ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2011; 7(5):567-574. doi:10.7150/ijbs.7.567 This issue Cite
Short Research Communication
miR-148a Promoted Cell Proliferation by Targeting p27 in Gastric Cancer Cells
1. State Key Laboratory of Proteomics, Genetic Laboratory of Development and Diseases, Institute of Biotechnology, Beijing, P.R. China
2. General Surgery Department, PLA General Hospital & PLA Postgraduate School of Medicine, Beijing, P.R. China
3. Model Organism Division, E-institutes of Shanghai Universities, Shanghai Jiaotong University, P.R. China
4. General Surgery Department, China-Japan Friendship Hospital, Beijing, P.R. China
5. Neurosurgery Department, China-Japan Friendship Hospital, Beijing, P.R. China
# Shui-Long Guo and Zheng Peng contributed equally to this work.
Abstract
Accumulating evidence has shown that miRNAs are aberrantly expressed in human gastric cancer and crucial to tumorigenesis. Herein, we identified the role of miR-148a in gastric cell proliferation. miR-148a knockdown inhibited cell proliferation in gastric cancer cell lines. Conversely, miR-148a overexpression promoted cell proliferation and cell cycle progression. p27, a key inhibitor of cell cycle, was verified as the target of miR-148a, indicating miR-148a might downregulate p27 expression to promote gastric cell proliferation. Moreover, we confirmed that miR-148a expression was frequently and dramatically downregulated in human advanced gastric cancer tissues, and observed a good inverse correlation between miR-148a and p27 expression in tumor samples. Thus, our results demonstrated that miR-148a downregulation might exert some sort of antagonistic function in cell proliferation, rather than promote cell proliferation in gastric cancer.
Keywords: gastric cancer, cell proliferation, miR-148a, p27
Introduction
Gastric cancer remains one of the most frequently occurring malignancies, and ranks as the second leading cause of mortality next to lung cancer [1]. Although alterations in a large number of oncogenes and tumor suppressor genes have been reported in gastric carcinomas [2], the molecular mechanisms underlying the pathogenesis of gastric carcinomas remain to be fully defined.
miRNAs are small, noncoding, single-stranded RNAs that bind to complementary sequences in the 3' untranslated region (UTR) of their target mRNAs and induce mRNA degradation or translational repression [3]. miRNAs are involved in various cell functions, including proliferation, differentiation and apoptosis. Characteristic miRNA signatures have been identified for human gastric cancer, which predict disease status and clinical outcome [4-9]. Among them, miR-148a was one of the most frequently downregulated miRNAs in miRNA profiles of gastric cancers [4, 7, 8], and correlated with tumor size [9] and node metastasis [7]. Previous studies have shown that miR-148a plays important roles in cancer cell proliferation, response to anti-cancer chemotherapy, and metastasis in some tumor types including cholangiocarcinoma, prostate, esophageal and colon cancers [10-15]. For instance, downregulation of cytokine interleukin-6-regualted miR-148a contributes to cell proliferation by its target DNMT1 in malignant cholangiocytes [10]. In prostate cancer PC3 cells, miR-148a could inhibit cell proliferation, migration and invasion, and increase the sensitivity to anti-cancer drug [11]. However, the functional roles and target genes of miR-148a in gastric cancer remain unknown.
In this study, we investigated the role of miR-148a in cell proliferation of gastric cancer cells. Unexpectedly, inhibition of miR-148a expression repressed cell proliferation in gastric cancer cell lines. Conversely, transient and stable overexpression of miR-148a promoted cell proliferation and cell cycle progression. Moreover, p27, a key inhibitor of cell cycle, was identified as the direct target of miR-148a, suggesting that miR-148a might exert its function through the downregulation of p27. Finally, we confirmed that miR-148a expression was frequently downregulated in human advanced gastric cancer tissues, and observed a good inverse correlation between miR-148a and p27 expression in tumor samples. Taken together, our findings suggested that miR-148a downregulation might exert antagonistic function in gastric cancer cell proliferation during gastric tumorigenesis.
