ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2018; 14(5):518-530. doi:10.7150/ijbs.23511 This issue Cite
Research Paper
Elongator promotes the migration and invasion of hepatocellular carcinoma cell by the phosphorylation of AKT
1. Department of Cell Biology and Institute of Bioengineering, School of Medicine, Soochow University, Suzhou, 215123 China
2. State Key laboratory of Medical Neurobiology, Fudan University, Shanghai, 200032 China
3. Department of Gastroenterology, The North District of the Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou, Jiangsu, 215008 China.
*These authors contribute equally.
Abstract
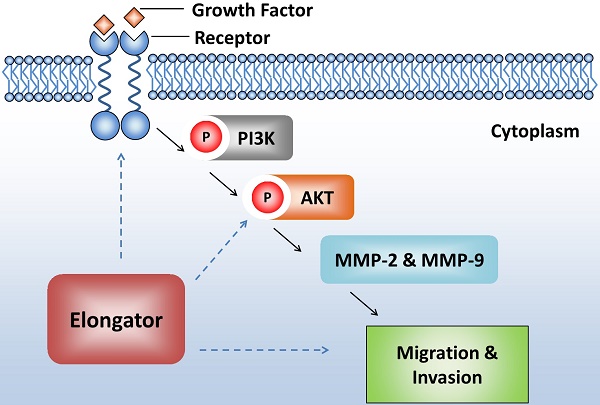
The Elongator is a complex with multiple subunits (Elp1-Elp6) which promotes transcript elongation and protein translation. In this study, we investigated the effects of Elongator on the migration and invasion of HCC cells as well as the underlying mechanisms. We showed that overexpression of Elp3 or Elp4 promoted the migration and invasion of HCC cells, which was abolished when either Elp3 or Elp4 was silenced. The expression of matrix metalloproteinase-2 (MMP-2) and MMP-9 were enhanced by phosphorylation of AKT. Elongator-driven migration and invasion and the expression of MMP-2 and MMP-9 were reduced in HCC cells treated with AKT inhibitor LY294002. Depletion of Elp3 also reduced the phosphorylation of AKT induced by growth factors. In vivo assay of lung metastasis in mice demonstrated that overexpression of Elp3 increased tumor nodules metastatic to lung. Importantly, Elp3 was up-regulated in human HCC tissues, which was correlated with the phosphorylation of AKT and expression of MMP-2. Collectively, these results suggested that Elongator activated migration and invasion of HCC cells by promoting the expression of MMP-2 and MMP-9 through the PI3K/AKT signaling pathway. Our work suggests that Elongator might be a potential marker which promotes the metastasis of HCC.
Keywords: Elp3, Elp4, hepatocellular carcinoma, migration, invasion, AKT
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignancy and the most frequent leading cause of cancer related mortality [1, 2]. High recurrence and high mortality were found in HCC patients with increasing incidence globally[3]. Early metastasis is recognized as the main cause for the low survival rates which is responsible for a poor chemotherapy and frequent relapse [4]. Intrahepatic and extrahepatic metastasis of HCC is very frequent and may still arise after remedy [5]. Currently, surgical resection and liver transplantation are main choices for the medical treatment of HCC. Radiofrequency ablation and chemoembolization are also applied for the HCC therapy [6, 7].
Elongator is a highly conserved multi-subunits complex composed of two subcomplexes, a core subcomplex (Elp1-Elp3) and an essential subcomplex (Elp4-Elp6) [8, 9]. Originally, Elongator was isolated as a component of a RNA polymerase II (RNAPII) holoenzyme from the yeast Saccharomyces cerevisiae [10]. Subsequently, human Elongator was purified from HeLa Cells [11]. Many reports provided evidences for a physiologically important role of Elongator complex in different cellular activities, including cytoskeleton organization, cellular motility, gene transcription, protein acetylation, tRNA modification, exocytosis and sensitivity to DNA damage agent [12-14]. Elongator is expressed both in nuclei and cytoplasm [12]. Depletion of Elongator reduced histone H3 acetylation, which resulted in lower RNAP II density through the coding region of target genes [15]. The function of Elongator in telomeric silencing and DNA repair was also described [16]. In cytoplasm, both Elp1 and Elp3 interact with microtubules. The migration and differentiation of projection neurons into the cerebral cortex were inhibited when either Elp1 or Elp3 was depleted during mouse corticogenesis[17].
PI3K/AKT signaling is activated by growth factors such hepatocyte growth factor (HGF) and epidermal growth factor (EGF), which regulates cell cycling, apoptosis, cell growth, and cell migration [18-21]. In response to EGF stimulation, several signaling cascades are initiated by epidermal growth factor receptor (EGFR), including MAPK/ERK, PI3K/AKT, and Stat pathways, which promotes cell survival, proliferation, migration, and invasion. HGF play critical roles in proliferation, cancer migration and metastasis [22]. Expression of HGF and HGF receptor, has been found to be correlated with human cancer in abnormal stages and poor prognosis [23].
Here, we showed that Elp3 and Elp4 promoted migration and invasion of HCC cells in vitro, and the tumor metastasis in mice. Further in vivo studies demonstrated that Elp3 was up-regulated in HCC tumor with enhanced expression of MMP-2 and MMP-9 through the PI3K/AKT signaling pathway.
Results
Elongator activates migration and invasion of HCC cells in vitro
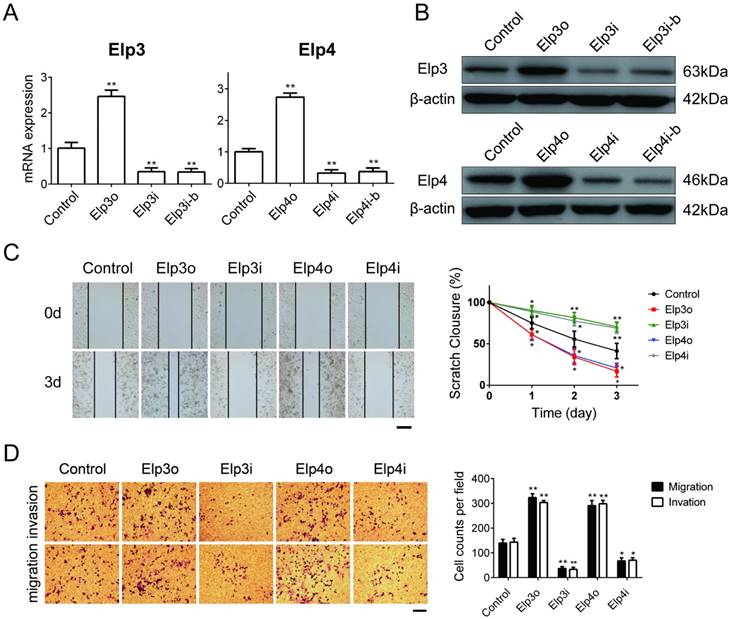
To explore the effects of Elongator on migration and invasion of HCC cells, we first established stable HepG2 cell lines that constantly overexpressed Elp3 (Elp3o) or Elp4 (Elp4o). We also generated HepG2 cell lines in which the expression of Elp3 or Elp4 were stably depleted by RNA interference (RNAi) using corresponding targeted shRNAs, as described in material and method. Elp3i and Elp3i-b were two different clones targeting the same sequence of Elp3 transcript. Elp4i and Elp4i-b were two different clones targeting the same sequence of Elp4 transcript. The mRNA and protein expression of Elp3 or Elp4 was efficiently raised in Elp3o or Elp4o cells, respectively, as determined by qRT-PCR (Figure 1A) and western blot (Figure 1B). The expression of Elp3 and Elp4 was significantly decreased in Elp3i/ Elp3i-b and Elp4i/ Elp4i-b cells (Figure 1A and 1B).
Then we characterized the effect of Elongator on motility of HCC cells. The wound-healing assay showed that Elp3o and Elp4o cells obtained quicker closure of the scratched wound compared with control HepG2 cells. Depletion of Elp3 (Elp3i) or Elp4 (Elp4i) significantly reduced the migratory capability of HepG2 cells (Figure 1C). Transwell assay was shown in Figure 1D. The number of cells that invaded or migrated to the lower chambers was remarkably increased for Elp3o or Elp4o cells. In contrast, reduction in cells invasion was observed for Elp3i and Elp4i cells. A rescue assay was shown in Supplementary Figure S2A. After the HepG2-Elp3i cells were transfected with Elp3o expression plasmid, the migration and invasion of cells were enhanced. These results suggested that Elongator promoted cell migration and invasion in HepG2 cells.
