10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2016; 12(11):1394-1404. doi:10.7150/ijbs.16517 This issue Cite
Research Paper
Conserved chemosensory proteins in the proboscis and eyes of Lepidoptera
1. State Key Laboratory for Biology of Plant Diseases and Insect Pests, Institute of Plant Protection, Chinese Academy of Agricultural Sciences, Beijing 100193, China.
2. State Key Laboratory of Integrated Management of Pest Insects and Rodents, Institute of Zoology, Chinese Academy of Sciences, Beijing 100101, P.R. China
3. Dipartimento di Biologia, Università degli Studi di Firenze, 50019 Sesto Fiorentino (Firenze), Italy.
*These Authors have contributed equally to this work.
Abstract
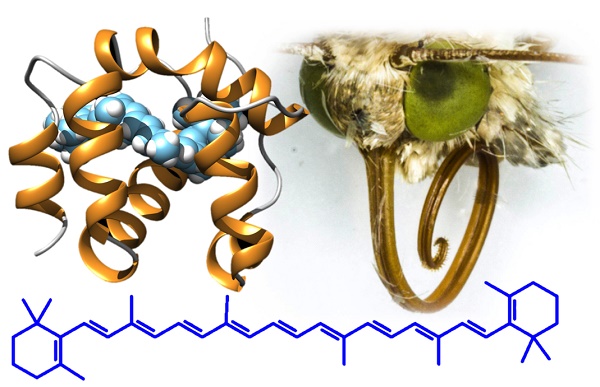
Odorant-binding proteins (OBPs) and chemosensory proteins (CSPs) are endowed with several different functions besides being carriers for pheromones and odorants. Based on a previous report of a CSP acting as surfactant in the proboscis of the moth Helicoverpa armigera, we revealed the presence of orthologue proteins in two other moths Plutella xylostella and Chilo suppressalis, as well as two butterflies Papilio machaon and Pieris rapae, using immunodetection and proteomic analysis. The unusual conservation of these proteins across large phylogenetic distances indicated a common specific function for these CSPs. This fact prompted us to search for other functions of these proteins and discovered that CSPs are abundantly expressed in the eyes of H. armigera and possibly involved as carriers for carotenoids and visual pigments. This hypothesis is supported by ligand-binding experiments and docking simulations with retinol and β-carotene. This last orange pigment, occurring in many fruits and vegetables, is an antioxidant and the precursor of visual pigments. We propose that structurally related CSPs solubilise nutritionally important carotenoids in the proboscis, while they act as carriers of both β-carotene and its derived products 3-hydroxyretinol and 3-hydroxyretinal in the eye. The use of soluble olfactory proteins, such as CSPs, as carriers for visual pigments in insects, here reported for the first time, parallels the function of retinol-binding protein in vertebrates, a lipocalin structurally related to vertebrate odorant-binding proteins.
Keywords: Odorant-binding protein, Chemosensory protein, Proboscis, Lepidoptera, Proteomics, Vision.

 Global reach, higher impact
Global reach, higher impact