Results and Discussion
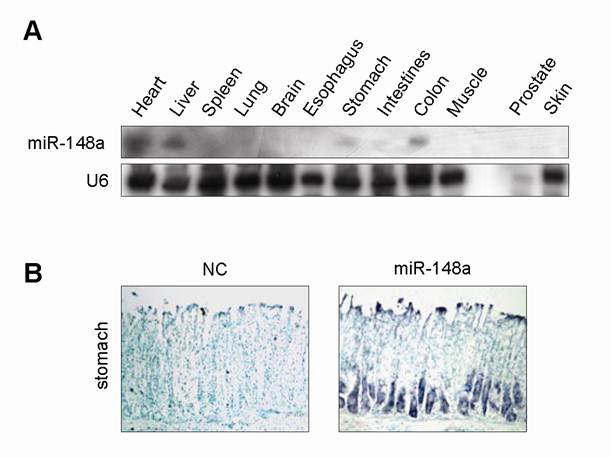
To explore the tissue-specific expression pattern of miR-148a, we detected miR-148a expression in different tissues of mouse by Northern blot. miR-148a expression was relatively high in stomach, heart, intestine, colon and liver (Fig. 1A). To visualize the spatial localization of miR-148a in whole-stomach tissue of mouse, we applied locked nucleic acid in situ hybridization. Unlike with the mismatch control probe (scrambled miRNA), hybridization with miR-148a-specific probe revealed that miR-148a was expressed at the bottom half of glandular gastric epithelium, where parietal and chief cells resided (Fig. 1B).
Tissue-specific and cell-specific expression pattern of miR-148a in mouse (A), miR-148a expression was detected in various tissues of mouse by Northern blot. (B), ISH analysis showed that miR-148a expression was located at bottom half of gastric epithelium in mouse. Original magnification, ×200.
miR-148a promoted cell proliferation in gastric cancer cells. (A), miR-148a expression was measured by Northern blot in 6 human gastric cancer cell lines. (B), miR-148a downregulation with AS was confirmed by Northern Blot in a representative transfectant. (C), miR-148a downregulation inhibited cell proliferation in AGS, BGC-823 and SGC-7901 cells. Gastric cancer cells were transfected with miR-148a AS or scrambled miRNA AS (as a negative-control, NC AS). Cell viability was determined by cell count 72 and 96 hours after transfection. The numbers of viable cells transfected with NC AS were set at 1. (D), miR-148a promoted cell proliferation in AGS cells. miR-148a overexpression with mimic were confirmed by Northern Blot in AGS cells. Cell viability was determined by cell count 72 and 96 hours after transfection. (E), Northern blot revealed that miR-148a expression was increased in stably miR-148a-expressing AGS cells (miR-148a) compared with AGS stably transfected with empty vector (vector). Cell count showed that the cell numbers at 48 and 72 hours were increased in stably miR-148a-expressing AGS cells. (F), Cell cycle analysis showed that AGS cells transiently transfected with miR-148a mimic exhibited increased population of cells at S phase 24 hours after transfection. (G), Stably miR-148a-expressing AGS cells (AGS-miR-148a) also showed increased population of cells at S phase. (H), FCAS analysis showed no obvious change was observed in apoptosis induced by Etoposide. Values represent mean ± SD. n=3. *P<0.05, **P<0.01.