To further confirm the migration-promoting effect of Elp3 and Elp4, an alternative HCC cells of SMMC-7721, were transiently transfected with the Elp3o, Elp4o, Elp3i or Elp4i plasmids respectively. The overexpression of Elp3 or Elp4 resulted in a promotion of migratory and invasive capabilities of SMMC-7721. Depletion of Elp3 or Elp4 inhibited cell migration and invasion in wound-healing assay and transwell assay (Supplementary Figure S1). An additional HCC cell line Hep3B was also applied for the transwell assay (Supplementary Figure S2). Overexpression of Elp3 or Elp4 promoted the migration and invasion of Hep3B cells, while depletion of Elp3 or Elp4 reduced their migration and invasion. These results were consistent with the observation in HepG2 cells, which is consistent with the results of HepG2 in Figure 1.
Elongator activates PI3K/AKT/MMPs signaling pathway
AKT signaling pathway plays an important role in migration and invasion of cancer cells. It has been reported that MMP-2 and MMP-9 expressions are critically mediated by the PI3K/AKT pathway [24-26]. MMP-2 and MMP-9 have been shown to stimulate extracellular matrix (ECM) degradation, which is required for cell migration and invasion [27-31].
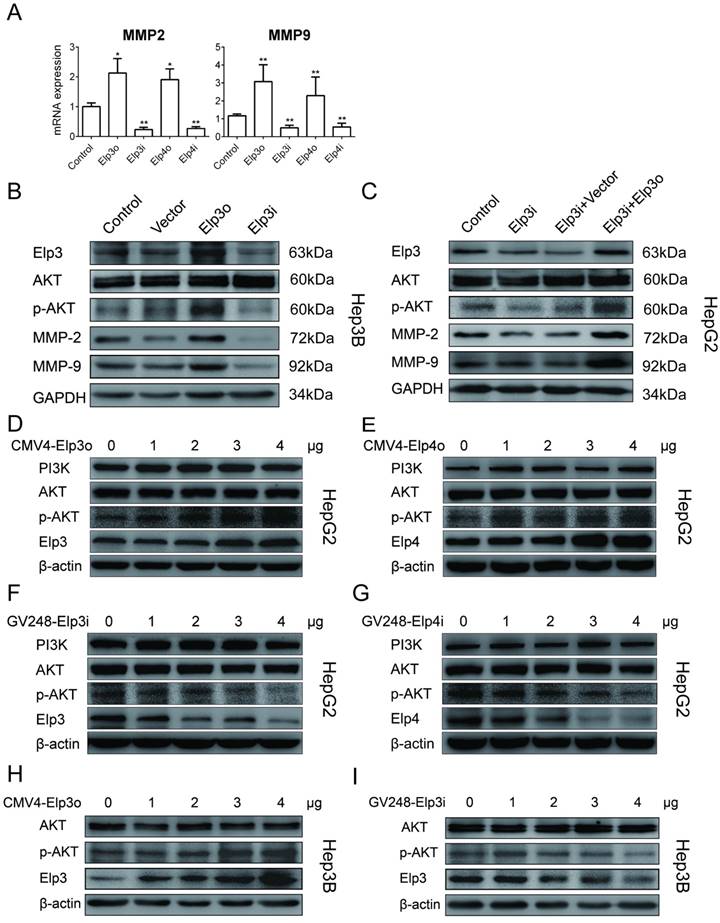
To further study the mechanisms underlying effects of Elongator on migration and invasion of HCC cells, we tested whether AKT activation was involved in Elongator function. As shown in Figure 2A, the mRNA expression of MMP-2 and MMP-9 were judged by qRT-PCR. The overexpression of Elp3 (Elp3o) or Elp4 (Elp4o) in HepG2 cells promoted the mRNA expression of MMP-2 and MMP-9. The depletion of Elp3 (Elp3i) or Elp4 (Elp4i) reduced the mRNA expression of MMP-2 and MMP-9.
Elp3 and Elp4 promote cell migration and invasion of HepG2 cells. The plasmids for Elp3 overexpression (Elp3o) and Elp4 overexpression (Elp4o), Elp3 interference (Elp3i) and Elp4 interference (Elp4i) were introduced into stably transfected HepG2 cell lines (A-D). (A) The mRNA expression of Elp3 or Elp4 in stably transfected HepG2 cell lines was examined by qRT-PCR as described in Materials and Methods. The relative mRNA levels of Elp3 or Elp4 was normalized against β-actin and depicted graphically. Elp3 and Elp4 mRNA levels in control HepG2 cells were set to 1 fold. (B) The protein expression of Elp3 and Elp4 in stably transfected HepG2 cells were examined by western blot analysis. A β-actin western blot is shown as loading control. (C) Wound-healing assay. Wound fields were observed directly after scribing (0 d), and cell migration was followed for 1, 2 and 3 days. Representative images taken at 0 and 3 days were shown up in the left panel. A quantification of the data obtained is illustrated on the right. For each experimental condition, the width of starching was set to 100% at time 0 and the width in other time points expressed relative to that. (D) Transwell assay. Cells on the lower surface of the chamber were stained by crystal violet after transfection. Representative images of migrated cells or invaded cells are shown in the left panel. The numbers of migration cells and invasion cells were counted. Data were calculated and presented as a histogram in the right panel. The results are expressed as the mean ± SD of three independent experiments. The figures show the data from a representative experiment performed in triplicates (*P< 0.05, **P< 0.01, independent Student t test).
Hep3B was transfected with Elp3o or Elp3i plasmids. As shown in Figure 2B, the phosphorylated AKT, MMP-2 and MMP-9 were significantly increased in Elp3o while reduced in Elp3i cells, which is consistent with results from HepG2 cells. A rescue assay was also performed, and shown in Figure 2C. Interestingly, when the Elp3i cells was transfected with Elp3o expression vector, the decreased phosphorylated AKT, MMP-2 and MMP-9 was significantly increased, compared with the control cells transfected with vector alone. The densitometric data was shown in Supplementary Figure S3A-3B.
To further confirm the effect of Elongator on AKT phosphorylation, a dose-dependent assay was carried out. HepG2 cells were transfected with increasing amount of plasmids of Elp3o (Figure 2D), Elp4o (Figure 2E), Elp3i (Figure 2F) and Elp4i (Figure 2G), respectively. The phosphorylated AKT increased when cells were transfected with increasing amount of Elp3o, or Elp4o. Inversely, when cells were transfected with increasing amount of Elp3i, or Elp4i, the phosphorylated AKT decreased. The densitometric data was shown in Supplementary Figure S3C-3F. Moreover, Hep3B cells were also transfected with increasing amount of plasmids of Elp3o (Figure 2H) and Elp3i (Figure 2I). The densitometric data was shown in Supplementary Figure S3G and S3H. These data indicate that the Elongator subunits of Elp3 and Elp4 enhanced AKT phosphorylation in HepG2 cells in a dose-dependent manner.
Elongator activates PI3K/AKT/MMPs signaling pathway. Stably transfected cells were maintained in the absence of serum for 24 h, and then, total RNA or proteins were isolated. Overexpressed Elp3 and Elp4 in HepG2 cells were abbreviated to Elp3o and Elp4o. Elp3i and Elp3i-b were two different clones targeting the same sequence of Elp3 transcript. Elp4i and Elp4i-b were also two different clones targeting the same sequence of Elp4 transcript. (A) The mRNA expression of MMP-2 and MMP-9 in stably transfected HepG2 cell lines was examined by qRT-PCR. Related mRNA levels of MMP-2 and MMP-9 were compared between control HepG2 cells and Elp3o or Elp4o cells, and between control HepG2 cells and Elp3i or Elp4i cells. MMP-2 and MMP-9 mRNA levels in control HepG2 cells were set to 1 fold. (B) The plasmids for Elp3 overexpression (Elp3o), Elp3 interference (Elp3i), or vector alone (Vector) were transfected into Hep3B cells. The protein expression of Elp3 and Elp4 in transiently transfected Hep3B cells were examined by western blot analysis. GAPDH was shown as a loading control. (C) Rescue Experiment. The plasmids for Elp3 overexpression (Elp3o) were transiently transfected into HepG2-Elp3i cells. Cells were subjected to western blot analysis with antibodies against AKT, phosphorylated AKT, MMP-2 and MMP-9. GAPDH was shown as a loading control. (D-G) HepG2 cells were transfected with increasing amount of CMV4-Elp3o (D), CMV4-Elp4o (E), GV248-Elp3i (F), or GV248-Elp4i (G). (H-I) Hep3B cells were transfected with increasing amount of CMV4-Elp3o (H) or GV248-Elp3i (I). Western blot analysis was performed with anti-PI3K, anti-AKT, anti-p-AKT, anti-Elp3 and anti-Elp4 antibodies. A β-actin western blot is shown as loading control. Results presented as mean ± SD (*P< 0.05, **P< 0.01, independent Student t test).
