloemena E, Ransom JH, Hanna MGJr, Pinedo HM. Active specific immunotherapy for stage II and stage III human colon cancer: a randomised trial. Lancet. 1999;353:345-50
41. Sadahiro S, Suzuki T, Ishikawa K, Yasuda S, Tajima T, Makuuchi H, Saitoh T, Murayama C. Prophylactic hepatic arterial infusion chemotherapy for the prevention of liver metastasis in patients with colon carcinoma: a randomized control trial. Cancer. 2004;100:590-7
42. Link KH, Kornmann M, Staib L, Redenbacher M, Kron M, Beger HG. Increase of survival benefit in advanced resectable colon cancer by extent of adjuvant treatment: results of a randomized trial comparing modulation of 5-FU + levamisole with folinic acid or with interferon-alpha. Ann Surg. 2005;242:178-87
43. Hoensch H, Groh B, Edler L, Kirch W. Prospective cohort comparison of flavonoid treatment in patients with resected colorectal cancer to prevent recurrence. World J Gastroenterol. 2008;14:2187-93
44. Pan MH, Lai CS, Wu JC, Ho CT. Molecular mechanisms for chemoprevention of colorectal cancer by natural dietary compounds. Mol Nutr Food Res. 2011;55:32-45
45. Lee CL, Tsai TY, Wang JJ, Pan TM. In vivo hypolipidemic effects and safety of low dosage Monascus powder in a hamster model of hyperlipidemia. Appl Microbiol Biotechnol. 2006;70:533-40
46. Lee CL, Hung HK, Wang JJ, Pan TM. Red mold dioscorea has greater hypolipidemic and antiatherosclerotic effect than traditional red mold rice and unfermented dioscorea in hamsters. J Agric Food Chem. 2007;55:7162-9
47. Lee CL, Kuo TF, Wang JJ, Pan TM. Red mold rice ameliorates impairment of memory and learning ability in intracerebroventricular amyloid beta-infused rat by repressing amyloid beta accumulation. J Neurosci Res. 2007;85:3171-82
48. Chen WP, Ho BY, Lee CL, Lee CH, Pan TM. Red mold rice prevents the development of obesity, dyslipidemia and hyperinsulinemia induced by high-fat diet. Int J Obes (Lond). 2008;32:1694-704
49. Lee CL, Wang JJ, Pan TM. Red mold rice extract represses amyloid beta peptide-induced neurotoxicity via potent synergism of anti-inflammatory and antioxidative effect. Appl Microbiol Biotechnol. 2008;79:829-41
50. Ho BY, Pan TM. The Monascus metabolite monacolin K reduces tumor progression and metastasis of Lewis lung carcinoma cells. J Agric Food Chem. 2009;57:8258-65
Author contact
![]() Corresponding author: Tzu-Ming Pan, PhD, Department of Biochemical Science and Technology, College of Life Science, National Taiwan University, Taipei, Taiwan, or King-Jen Chang, MD, PhD, Department of Surgery, National Taiwan University Hospital, No. 7, Chung-Shan S. Rd, Taipei, Taiwan. E-mail: tmpanedu.tw or kingjenedu.tw, Fax: +886-2-33663838.
Corresponding author: Tzu-Ming Pan, PhD, Department of Biochemical Science and Technology, College of Life Science, National Taiwan University, Taipei, Taiwan, or King-Jen Chang, MD, PhD, Department of Surgery, National Taiwan University Hospital, No. 7, Chung-Shan S. Rd, Taipei, Taiwan. E-mail: tmpanedu.tw or kingjenedu.tw, Fax: +886-2-33663838.

 Global reach, higher impact
Global reach, higher impact