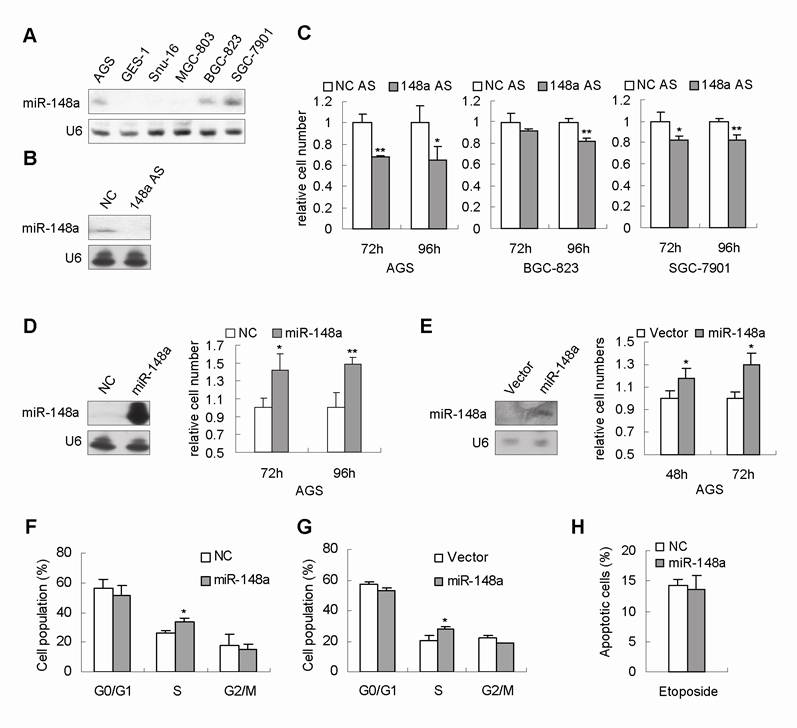
Several independent lines of evidence have shown that miR-148a was one of the most frequently downregulated microRNAs in human gastric carcinomas [4, 7, 8]. These studies led us to speculate that miR-148a dowregulation could contribute to gastric cell proliferation. We first detected miR-148a expression in 6 human gastric cancer cell lines. Northern blot results showed that miR-148a expression in AGS, BGC-823 and SGC-7901 cells was relatively higher than that in GES-1, BGC-803 and SNU-16 cells (Fig. 2A). Next, to evaluate the role of miR-148a downregulation in gastric cancer cells, we engineered miR-148a with antisense oligonucleotides (miR-148a AS) to decrease the endogenous miR-148a expression in 3 human gastric cancer cell lines, AGS, BGC-823 and SGC-7901, respectively. The downregulation of miR-148a were confirmed by Northern Blot (Fig. 2B). Cellular proliferation assays were performed on the miR-148a-AS-tranfected cells. To our surprise, miR-148a knockdown inhibited cell proliferation by 20-40% after 96 hours in AGS, BGC-823, and SGC-7901 cells (Fig. 2C, p<0.05).
To substantiate the function of miR-148a in gastric cell proliferation, we transfected AGS cells with miR-148a mimic. Northern Blot confirmed the increased expression of miR-148a in the transfectant (Fig. 2D). Viable cell numbers were counted at 72 and 96 hours after transfection. Consistent with our prediction, the cell numbers of miR-148a-transfected AGS cells at 72 and 96 hours were found to be increased by 42% and 48%, respectively, compared with that of scrambled-miRNA-transfected cells (Fig. 2D, p<0.05). Moreover, we established a stable cell line (AGS-miR-148a) in which miR-148a expression was significantly higher than that in AGS stably transfected with empty vector (AGS-vector) (Fig. 2E). Consistently, the cell numbers of AGS-miR-148a at 48 and 72 hours were increased by 18% and 30%, respectively, compared with that in control cells (Fig. 2E, p<0.05).
It is known that abnormal cell proliferation is related to the altered cell cycle. Cell cycle analysis was conducted in AGS cells transiently transfected with miR-148a mimic. The proportion of cells corresponding to S phase was higher in miR-148a-transfected AGS (34%±2.9%) than that in control cells (26%±2.1%, p<0.05, Fig. 2F). Similar results were obtained in the stable miR-148a-expressing AGS cells (Fig. 2G). These data suggested that miR-148a could promote cell proliferation by increasing the population of cells in S phase. In addition, we checked the effect of miR-148a on apoptosis by FACS analysis, and no obvious differences were observed between miR-148a-tansfected AGS cells and control cells (Fig. 2H).