PI3K/AKT signaling pathway is required for the Elongator-mediated migration and invasion of HCC cells
To further confirm the promotion effect of Elongator on the migration and invasion of HCC cells by the activation of the PI3K/AKT signaling pathway, the phosphorylated AKT inhibitor LY294002 was applied which specifically blocks PI3K/AKT pathway.
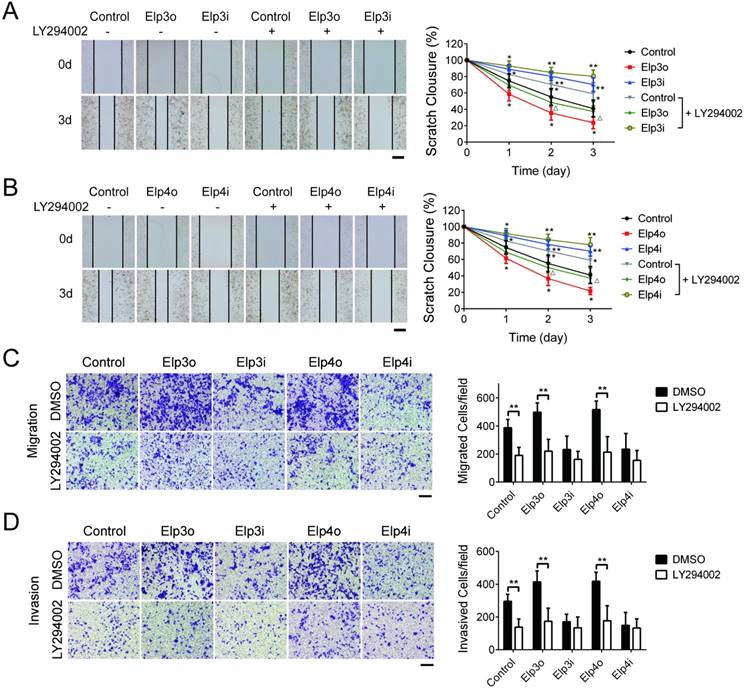
Stably transfected HCC cells overexpressing Elp3 (Elp3o) and Elp4 (Elp4o) or depleted of Elp3 (Elp3i) or Elp4 (Elp4i) were treated with 25 μM LY294002 or DMSO for 2 hours and then subjected to wound-healing assay. The quantification of wound closure was performed, shown in Figure 3A and 3B. When the HepG2cells were treated with LY294002, the migration of HepG2 cells promoted by Elp3 or Elp4 reduced significantly. Transwell assay was also performed, shown in Figure 3C and 3D. The migratory abilities were reduced prominently in Elp3o or Elp4o with LY294002 treatment. To exclude the possible effect of AKT inhibitor LY294002 on the growth of HCC cells, cell viability assay was performed in Supplementary Figure S4. The results demonstrated that the growth of HepG2 cells was not reduced upon the addition of LY294002. These results indicated that the Elongator-mediated migration of HepG2 cells was through the activation of PI3K/AKT pathway, although other factors and pathway may also be involved.
To further investigate the effect of Elongator in the activation of AKT pathway, we examined if the expression of Elp3 or Elp4 was inhibited by LY294002. The mRNA and protein expression of Elp3 or Elp4 was examined as shown in Figure 3E and 3F respectively. Both mRNA and protein expression were not altered by LY294002 in Elp3o, Elp4o, Elp3i or Elp4i cells. Blocking the PI3K/AKT pathway did not regulate Elp3 and Elp4 in either transcriptional or translational level.
PI3K/AKT signaling pathway is required for the Elongator-mediated migration and invasion of HCC cells. The plasmids for Elp3 overexpression (Elp3o) and Elp4 overexpression (Elp4o), Elp3 interference (Elp3i) and Elp4 interference (Elp4i) were introduced into stably transfected HepG2 cell lines. Stably transfected cells were maintained with the inhibitor LY294002 (25 μM for 2 hours) when indicated. (A-B) Wound-healing assay. Cells were maintained in the absence of serum and allowed to migrate. Wound fields were observed directly after scribing (0 d), and cell migration was followed for 1, 2 and 3 days. Pictures were taken from indicated time phase during 3 days of migration. Representative images were shown in left. Diagrams in right showed the percentage of wound closure at 0, 1, 2 and 3 days. For each experimental condition, the width of starching was set to 100% at time 0 and the width in other time phase expressed relative to that. (C-D) Transwell assay. Migration through membrane (C) and invasion through matrigel (D) were carried out with FBS (10%) as achemoattractant in transwell assay. Cells on the lower surface of the chamber were stained by crystal violet after transfection. Representative images of migrating cells or invading cells after staining were taken either in the absence or presence of the inhibitor LY294002. Histograms show the number of migrating cells and invading cells in right. (E) Total RNA was extracted and qRT-PCR was performed with primer pairs to amplify Elp3 and Elp4. The relative mRNA levels of Elp3 or Elp4 were examined. Elp3 and Elp4 mRNA levels in control HepG2 cells were set to 1 fold. (F) Cell lysates were extracted and subjected to western blot analysis with antibodies as indicated. The protein expressions of Elp3 and Elp4 were compared between inhibitor treatment and untreatment. (G-H) Total cell lysates of cells were probed for the expression of Elp3 (G) or Elp4 (H) levels normalized with β-actin by western blot analysis. Western blot was performed with specific antibodies against PI3K, AKT, phosphorylated AKT, MMP-2 and MMP-9.The densitometric analysis for the western blot was shown at the bottom. (I) The mRNA expression of MMP-2 and MMP-9 were compared between before and after inhibitor treatment in each group of cells respectively. The graphs represent densitometric results from three independent experiments. Each value represents the mean ± SD (*P< 0.05, **P< 0.01, independent Student t test).
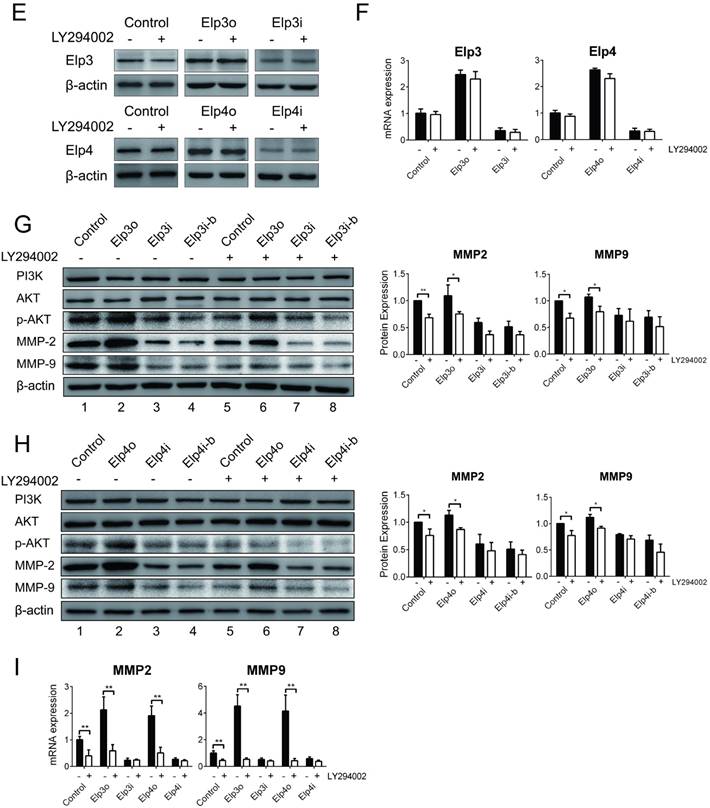
We then examined the effect of LY294002 on the phosphorylation of AKT. As shown in Figure 3G (Elp3) and Figure 3H (Elp4), the expression of PI3K remain constant before and after the treatment of LY294002. The phosphorylated AKT was significantly decreased in cells of Elp3o or Elp4o after treatment with LY294002 (compare lane 2 and 6). There is little effect of LY294002 on the phosphorylation of AKT in cells of Elp3i (compare lane 3-4 with lane 7-8) or Elp4i (compare lane 3-4 with lane 7-8). These results indicate Elongator is required for the phosphorylation of AKT.
