ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2023; 19(14):4644-4656. doi:10.7150/ijbs.84108 This issue Cite
Research Paper
Inhibition of Galectin-9 sensitizes tumors to anthracycline treatment via inducing antitumor immunity
1. Department of Oncology, The Seventh Affiliated Hospital, Sun Yat-Sen University, Shenzhen, P. R. China.
2. Departments of Molecular and Cellular Oncology, The University of Texas MD Anderson Cancer Center, Houston, TX 77030, USA.
3. Department of Medical Oncology, Harbin Medical University Cancer Hospital, Harbin, P. R. China.
4. Department of Biological Science and Technology, College of Life Sciences, China Medical University, Taichung, Taiwan.
5. Cancer Biology and Precision Therapeutics Center and Research Center for Cancer Biology, China Medical University, Taichung, Taiwan.
6. Department of Cardiac Vascular Center, The Seventh Affiliated Hospital, Sun Yat-Sen University, Shenzhen, P. R. China.
7. Antibody Therapeutics, Inc., Hayward, CA 94545, USA.
8. Graduate Institute of Biomedical Sciences, China Medical University, Taichung, Taiwan.
9. Tianjin Key Laboratory of Lung Cancer Metastasis and Tumor Microenvironment, Lung Cancer Institute, Tianjin Medical University General Hospital, Tianjin, China.
10. Institute of Biomedical Sciences, Academia Sinica, Taipei, Taiwan.
11. Department of Pathology, Harbin Medical University Cancer Hospital, Harbin, P. R. China.
12. Department of Pathology, Affiliated Hospital of Qingdao University, Harbin, P. R. China.
13. State Key Laboratory for Chemistry and Molecular Engineering of Medicinal Resources, School of Chemistry & Pharmacy, Guangxi Normal University, Guilin, Guangxi, P. R. China.
14. Department of Hematology and Hematopoietic Cell Transplantation, City of Hope National Medical Center, Duarte, California, USA.
15. Institute of Biochemistry and Molecular Biology, China Medical University, Taichung, Taiwan.
16. Molecular Medicine Center, China Medical University Hospital, China Medical University, Taichung, Taiwan.
17. Department of Biotechnology, Asia University, Taichung, Taiwan.
*These authors contributed equally to this work.
Abstract
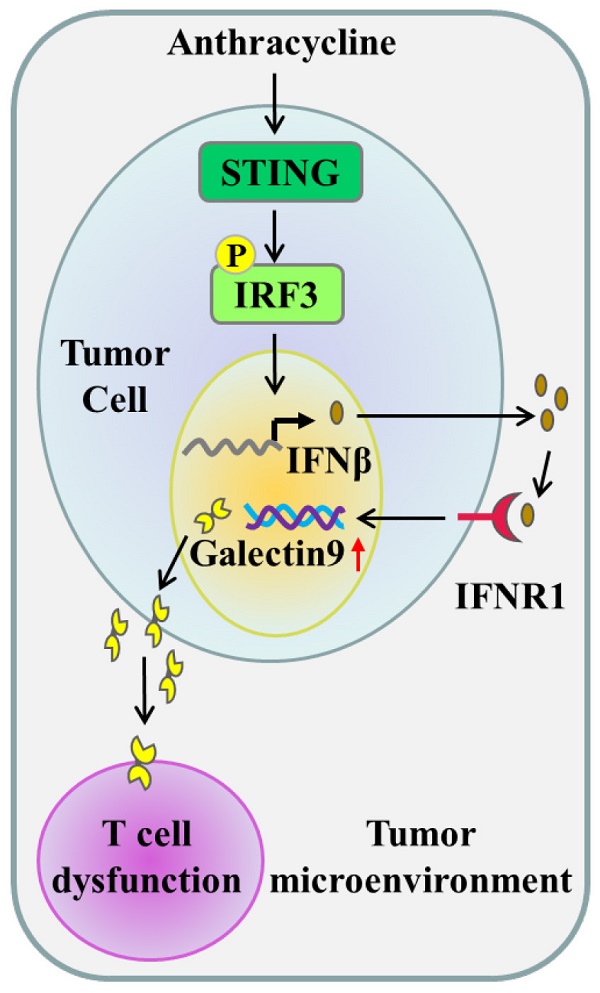
Anthracyclines are a class of conventionally and routinely used first-line chemotherapy drugs for cancer treatment. In addition to the direct cytotoxic effects, increasing evidence indicates that the efficacy of the drugs also depends on immunomodulatory effects with unknown mechanisms. Galectin-9 (Gal-9), a member of the β-galactoside-binding protein family, has been demonstrated to induce T-cell death and promote immunosuppression in the tumor microenvironment. Here, we asked whether anthracycline-mediated immunomodulatory activity might be related to Gal-9. We found that combining doxorubicin with anti-Gal-9 therapy significantly inhibited tumor growth and prolonged overall survival in immune-competent syngeneic mouse models. Moreover, Gal-9 expression was increased in response to doxorubicin in various human and murine cancer cell lines. Mechanistically, doxorubicin induced tumoral Gal-9 by activating the STING/interferon β pathway. Clinically, Gal-9 and p-STING levels were elevated in the tumor tissues of breast cancer patients treated with anthracyclines. Our study demonstrates Gal-9 upregulation in response to anthracyclines as a novel mechanism mediating immune escape and suggests targeting Gal-9 in combination with anthracyclines as a promising therapeutic strategy for cancer treatment.
Keywords: anthracyclines, Galectin-9, tumor microenvironment, STING, antitumor immunity