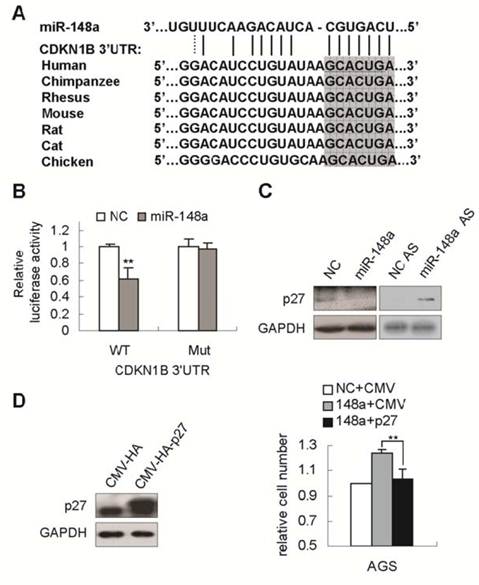
To determine the mechanisms by which miR-148a promotes gastric cell proliferation, bioinformatics analysis was performed to search for miR-148a targets. CDNK1B (encoding p27), a gene critical for cell cycle [15], was predicted as a putative target of miR-148a (Fig. 3A). p27 mRNA 3'UTR has a single predicted binding site that is highly conserved in mammals, and in chicken (Fig. 3A). To determine whether miR-148a could directly regulate p27, p27 mRNA 3'UTR encompassing the predicted binding site with or without mutation that would disrupt miRNA interaction (underlined in Fig. 3A), were inserted to the downstream of luciferase open reading frame in pGL3-CM reporter vector. When introduced into SGC-7901 cells, the wild type p27 3'UTR reporter showed a 39% reduction in luciferase activity in the miR-148a-transfected cells compared with that in scrambled-miRNA-transfected cells (Fig. 3B, p<0.01). Mutation of miR-148a binding site abrogated the repression of luciferase activity caused by miR-148a overexpression (Fig. 3B). Next, we pursued the ability of miR-148a to regulate the expression of endongenous p27. miR-148a overexpression with mimic in AGS cells dramatically repressed p27 expression (Fig. 3C). Conversely, miR-148a knockdown by AS dramatically enhanced p27 expression in BGC-823 cells (Fig. 3C), indicating that endogenous miR-148a could regulate p27 abundance. To examine the functional interrelationship between miR-148a and p27, AGS cells was simultaneously transfected with miR-148a mimic and p27-expressing vector (CMV-p27). As expected, overexpression of p27 dramatically rescued miR-148a-induced growth promotion (Fig. 3D, p<0.01), indicating that miR-148a enhanced cell proliferation possibly by repressing p27.
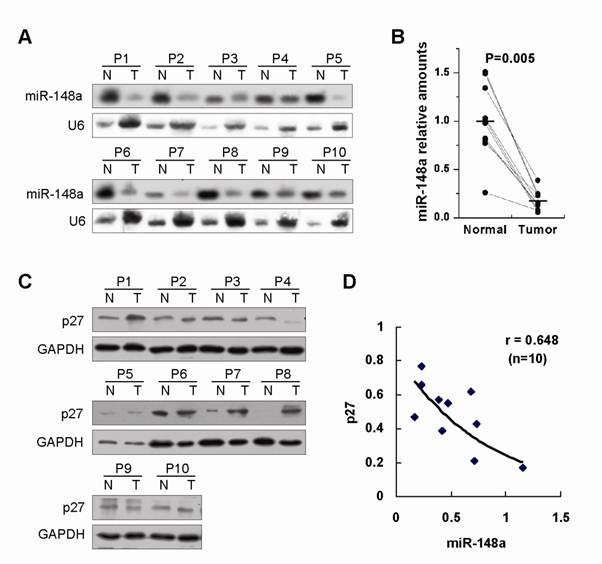
Finally, we detected the expression of miR-148a and p27 in 10 advanced gastric primary tumors, each one paired with adjacent nontumor gastric tissue from the same patient (Fig. 4A and 4C). Northern blot assays showed that the reduced expression of miR-148a was found in 100% (10/10) of human advanced gastric tumor tissues as compared with adjacent normal counterparts (Fig. 4A and 4B, p=0.005), indicating that miR-148a downregulation was a very common event in advanced gastric cancer. Furthermore, the inverse correlation was observed between miR-148a and p27 in tumor samples (r=0.648, Fig. 4D), suggesting that miR-148a could negatively regulate p27 expression in human gastric cancer.