Then we examined the expression of MMP-2 and MMP-9, which are downstream effectors of AKT phosphorylation. The expression of both MMP-2 and MMP-9 were reduced by LY294002 in cells of Elp3o (Figure 3G, compare lane 2 with lane 6) or Elp4o (Figure 3H, compare lane 2 with lane 6). However, there is a little effect of LY294002 on the expression of both MMP-2 and MMP-9 in cells of Elp3i (Figure 3G, compare lane 3-4 with lane 7-8) or Elp4i (Figure 3H, compare lane 3-4 with lane 7-8). The expression pattern of both MMP-2 and MMP-9 corresponded to the phosphorylated AKT. Elp3o and Elp4o cells showed similar reactions to LY294002 treatment. So did Elp3i and Elp4i. It seems that Elp3 and Elp4 worked coordinately in the intact Elongator complex.
To confirm the effect of LY294002 on the expression of MMP-2 and MMP-9, qRT-PCR was also performed. As shown in Figure 3I, the mRNA expression of both MMP-2 and MMP-9 was reduced significantly in Elp3o cells or Elp4 cells. However, there is little effect of LY294002 on the expression of MMP-2 and MMP-9 in Elp3i or Elp4i. Similar tendency of expression of MMP-2 and MMP-9 was observed over p-AKT. These results suggested that Elongator was required for the expression of MMPs activated by phosphorylated AKT.
Elongator is required for the phosphorylation of AKT induced by growth factor. (A) Cells were serum starved overnight and then were stimulated with 50 ng/ml EGF for 10 min. The expressions of phosphorylated AKT, AKT and Elp3 in stably transfected HepG2 cells were analyzed by Western blot. A β-actin western blot was shown as loading control. (B) Cells were maintained in the absence of serum overnight, and then were stimulated with 20 ng/ml HGF for 10 min. The protein expression of AKT and phosphorylated AKT in stably transfected HepG2 cells were examined by western blot analysis. (C) Rescue Experiment. The plasmids for Elp3 overexpression (Elp3o) were transiently transfected into HepG2-Elp3i cells. Cells were subjected to western blot analysis with antibodies against AKT, phosphorylated AKT and Elp3. β-actin was shown as a loading control. (D) Rescue Experiment. The plasmids for Elp3 overexpression (Elp3o) were transfected into HepG2-Elp3i cells. The protein expressions of phosphorylated AKT, AKT and Elp3 in transiently transfected HepG2-Elp3i cells were examined by western blot analysis. β-actin was shown as a loading control. (E) HepG2-Elp3i cells were transfected with increasing amount of CMV4-Elp3o. Western blot analysis was performed with anti-AKT, anti-p-AKT and anti-Elp3 antibodies. A β-actin western blot was shown as loading control.
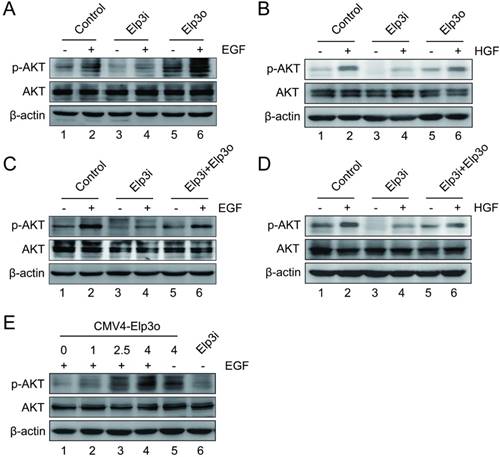
Elongator is required for the phosphorylation of AKT induced by growth factor
PI3K/AKT signaling is activated by growth factors such as hepatocyte growth factor (HGF) and epidermal growth factor (EGF). These growth factors have been reported to regulate several cellular biological functions such as cell cycling, apoptosis, cell growth, and cell migration [18, 21, 22]. To further characterize the functional implication of the Elongator-induced AKT phosphorylation, we examined the role of Elp3 in the phosphorylation of AKT when cells were activated by growth factors. Cells were exposed to the growth factors. The total AKT and phosphorylated AKT were then assessed by immunoblotting analysis.
As shown in Figure 4A, little difference was observed on the phosphorylation of AKT in HepG2-Elp3i cells with or without EGF treatment (compare lane 3 and lane 4). Both HepG2 and HepG2-Elp3o cells showed much higher level of phosphorylated AKT than HepG2-Elp3i cells did (compare lane 2, lane 4 and lane 6). The densitometric analysis was shown in Supplementary Figure S5A. The phosphorylation of AKT was increased by the overexpression of Elp3 (Figure 4A, lane 5), which was further enhanced by the presence of EGF (Figure 4A, lane 6). These results suggest that Elp3 is required for the phosphorylation of AKT activated by EGF. The similar observation was observed and confirmed in cells treated with HGF, as shown in Figure 4B.
A rescue assay was performed as shown in Figure 4C. The vector for Elp3 expression was introduced into HepG2-Elp3i cells. After treatment with EGF, the phosphorylation of AKT was significantly increased, compared with Elp3i cells transfected with empty vector. And EGF effectively activated the phosphorylation of AKT when the expression of Elp3 was restored in Elp3i cells (compare lane 4 and lane 6). The similar observation was observed and confirmed in cells treated with HGF, as shown in Figure 4D. A dose-dependent assay was also performed, as shown in Figure 4E. The phosphorylation of AKT was increased by the expression of Elp3 in a dose-dependent manner upon EGF treatment. The densitometric data was shown in Supplementary Figure S5. These results further demonstrated that Elongator is required for the phosphorylation of AKT induced by growth factors.
Elongator enhances the metastasis of HCC to lung
The novel finding that Elongator promoted the migration and invasion of HCC cells in vitro prompted us to examine whether Elongator enhances the metastasis of HCC cells in vivo. Mouse tumor metastasis by tail vein injection was performed to investigate the effect of Elp3 on metastasis. Mice were injected with Elp3o cells, or Elp3i cells into the tail vein. The HepG2 cells were transfected with empty vector (Vector), HepG2 cells alone (Control). PBS (NC) was also administrated as the controls. After injection for 8 weeks, mice were sacrificed and the metastatic nodules were visible at lung surfaces (Figure 5A, up panel). The lung tissues were then examined by hematoxylin and eosin (H&E) staining (Figure 5A low panel) to confirm that the nodules on the surfaces of mice lungs were metastatic tumors. Statistical chart on the number of metastatic tumors is shown in Figure 5B.
Elongator enhances the metastasis of HCC to lung. (A) 1×106 tumor cells suspended in 200 μl of normal saline were inoculated into the tail-vein of six weeks old female SCID beige mice. The normal saline without any cells was set as negative control (NC). HepG2 cells (HepG2) and HepG2 transfected with empty vector (Vector) were set as additional controls. Overexpressed Elp3 and interference Elp3 cell lines were respectively abbreviated to Elp3o and Elp3i. Lung tissues were dissected at 8th week after inoculation. Photographs of lung tissue (upper) and representative images of metastatic tumor via H&E staining (lower) were taken. (B) Tumor metastatic nodules of different cell groups were calculated and compared after 8 weeks. Results presented as mean ± SD (*P< 0.05, **P< 0.01, independent Student t test). (C) Protein expression was detected by IHC. Representative images of IHC using anti-Elp3, anti-Elp4, anti-MMP-2, anti-MMP-9 and anti-p-AKT, are shown.
