ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2024; 20(1):61-77. doi:10.7150/ijbs.83599 This issue Cite
Research Paper
Cancer Differentiation Inducer Chlorogenic Acid Suppresses PD-L1 Expression and Boosts Antitumor Immunity of PD-1 Antibody
1. Institute of Medicinal Biotechnology, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100050, China.
2. State Key Laboratory of Bioactive Substances and Function of Natural Medicine, Institute of Materia Medica, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100050, China.
3. State Key Latoratory of Phytochemistry and Plant Resource in West China, Kunming Institute of Botany, Chinese Academy of Sciences, Kunming, Yunnan, 650201, China.
4. Jiuzhang Biochemical Engineering Science and Technology Development Co., Ltd, Chengdu, Sichuan, 610041, China.
5. Department of Neuro-oncology, Cancer Center, Beijing Tiantan Hospital, Capital Medical University, Beijing 100070, China.
# Equal contribution to the work.
Abstract
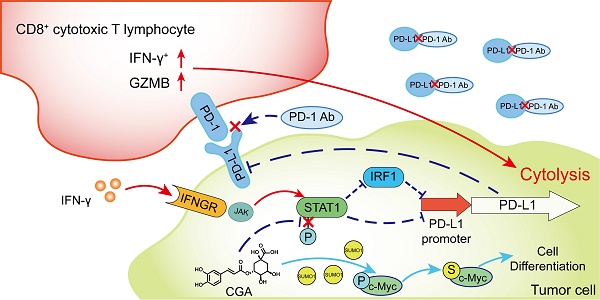
As immune checkpoint inhibitors have shown good clinical efficacy, immune checkpoint blockade has become a vital strategy in cancer therapy. However, approximately only 12.5% patients experience benefits from immunotherapy. Herein, we identified the cancer differentiation inducer chlorogenic acid (CGA, now in the phase II clinical trial in China for glioma treatment) to be a small-molecular immune checkpoint inhibitor that boosted the antitumor effects of the anti-PD-1 antibody. CGA suppressed the expression of PD-L1 induced by interferon-γ in tumor cell culture through inhibition of the p-STAT1-IRF1 pathway and enhanced activity of activated T-cells. In two murine tumor xenografts, combination therapy of CGA with anti-PD-1 antibody decreased the expression of PD-L1 and IRF1 and increased the inhibitory effect of the anti-PD-1 antibody on tumor growth. Particularly, the activity of tumor infiltrated T cells was enhanced by CGA. CGA improved the gene expression of granzymes in tumor-infiltrated immune cells. In conclusion, through induction of differentiation, CGA appeared to suppress the expression of PD-L1 on cancer cells, effectively promoting infiltrated T cells in the tumor and boosting the antitumor effect of the anti-PD-1 antibody. Thus, CGA might serve as a promising agent to enhance anticancer immunotherapy if combined with anti-PD-1 antibodies.
Keywords: Chlorogenic acid, Cancer differentiation inducer, PD-L1, Anti-PD-1 antibody, Combination therapy