Previous studies have shown that miR-148a could target its distinct targets to regulate cell proliferation in some cancer cells [10,12,16]. DNMT1, a direct target of miR-148a, plays important role to suppress cell proliferation in malignant cholangiocytes [10]. miR-148a, also targets and suppresses Microphthalmia associated transcription factor (Mitf), an important regulator involved in melanoma progression [16]. In the present study, we identified a new target, p27, possibly by which miR-148a could promote cell cycle progression and cell proliferation. Similarly, miR-148a could promote cell growth by repressing its target CAND1 expression in prostate cancer LNCaP cells [12]. These findings indicated that the different roles of miR-148a in regulating cell proliferation might depend on its distinct targets in different tumor cell types. miR-148b, which has the same “seed sequence” as miR-148a, was downregulated in gastric cancer, and could inhibit cell proliferation by targeting CCKBR [17]. Although CCKBR was a predicted target of miR-148a by bioinformatic analysis, it was not a real target of miR-148a by Western blot and luciferase reporter assay in our experimental system (data not shown), indicating that miR-148a and miR-148b, both belonging to the same microRNA family, might exert opposite roles in one cell type possibly by regulating distinct targets.
p27 was a direct target of miR-148a in gastric cancer cells. (A), The possible miR-148a binding site in CDNK1B (p27) mRNA 3'UTR was predicted by bioinformatic analyses. The mutant seed sequence was underlined. p27 had a single predicted binding site in mRNA 3'UTR that was highly conserved in mammals, and in chicken (the complementary sequences of seed sequence of miR-148a were highlighted). (B), Luciferase activities were measured in SGC-7901 cells cotransfected with the reporter constructs containing 3'UTR of p27 with or without mutant (underlined in A) and miR-148a mimic or negative-control miRNA. (C), Western blot assays confirmed that p27 expression were downregulated by miR-148a mimic in AGS cells and upregulated by miR-148a AS in BGC-823 cells. (D), p27 expression was significantly increased in p27-transfected AGC cells (CMV-HA-p27) compared with that in control cells (CMV-HA). The numbers of viable AGS cells were determined by cell count 72 hours after cotransfection with miR-148a mimic and CMV-HA-p27 vector. Values represent mean ± SD. n=3. **P<0.01
miR-148a was frequently and dramatically downregulated in human advanced gastric cancer and showed an inverse correlation with p27 expression in tumor samples. (A), miR-148a expression was determined by Northern blot in 10 advanced gastric primary tumors, each one paired with adjacent nontumor gastric tissue from the same patient. (B), Statistically significant downregulation of miR-148a in gastric cancers over matching normal tissues. P=0.005 by Wilcoxon signed rank test. Bars represent mean. (C), p27 expression was examined by Western blot in the same samples. (D), The relationship between miR-148a and p27 expression was detected by Spearman rank correlation analysis in tumor samples. r=0.648. p<0.05.