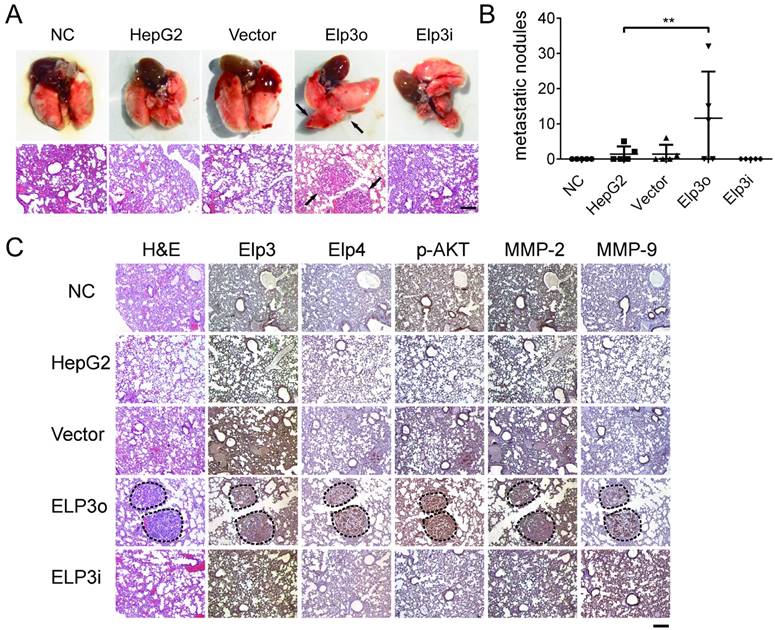
Histological examination showed that the number of lung metastatic tumors in mice with Elp3o cells was significantly higher than the tumor number in mice with HepG2 cells. Little metastatic tumor was found in mice injected with Elp3i cells. As HepG2 cells are of low metastasis [32, 33], the metastasis of HepG2 cells was much more sensitive to Elp3 overexpression, rather than Elp3 depletion.
The findings that Elongator enhances the metastasis of HCC in vivo, suggests Elongator as a potential target for HCC treatment. Immunohistochemical staining was also performed with the lung tissues, demonstrating the increased expression of p-AKT, MMP-2 and MMP-9 in the metastatic tumors from Elp3o mice (Figure 5C), which was consistent with in vitro assay.
Elongator is up-regulated in HCC tissues
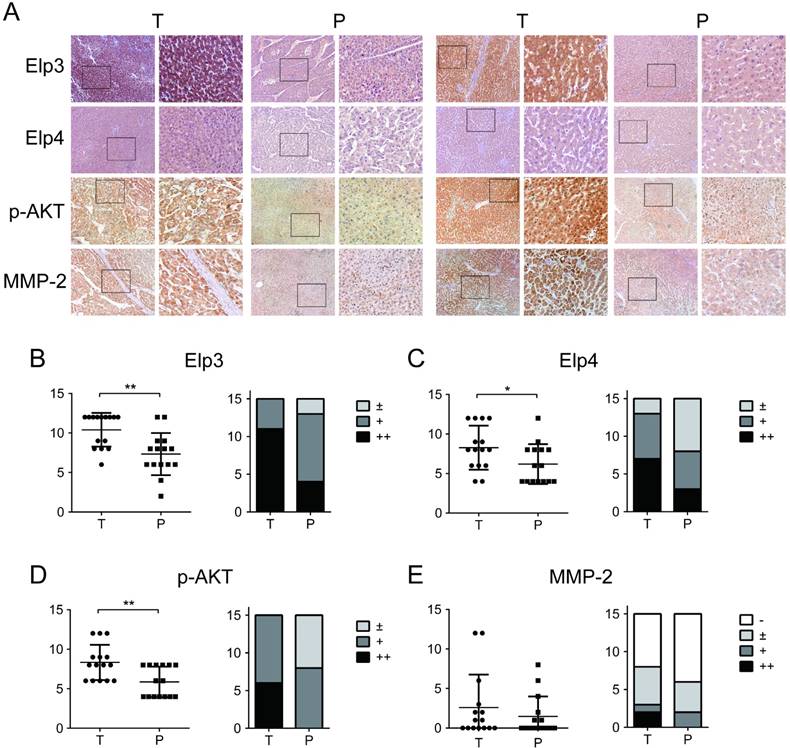
To further investigate the clinical significance of Elp3 in the metastasis of HCC, we examined the expression of Elp3, Elp4, p-AKT and MMP-2 in 15 paired HCC patients by immunohistochemical (IHC) analysis. IHC staining of Elp3, Elp4,p-AKT and MMP-2 in tumor tissues (T) and adjacent para-tumor tissues (P) were shown in Figure 6A. Statistical analysis demonstrated that the expression of both Elp3 and Elp4 in tumor tissues were stronger than that in corresponding para-tumor tissues, as shown in Figure 6B-C. The positive expression of Elp3 was detected in 15/15 (100 %) of tumor tissues and average scoring was 10.40±2.13. For para-tumor tissues, the positive expression rate of Elp3 was 13/15 (86.67%) and average scoring was 7.33±2.66.
The phosphorylation of AKT was also up-regulated in HCC tissues compared with para-tumor tissues (Figure 6D), which was highly consistent with the enhanced expression of Elp3 in these tissues. The positive expression rate of p-AKT was 15/15 (100 %) of tumor tissues and average scoring was 8.33±2.23. The positive expression rate of p-AKT was 8/15 (53.33%) and average scoring was 5.87±1.92 in adjacent para-tumor tissues. The positive expression rate and positive staining score of Elp3, Elp4 and p-AKT in HCC tissues were significantly increased compared to the para-tumor tissues (P <0.05). These results were consistent with our observation in vitro.
However, the high expression of MMP-2 was only detected in 20 % (3 of 15) HCC cases and the remaining 80 % (12 of 15) were scored as no or low MMP-2 expression. There was no significant difference in MMP-2 positive expression rate and positive staining score between HCC tissues and para-tumor tissues (P >0.05) (Figure 6E). Actually no metastatic tumor was found in 14/15 of HCC, which may explain no or low expression of MMP-2 in these tumors.
Discussion
Metastasis of cancer is a multistep and complicated process. The interactions of cancer cells with extracellular matrix (ECM) is required for metastasis [34]. The matrix metalloproteinase (MMPs), a family of zinc-containing endopeptidases, is necessary for photolytic degradation of the ECM. Among all MMPs, MMP-2 and MMP-9 are known as key enzymes in the degradation of type IV collagen. The unnatural expression of MMPs is thought to be a crucial determinant factor in cell migration [35, 36]. The expression of MMP-2 and MMP-9 were mediated by the PI3K/AKT pathway. Activation of AKT is required for cancer invasion and metastasis by stimulating the secretion of MMPs [37-39].
In this work, we demonstrated that Elp3 and Elp4 enhance migratory and invasive capability of HCC cells by up-regulating MMP-2 and MMP-9. Silencing of Elp3 and Elp4 down-regulate the expression of MMP-2 and MMP-9 and reduce the migration and invasion of HCC cells. Importantly, Elp3 and Elp4 activate the phosphorylation of AKT in HCC cells in a dose-dependent manner. Blocking of phosphorylation of AKT by the inhibitor LY294002 significantly suppressed Elongator-mediated cell migration and invasion. The phosphorylation of AKT stimulated by both Elp3 and Elp4 was significantly reduced by LY294002 (Figure 3), apparently through PI3K dependent pathway. However, the inhibition was not complete and certain amount of phosphorylated AKT still remained, especially in Elp3o cells, which possibly resulted from PI3K-independent pathway [40]. Taken together, our results showed that Elp3 and Elp4 promoted migration and invasion of HCC cells through AKT/MMPs signaling pathway.
PI3K/AKT signaling pathway could be activated by various growth factors, such as EGF and HGF. Previous studies reported that EGF and HGF could stimulate cell motility and migration by PI3K/AKT signaling pathway, and play important roles in proliferative ability and invasive property of HCC cell lines [41, 42]. Additionally, the activation of EGF/EGFR or HGF/c-MET signaling pathway is associated with the development of HCC and may count for the poor prognosis of the patients [43-45]. Several drugs and protein cofactors described in these studies could promote or inhibit the inducement of cell migration and activation of PI3K/AKT signaling pathway by interacting with the receptor of EGF or HGF [45, 46]. In our studies, the fact that depletion of Elp3 impaired the activation of AKT induced by EGF/HGF, suggested that Elp3 is involved in the activation of AKT phosphorylation in EGF/HGF-induced AKT signaling pathway.