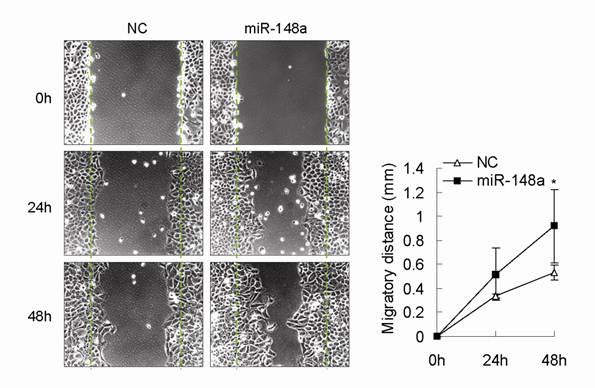
In this study, we observed that miR-148a was frequently downregulated in human advanced gastric cancer samples. We also showed that inhibition of miR-148a suppressed cell proliferation in different gastric cancer cell lines possibly by upregulating its target p27, suggesting that miR-148a downregulation in human gastric cancer might inhibit but not promote gastric cancer cell proliferation during gastric tumorigenesis. Some evidence has shown that miR-148a is associated with migration, metastasis, and drug resistance in some tumor types [7,11,13,14], indicating that miR-148a downregulation might contribute to these biological processes in gastric cancer. In fact, we also found that miR-148a overexpression with mimic could promote gastric cell migration (Fig. S1, p<0.05).
Conclusion
In this study, we investigated the role of miR-148a in regulating cell proliferation of gastric cancer cells. We observed that miR-148a expression was frequently and dramatically downregulated in human advanced gastric cancer tissues compared with the matched adjacent normal tissues. Unexpectedly, miR-148a knockdown inhibited cell proliferation in gastric cancer cell lines. We further showed that transient and stable overexpression of miR-148a could promote cell proliferation and cell cycle progression. Moreover, p27, a key inhibitor of cell cycle, was verified as a direct target of miR-148a, suggesting that miR-148a might promote gastric cell proliferation by depressing p27 expression. Collectively, our findings indicated that miR-148a downregulation might exert some sort of antagonistic function in gastric cancer cell proliferation during gastric tumorigenesis.
Methods
Human gastric tissues. Paired gastric cancer and adjacent non-tumor gastric tissues were obtained after review and approval by the General Surgery Department of PLA General Hospital of China. Both tumor and non-tumor tissues were histologically confirmed. Informed consent was obtained from each patient before operation. Tissue samples were immediately frozen in liquid nitrogen until RNA extraction.
Cell culture and transfection. The gastric cancer cell lines AGS, GES-1, SNU-16, MGC-803, BGC-823 and SGC-7901 were cultured in Dulbecco's Modified Eagle Medium supplemented with 2mM L-glutamine, 1% penicillin/streptomycin, and 10% fetal bovine serum (Hyclone) at 37℃ and 5% CO2. AGS, BGC-823 and SGC-7901 cells were individually plated onto tissue culture plates or dishes 24 hours before transfection. Transient transfection of miRNA mimics or anti-sense (AS) and control oligonucleotides (Genepharm) at a final concentration of 50 nM was accomplished with lipofectmine 2000 reagent (Invitrogen). To establish stably transfected AGS, pIRES2-EGFP vector (BD Bioscience) was inserted with 164bp genomic sequence of miR-148a precursor. The primers for PCR amplification are: sense, 5'- AAAAGATCTGAACACACCTGCAGGAAGAA-3', anti-sense, 5'- AAAAAAGCTTCTGGCGTCTGGAGCACTG-3'. Stably transfected cells were selected using G418 (500μg/ml, Gibco).
Cell cycle analysis. Cells were harvested and processed by standard methods by using propidium iodide staining of cell DNA. Cell samples were analyzed with a FACScan flow cytometer (BD Biosciences). Histograms were analyzed for cell cycle compartments using ModFit version 2.0 (Verity Software House).
Proliferation assays. 2×104 Cells were seeded into 24-well plate in triplicate, and the cell number was counted 48, 72 or 96 hours later under microscope using a hemocytometer.
Apoptosis assay. AGS cells were treated with etoposide (30μg/ml) or a matched concentration of diluents for 48 hours. After the treatment, cells were rinsed twice in cold PBS, and then analyzed for apoptosis by a PE-labeled Annexin-V/7-AAD assay. Cell samples (10,000 events) were analysed in a FACScan flow cytometer (BD Biosciences), and data were processed using FACScan software.
Luciferase activity assay. The 3'UTR region of human p27 mRNA that containing miR-148a target site was amplified using specific primers: sense, 5'- CCGCTCGAGGTTTATCAGATACATCACTGCTTGA-3', anti-sense, 5'- CCGACGCGTTTGGCTCAGTATGCAACCTTTT-3', and cloned into pGL3-CM to generate luciferase reporter vector. The mutant vector of p27 3′UTR was also constructed. The method for luciferase assay was described previously [18]. In brief, reporter vectors were co-transfected with renilla luciferase expressing vector to normalize transfection efficiency. Luciferase activity assays were done at 48 hours after transfection using a Dural-luciferase® reporter assay system (promega) using LB 960 Centro XS3 luminometer (Berthold Technologies).
Northern blot. Northern blot analysis was performed as described using 20 μg total RNA from each sample [18]. Probes were labeled with 32P γ-ATP using T4 polynucleotide kinase (New England Biolabs).
miRNA in situ hybridization. miRNA in situ hybridization was performed as previously described [19] using locked nucleic acid (LNA) probes complementary to mouse mature miR-148a (Exiqon) or scrambled control sequence (Exiqon). LNA probes were hybridized to mouse gastric tissue at 60°C.
Western blot. Cells were homogenized in lysis buffer supplemented with protease inhibitor cocktail (Roche). Proteins (25 ug) were electrophoresed on SDS-PAGE and transferred onto PVDF membranes. Immunoblotting was done using human p27 (Cell Signaling) and GAPDH (ZSGB-Bio) antibodys.
Statistical analysis. All values were expressed as mean±SD. Statistical analysis was performed by 2-tailed Student's t test, and results were considered statistically significant at P < 0.05. Wilcoxon signed rank test was used to analyze the statistically significant downregulation of miR-148a expression in gastric cancers. Correlation coefficient was calculated by Spearman rank correlation analysis.
Ackowledgments
This work was supported by grants 2011CB504200 and 2011CB901601 from Chinese National Key Program on Basic Research, grant 2009ZX09501-027 from the Chinese Key Program for Drug Invention, grant 2009ZX10002-016 from the Chinese Key Project for the Infections Disease, grants 31030040, 30700423 and 30900863 from National Natural Science Foundation of China, and grant 5082018 from Beijing Natural Science Foundation of China.
Conflict of Interests
The authors have declared that no conflict of interest exists.