We also confirm Elp3 promotes invasion and migration through AKT signaling activation in vivo. Tumor metastasis developed in mice injected with HCC cells which overexpressed Elp3, rather than the cells depleted of Elp3. The IHC from mice pulmonary cross section were consistent with in vitro assays. Human HCC cells of Elp3o promoted migration and invasion in vitro, and its capacity to develop tumor metastasis in mice as well. The results demonstrate that metastasis of HepG2 cells are much more sensitive to Elp3 overexpression, rather than Elp3 depletion, possibly because HepG2 cells is of low metastasis [32, 33].
Furthermore, we also showed that Elp3 was frequently up-regulated in HCC samples. The IHC results for Elp3 expression and the phosphorylation level of AKT in HCC cells demonstrated a correlation with Elp3 expression. These results are highly consistent with our in vitro studies, showing that abnormally high expression of Elp3 could promote the phosphorylation level of AKT.
Elongator is up-regulated in HCC tissues. (A) Relative immunohistochemistry (IHC) staining in paired HCC tissue samples. The expression of Elp3, Elp4, p-AKT and MMP-2 in tumor tissues (T) and paired para-tumor tissues (P) from 15 HCC patients was determined by immunohistochemistry. Representative IHC images of matched tumor/para-tumor tissues were shown. (B) Semiquantitative analysis of Elp3 expression levels in tumor tissues and para-tumor tissues (H-score; left panel). The expression of Elp3 was classified into four degree ('-', '±', '+' and '++') dependent on H-score evaluation, shown in MATERIALS AND METHODS. The numbers of cases in different group were calculated in histogram (Case Number; right panel). (C) Semiquantitative analysis of Elp4 expression levels in tumor tissues and para-tumor tissues (left panel: H-score; right panel: Case Number). (D) Semiquantitative analysis of p-AKT expression levels in tumor tissues and para-tumor tissues (left panel: H-score; right panel: Case Number). (E) Semiquantitative analysis of MMP-2 expression levels in tumor tissues and para-tumor tissues (left panel: H-score; right panel: Case Number). (*P< 0.05, **P< 0.01)
Interesting, a previous study reported that a reduced expression of Elp3 was observed to be correlated to a poor prognosis of endometrioid adenocarcinoma based on immunohistochemical examination [47]. In vitro analysis and in vivo model are required to further confirm such an interesting observation. Multiple target genes may also contribute to the various and even contradictory conclusion on Elongator. However, our result is in agreement with many other reports [15, 17, 48,49].
Elongator deficiency enhanced the expression of pro-apoptotic factors such as Bax of p53-dependent genes in colon cancer cells, although no modulation was observed on the sensitivity to apoptosis [50]. Our results demonstrate Elongator promotes the migration and invasion of HCC cells, which may overlap the effect of p53-dependent pathway targeted by Elongator.
Previous study has proved that Elp1-Elp6 act as integral Elongator subunit to control cell motility and tumorigenicity. As a crucial actor for assembling Elongator into a functional complex, Elp1 stabilized the catalytic subunit Elp3. Elp5 constitutively binds Elp1 and Elp3. It is also required for optimal binding of Elp3 to Elp4. Here, we show that both Elp3 and Elp4 function in the migration and invasion of HCC cells, demonstrating that Elp3 and Elp4 work in an intact complex of Elongator. Similar results from the overexpression of Elp3 and Elp4 were possibly due to the enhancement of assembly of Elongator complex by the overexpession of individual subunit [51-54].
Our study has demonstrated that Elp3 and Elp4 of Elongator promoted metastasis of HCC cells through AKT/MMPs pathway. The identification of Elongator involved in the mobility of HCC cells help to clarify the mechanism of early metastasis of HCC, which may potentially benefit the clinic medicine in HCC.
Materials and Methods
Plasmids, Cell Lines and Culture, Reagents
The plasmid pFLAG-CMV4 was a gift from Dr. Danny Reinberg (NYU School of Medicine, Department of Biochemistry and Molecular Pharmacology Smilow Research Center, Room 213). The plasmid GV248 was purchased from Genechem Co. (Shanghai, China). Four shRNAs targeting different sequences were screened for the efficiency of knockdown of Elp3 and Elp4 respectively and shRNA with the strongest effect was selected for the establishment of stable cell lines applied in this paper. HepG2 and SMMC-7721 human HCC cell lines were obtained from our own laboratory of the Department of Cellular and Molecular Biology at Soochow University. HepG2 cells were grown in DMEM (Gibco-BRL, Shanghai, China) supplemented with 10% fetal bovine serum (FBS) (Sino-American Biotechnology Co, Shanghai, China), 1% antibiotics at 37 ℃ in 5% CO2. SMMC-7721 were maintained in RPMI-1640 (Gibco-BRL, Shanghai, China) supplemented with 10% FBS. LY294002 was purchased from Selleck Chemicals (Shanghai, China). Antibodies against Elp3, Elp4 were purchased from Abcam Biotechnology Company (Shanghai, China). Antibodies against MMP-2, MMP-9, PI3K, AKT, p-AKT, GAPDH and β-actin were purchased from Cell Signaling Technology Company (Shanghai, China).
Reverse transcription and Quantitative real time Polymerase Chain Reaction (qRT-PCR)
Total RNA was extracted using the Trizol reagent (Invitrogen, USA) according to the manufacturer's instructions. The RNA concentrations were quantified with the QubitFluorometer. Reverse transcription PCR was carried out by the Thermo script RT system (Invitrogen, Shanghai, China) according to the manufacturer's protocol. qPCR was performed in triplicate in a total volume of 20 μl containing 10 μl SsoFastEvaGreensupermix (Bio-Rad) with SYBR Green. The reaction was run on a Mini OpticonTM Real-time PCR instrument. Melting curve analysis was done to confirm the specificity of different genes. Fold-changes of genes with different treatment were calculated by normalizing the Ct values to the GADPH internal control. Primers for Elp3 were 5'-TTTGTAAAATGCCACAGGAGC -3' and 5'-GGCTCCTCTTGTAGAACTGCC-3'. Primers for Elp4 were 5'-GTTAAAAAATGAGAAATGGCGG-3' and 5'-TTCCTCCTTAGTCGCTGCAT-3'. Primers for MMP-2 were 5'-GACAACGCCCCCATACCA G-3' and 5'-CACTCGCCCCGTGTGTTAGT-3'. Primers for MMP-9 were 5'-ACGCAGACATCGTCATCCAGT-3' and 5'-ACGCAGACATCGTCATCCAGT-3'. Primers for GAPDH were 5'-GACCTGACCTGCCGTCTA-3' and 5'-AGGAGTGGGTGTCGCTGT-3'.
Western blot analysis
Briefly, each protein sample was separated by radical electrophoresis apparatus and fractionated by 10% sodium dodecyl-polyacrylamide gel electrophoresis (SDS-PAGE). The proteins were transferred onto polyvinylidenedifluoride (PVDF) membranes. After being blocked in 5% BSA in Tris-buffered saline with Tween-20 buffer (TBST) at 37 °C for 1 h, the membrane was incubated with various primary antibodies overnight at 4 °C with gently shaking and then followed with appropriate secondary antibodies for 2 h at room temperature. Signals were visualized by enhanced chemiluminescence (ECL) detection reagents according to the manufacturer's instructions. Western blot analysis with antibodies was performed using standard protocol. The densitometric analyses of bands were semi-quantified with ImageJ software.
Wound-healing assays
Cells were seeded on 6-well plate in triplicates. The wound was induced with a pipette tip scratch in the confluent cell monolayer, followed by serum starvation for 24 h. The cells were treated under appropriate conditions for 0-3 days. For each well, pictures were taken at time 0 d (just after the scratch), 1 d, 2 d and 3 d.
Transwell assays
For the transwell assays, serum-starved cells were trypsinized and plated at upper chamber 2×104 cells perchamber well, together with serum-free DMEM and 10% FBS placed in the low chamber. The cells were then allowed to migrate for 6 hours at 37 ℃. After the incubation, non-migrating cells were removed from the upper chamber with a cotton swap and the membrane inserts were stained with crystal violet solution. Motility was quantified by counting the cells that had migrated to the undersurface in the serum medium. Each well was photographed under a light microscope at a magnification of 100× and then counted.
In vivo mouse metastasis models
The animal operations and procedures were conducted in accordance with a protocol approved by the Committee on the Use of Live Animals in Teaching and Research of Soochow University. The mice were randomly divided into 5 groups: (1) PBS group, (2) HepG2 group, (3) HepG2-Vec group, (4) HepG3-Elp3o group, (5) HepG2-Elp3i, each group consisting of 5 mice. For lung metastasis study, 5×105 viable HepG2-luc hepatocellular carcinoma cells resuspended in 0.1ml PBS were injected subcutaneously into the tail vein of 15-20 g female nude mice (Animal Centre of Soochow University). Tumor cells metastatic nodules developed in the nude mice for 8 weeks later after injection. Mice were monitored daily. At the end of the eighth week, lung tissues were dissociated and evaluated.
Immunohistochemistry and Evaluation
Tissues from nude mice or human HCC samples were subjected to formalin fixation, paraffin embedding and sectioning for immunohistochemistry assays. The slides were detected by Bio-Streptavidin HRP detection systems. Sections were examined at high power (100×) under a standard light microscope. IHC staining was evaluated by taking into account both the intensity of staining and the percentage of positive cells [4, 55,56]. The reaction was developed under the visual supervision of a pathologist. For the intensity staining, a score index of 0, 1, 2 and 3 corresponding to negative, weak, moderate and strong staining intensity was used. The extent of staining was based on the percentage of positive cells: 0 (negative), 1 (1%-10%), 2 (11%-33%), 3 (34%-66%), or 4 (66%-100%). The final score was calculated by multiplying the staining area and the intensity score. Each case was ultimately considered negative if the final score was 0 (-) or 1 to 4 (±) and positive if the final score was 5 to 8 (+) or 9 to 12 (+ +). The final H-scores for each sample were assessed by summarizing the results of the intensities and extents of staining.
Statistical analysis
Values are expressed as the mean ± SD. Student's t-test was used to determine the significance of the difference between compared groups. All statistical treatments were performed using the GraphPad software. P<0.05 indicated statistical significant differences.
Supplementary Material
Supplementary figures.
Acknowledgements
This work was supported by Nature Science Foundation of China (81272301), Suzhou municipal project for science and technology of people's livelihood SYS201748, a project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions. This work was also supported by the Natural Science Foundation of Jiangsu Province (BK20161232) and Suzhou Special Project of Diagnosis and Treatment for key Clinical Disease (LCZX201715).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA: a cancer journal for clinicians. 2011;61:69-90
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA: a cancer journal for clinicians. 2015;65:5-29
3. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA: a cancer journal for clinicians. 2013;63:11-30
4. Huang W, Chen Z, Shang X, Tian D, Wang D, Wu K. et al. Sox12, a direct target of FoxQ1, promotes hepatocellular carcinoma metastasis through up-regulating Twist1 and FGFBP1. Hepatology. 2015;61:1920-33
5. Uchino K, Tateishi R, Shiina S, Kanda M, Masuzaki R, Kondo Y. et al. Hepatocellular carcinoma with extrahepatic metastasis: clinical features and prognostic factors. Cancer. 2011;117:4475-83
6. Park EK, Kim HJ, Kim CY, Hur YH, Koh YS, Kim JC. et al. A comparison between surgical resection and radiofrequency ablation in the treatment of hepatocellular carcinoma. Annals of surgical treatment and research. 2014;87:72-80
7. Yamashita Y, Imai D, Bekki Y, Takeishi K, Tsujita E, Ikegami T. et al. Surgical outcomes of anatomical resection for solitary recurrent hepatocellular carcinoma. Anticancer research. 2014;34:4421-6
8. Glatt S, Letoquart J, Faux C, Taylor NM, Seraphin B, Muller CW. The Elongator subcomplex Elp456 is a hexameric RecA-like ATPase. Nature structural & molecular biology. 2012;19:314-20
9. Otero G, Fellows J, Li Y, de Bizemont T, Dirac AM, Gustafsson CM. et al. Elongator, a multisubunit component of a novel RNA polymerase II holoenzyme for transcriptional elongation. Molecular cell. 1999;3:109-18
10. Krogan NJ, Greenblatt JF. Characterization of a six-subunit holo-elongator complex required for the regulated expression of a group of genes in Saccharomyces cerevisiae. Molecular and cellular biology. 2001;21:8203-12
11. Hawkes NA, Otero G, Winkler GS, Marshall N, Dahmus ME, Krappmann D. et al. Purification and characterization of the human elongator complex. The Journal of biological chemistry. 2002;277:3047-52
12. Versees W, De Groeve S, Van Lijsebettens M. Elongator, a conserved multitasking complex?. Molecular microbiology. 2010;76:1065-9
13. Svejstrup JQ. Elongator complex: how many roles does it play?. Current opinion in cell biology. 2007;19:331-6
14. Creppe C, Buschbeck M. Elongator: an ancestral complex driving transcription and migration through protein acetylation. Journal of biomedicine & biotechnology. 2011;2011:924898
15. Close P, Hawkes N, Cornez I, Creppe C, Lambert CA, Rogister B. et al. Transcription impairment and cell migration defects in elongator-depleted cells: implication for familial dysautonomia. Molecular cell. 2006;22:521-31
16. Li Q, Fazly AM, Zhou H, Huang SB, Zhang ZG, Stillman B. The Elongator Complex Interacts with PCNA and Modulates Transcriptional Silencing and Sensitivity to DNA Damage Agents. PLoS genetics. 2009:5
17. Creppe C, Malinouskaya L, Volvert ML, Gillard M, Close P, Malaise O. et al. Elongator controls the migration and differentiation of cortical neurons through acetylation of alpha-tubulin. Cell. 2009;136:551-64
18. Kakinuma N, Roy BC, Zhu Y, Wang Y, Kiyama R. Kank regulates RhoA-dependent formation of actin stress fibers and cell migration via 14-3-3 in PI3K-Akt signaling. The Journal of cell biology. 2008;181:537-49
19. Guo B, Gao J, Zhan J, Zhang H. Kindlin-2 interacts with and stabilizes EGFR and is required for EGF-induced breast cancer cell migration. Cancer letters. 2015;361:271-81
20. Baselga J, Swain SM. Novel anticancer targets: revisiting ERBB2 and discovering ERBB3. Nature reviews Cancer. 2009;9:463-75
21. Du Z, Caenepeel S, Shen Y, Rex K, Zhang Y, He Y. et al. Preclinical Evaluation of AMG 337, a Highly Selective Small Molecule MET Inhibitor, in Hepatocellular Carcinoma. Molecular cancer therapeutics. 2016;15:1227-37
22. Citri A, Yarden Y. EGF-ERBB signalling: towards the systems level. Nature reviews Molecular cell biology. 2006;7:505-16
23. Jiang WG, Martin TA, Parr C, Davies G, Matsumoto K, Nakamura T. Hepatocyte growth factor, its receptor, and their potential value in cancer therapies. Critical reviews in oncology/hematology. 2005;53:35-69
24. Wang RY, Chen L, Chen HY, Hu L, Li L, Sun HY. et al. MUC15 inhibits dimerization of EGFR and PI3K-AKT signaling and is associated with aggressive hepatocellular carcinomas in patients. Gastroenterology. 2013;145:1436-48 e1-12
25. Li H, Huang F, Fan L, Jiang Y, Wang X, Li J. et al. Phosphatidylethanolamine-binding protein 4 is associated with breast cancer metastasis through Src-mediated Akt tyrosine phosphorylation. Oncogene. 2014;33:4589-98
26. Guo J, Jie W, Shen Z, Li M, Lan Y, Kong Y. et al. SCF increases cardiac stem cell migration through PI3K/AKT and MMP2/9 signaling. International journal of molecular medicine. 2014;34:112-8