References
1. Parkin DM, Bray F, Ferlay J. et al. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55(2):74-108
2. Nobili S, Bruno L, Landini I. et al. Genomic and genetic alterations influence the progression of gastric cancer. World J Gastroenterol. 2010;17(3):290-9
3. Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(2):281-97
4. Ueda T, Volinia S, Okumura H. et al. Relation between microRNA expression and progression and prognosis of gastric cancer: a microRNA expression analysis. Lancet Oncol. 2010;11(2):136-46
5. Guo J, Miao Y, Xiao B. et al. Differential expression of microRNA species in human gastric cancer versus non-tumorous tissues. J Gastroenterol Hepatol. 2009;24(4):652-7
6. Li X, Zhang Y, Ding J. et al. Survival prediction of gastric cancer by a seven-microRNA signature. Gut. 2010;59(5):579-85
7. Tchernitsa O, Kasajima A, Schafer R. et al. Systematic evaluation of the miRNA-ome and its downstream effects on mRNA expression identifies gastric cancer progression. J Pathol. 2010;222(3):310-9
8. Tsukamoto Y, Nakada C, Noguchi T. et al. MicroRNA-375 is downregulated in gastric carcinomas and regulates cell survival by targeting PDK1 and 14-3-3zeta. Cancer Res. 2010;70(6):2339-49
9. Chen Y, Song Y, Wang Z. et al. Altered expression of MiR-148a and MiR-152 in gastrointestinal cancers and its clinical significance. J Gastrointest Surg. 2010;14(7):1170-9
10. Braconi C, Huang N, Patel T. MicroRNA-dependent regulation of DNA methyltransferase-1 and tumor suppressor gene expression by interleukin-6 in human malignant cholangiocytes. Hepatology. 2010;51(3):881-90
11. Fujita Y, Kojima K, Ohhashi R. et al. MiR-148a attenuates paclitaxel resistance of hormone-refractory, drug-resistant prostate cancer PC3 cells by regulating MSK1 expression. J Biol Chem. 2010;285(25):19076-84
12. Murata T, Takayama K, Katayama S. et al. miR-148a is an androgen-responsive microRNA that promotes LNCaP prostate cell growth by repressing its target CAND1 expression. Prostate Cancer Prostatic Dis. 2010;13(4):356-61
13. Lujambio A, Calin GA, Villanueva A. et al. A microRNA DNA methylation signature for human cancer metastasis. Proc Natl Acad Sci U S A. 2008;105(36):13556-61
14. Hummel R, Watson DI, Smith C. et al. Mir-148a Improves Response to Chemotherapy in Sensitive and Resistant Oesophageal Adenocarcinoma and Squamous Cell Carcinoma Cells. J Gastrointest Surg. 2011Mar;15(3):429-38
15. Chu IM, Hengst L, Slingerland JM. The Cdk inhibitor p27 in human cancer: prognostic potential and relevance to anticancer therapy. Nat Rev Cancer. 2008;8(4):253-67
16. Haflidadottir BS, Bergsteinsdottir K, Praetorius C. et al. miR-148 regulates Mitf in melanoma cells. PLoS One. 2010;5(7):e11574
17. Song YX, Yue ZY, Wang ZN. et al. MicroRNA-148b is frequently down-regulated in gastric cancer and acts as a tumor suppressor by inhibiting cell proliferation. Mol Cancer. 2010;10:1
18. Sun Q, Zhang Y, Yang G. et al. Transforming growth factor-beta-regulated miR-24 promotes skeletal muscle differentiation. Nucleic Acids Res. 2008;36(8):2690-9
19. Obernosterer G, Martinez J, Alenius M. Locked nucleic acid-based in situ detection of microRNAs in mouse tissue sections. Nat Protoc. 2007;2(6):1508-14
Figures
miR-148a promoted gastric cell migration in AGS cells. Scratch wound-healing assay was conducted in miR-148a-transfected AGS cells and control cells. Migration distance was measured at 0, 24, 48 hours after cells were scratched. Values represent mean ± SD. n=3. *P<0.05.
Author contact
![]() Corresponding author: Xiao Yang, Ph.D., Institute of Biotechnology, 20 Dongdajie, Beijing 100071, P.R. China. Tel/Fax: 86-10-63895937(O); E-mail: yangxbmi.ac.cn. Or to: Yan Teng, Ph.D., Institute of Biotechnology, 20 Dongdajie, Beijing 100071, P.R. China. E-mail: tengyan0919com
Corresponding author: Xiao Yang, Ph.D., Institute of Biotechnology, 20 Dongdajie, Beijing 100071, P.R. China. Tel/Fax: 86-10-63895937(O); E-mail: yangxbmi.ac.cn. Or to: Yan Teng, Ph.D., Institute of Biotechnology, 20 Dongdajie, Beijing 100071, P.R. China. E-mail: tengyan0919com
Received 2011-2-21
Accepted 2011-4-30
Published 2011-5-5