27. Yoo YA, Kang MH, Lee HJ, Kim BH, Park JK, Kim HK. et al. Sonic hedgehog pathway promotes metastasis and lymphangiogenesis via activation of Akt, EMT, and MMP-9 pathway in gastric cancer. Cancer research. 2011;71:7061-70
28. Wang C, Wen J, Zhou Y, Li L, Cui X, Wang J. et al. Apelin induces vascular smooth muscle cells migration via a PI3K/Akt/FoxO3a/MMP-2 pathway. The international journal of biochemistry & cell biology. 2015;69:173-82
29. Shi H, Wu Y, Wang Y, Zhou M, Yan S, Chen Z. et al. Liquiritigenin Potentiates the Inhibitory Effects of Cisplatin on Invasion and Metastasis Via Downregulation MMP-2/9 and PI3 K/AKT Signaling Pathway in B16F10 Melanoma Cells and Mice Model. Nutrition and cancer. 2015;67:761-70
30. Pan HC, Jiang Q, Yu Y, Mei JP, Cui YK, Zhao WJ. Quercetin promotes cell apoptosis and inhibits the expression of MMP-9 and fibronectin via the AKT and ERK signalling pathways in human glioma cells. Neurochemistry international. 2015;80:60-71
31. Chin YR, Toker A. Function of Akt/PKB signaling to cell motility, invasion and the tumor stroma in cancer. Cellular signalling. 2009;21:470-6
32. Li Y, Guo J, Wang C, Fan Z, Liu G, Wang C. et al. Circulation times of prostate cancer and hepatocellular carcinoma cells by in vivo flow cytometry. Cytometry Part A: the journal of the International Society for Analytical Cytology. 2011;79:848-54
33. Ke AW, Shi GM, Zhou J, Wu FZ, Ding ZB, Hu MY. et al. Role of overexpression of CD151 and/or c-Met in predicting prognosis of hepatocellular carcinoma. Hepatology. 2009;49:491-503
34. Berrier AL, Yamada KM. Cell-matrix adhesion. Journal of cellular physiology. 2007;213:565-73
35. Vihinen P, Ala-aho R, Kahari VM. Matrix metalloproteinases as therapeutic targets in cancer. Current cancer drug targets. 2005;5:203-20
36. Freije JM, Balbin M, Pendas AM, Sanchez LM, Puente XS, Lopez-Otin C. Matrix metalloproteinases and tumor progression. Advances in experimental medicine and biology. 2003;532:91-107
37. Chen JS, Wang Q, Fu XH, Huang XH, Chen XL, Cao LQ. et al. Involvement of PI3K/PTEN/AKT/mTOR pathway in invasion and metastasis in hepatocellular carcinoma: Association with MMP-9. Hepatology research: the official journal of the Japan Society of Hepatology. 2009;39:177-86
38. Park CM, Park MJ, Kwak HJ, Lee HC, Kim MS, Lee SH. et al. Ionizing radiation enhances matrix metalloproteinase-2 secretion and invasion of glioma cells through Src/epidermal growth factor receptor-mediated p38/Akt and phosphatidylinositol 3-kinase/Akt signaling pathways. Cancer research. 2006;66:8511-9
39. Chen JS, Huang XH, Wang Q, Huang JQ, Zhang LJ, Chen XL. et al. Sonic hedgehog signaling pathway induces cell migration and invasion through focal adhesion kinase/AKT signaling-mediated activation of matrix metalloproteinase (MMP)-2 and MMP-9 in liver cancer. Carcinogenesis. 2013;34:10-9
40. Mahajan K, Mahajan NP. PI3K-independent AKT activation in cancers: a treasure trove for novel therapeutics. Journal of cellular physiology. 2012;227:3178-84
41. Zhang Y, Wang L, Zhang M, Jin M, Bai C, Wang X. Potential mechanism of interleukin-8 production from lung cancer cells: an involvement of EGF-EGFR-PI3K-Akt-Erk pathway. Journal of cellular physiology. 2012;227:35-43
42. Wang SY, Chen B, Zhan YQ, Xu WX, Li CY, Yang RF. et al. SU5416 is a potent inhibitor of hepatocyte growth factor receptor (c-Met) and blocks HGF-induced invasiveness of human HepG2 hepatoma cells. Journal of hepatology. 2004;41:267-73
43. Yoneda N, Sato Y, Kitao A, Ikeda H, Sawada-Kitamura S, Miyakoshi M. et al. Epidermal growth factor induces cytokeratin 19 expression accompanied by increased growth abilities in human hepatocellular carcinoma. Laboratory investigation; a journal of technical methods and pathology. 2011;91:262-72
44. Li H, Ren Z, Kang X, Zhang L, Li X, Wang Y. et al. Identification of tyrosine-phosphorylated proteins associated with metastasis and functional analysis of FER in human hepatocellular carcinoma cells. BMC cancer. 2009;9:366
45. Li Y, Huang X, Zhong W, Zhang J, Ma K. Ganglioside GM3 promotes HGF-stimulated motility of murine hepatoma cell through enhanced phosphorylation of cMet at specific tyrosine sites and PI3K/Akt-mediated migration signaling. Molecular and cellular biochemistry. 2013;382:83-92
46. Xiang Q, Chen W, Ren M, Wang J, Zhang H, Deng DY. et al. Cabozantinib suppresses tumor growth and metastasis in hepatocellular carcinoma by a dual blockade of VEGFR2 and MET. Clinical cancer research: an official journal of the American Association for Cancer Research. 2014;20:2959-70
47. Wang Y, Ikeda JI, Rahadiani N, Mamat S, Ueda Y, Tian T. et al. Prognostic significance of elongator protein 3 expression in endometrioid adenocarcinoma. Oncology letters. 2012;3:25-9
48. Close P, Gillard M, Ladang A, Jiang ZS, Papuga J, Hawkes N. et al. DERP6 (ELP5) and C3ORF75 (ELP6) Regulate Tumorigenicity and Migration of Melanoma Cells as Subunits of Elongator. Journal of Biological Chemistry. 2012;287:32535-45
49. Wynshaw-Boris A. Elongator bridges tubulin acetylation and neuronal migration. Cell. 2009;136:393-4
50. Cornez I, Creppe C, Gillard M, Hennuy B, Chapelle JP, Dejardin E. et al. Deregulated expression of pro-survival and pro-apoptotic p53-dependent genes upon Elongator deficiency in colon cancer cells. Biochemical pharmacology. 2008;75:2122-34
51. Gu J, Sun D, Zheng Q, Wang X, Yang H, Miao J. et al. Human Elongator complex is involved in cell cycle and suppresses cell growth in 293T human embryonic kidney cells. Acta biochimica et biophysica Sinica. 2009;41:831-8
52. Xu H, Lin Z, Li F, Diao W, Dong C, Zhou H. et al. Dimerization of elongator protein 1 is essential for Elongator complex assembly. Proceedings of the National Academy of Sciences of the United States of America. 2015;112:10697-702
53. Kim JH, Lane WS, Reinberg D. Human Elongator facilitates RNA polymerase II transcription through chromatin. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:1241-6
54. Winkler GS, Kristjuhan A, Erdjument-Bromage H, Tempst P, Svejstrup JQ. Elongator is a histone H3 and H4 acetyltransferase important for normal histone acetylation levels in vivo. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:3517-22
55. Das P, Rawat R, Verma AK, Singh G, Vallonthaiel AG, Yadav R. et al. Immunohistochemical Expression of Antitissue Transglutaminase 2 in Tissue Injuries: An Interpretation Beyond Celiac Disease. Applied immunohistochemistry & molecular morphology: AIMM / official publication of the Society for Applied Immunohistochemistry. 2016
56. Putti TC, El-Rehim DM, Rakha EA, Paish CE, Lee AH, Pinder SE. et al. Estrogen receptor-negative breast carcinomas: a review of morphology and immunophenotypical analysis. Modern pathology: an official journal of the United States and Canadian Academy of Pathology, Inc. 2005;18:26-35
Author contact
![]() Corresponding authors: Wenxiang Wei, Department of Cell Biology and Institute of Bioengineering, School of Medicine, Soochow University, Suzhou, 215123 China. 86-512-5188-0107; wenxiangwedu.cn. and Zhi Pang, Department of Gastroenterology, The North District of the Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou, Jiangsu, 215008 China. 86-512-6236-3517; pangzhi0273com
Corresponding authors: Wenxiang Wei, Department of Cell Biology and Institute of Bioengineering, School of Medicine, Soochow University, Suzhou, 215123 China. 86-512-5188-0107; wenxiangwedu.cn. and Zhi Pang, Department of Gastroenterology, The North District of the Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou, Jiangsu, 215008 China. 86-512-6236-3517; pangzhi0273com
Received 2017-10-27
Accepted 2018-3-1
Published 2018-4-5
