Impact Factor ISSN: 1449-2288
Int J Biol Sci 2016; 12(11):1341-1356. doi:10.7150/ijbs.15992 This issue Cite
Research Paper
Ginsenoside Rg1 Protects against Oxidative Stress-induced Neuronal Apoptosis through Myosin IIA-actin Related Cytoskeletal Reorganization
1. State Key Laboratory of Natural Products, Jiangsu Key Laboratory of TCM Evaluation and Translational Research, Department of Complex Prescription of TCM, China Pharmaceutical University, 639 Longmian Road, Nanjing, 211198, China;
2. Department of Neurology, Jinling Hospital, Nanjing University School of Medicine, 305 East Zhongshan Road, Nanjing, 210002, China.
3. Pharmacy Department, The First Affiliated Hospital of Nanchang University, 17 Yongwai Street, Nanchang, 330006, China.
4. Cellular and Molecular Biology Center, China Pharmaceutical University, 639 Longmian Road, Nanjing, 211198, China.
5. State Key Laboratory of Quality Research in Chinese Medicine, Macau Institute for Applied Research in Medicine and Health, Macau University of Science and Technology, Macau, China.
*The authors contributed equally to this work.
Abstract
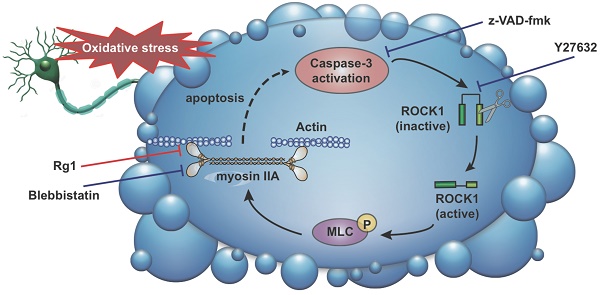
Oxidative stress-induced cytoskeletal dysfunction of neurons has been implicated as a crucial cause of cell apoptosis or death in the central nervous system (CNS) diseases, such as neurodegenerative and psychiatric diseases. The application of neuroprotectants rescuing the neurons from cytoskeletal damage and apoptosis can be a potential treatment for these CNS diseases. Ginsenoside Rg1 (Rg1), one of the major active components of ginseng, has been reported possessing notable neuroprotective activities. However, there is rare report about its effect on cytoskeleton and its undergoing mechanism. The current study is to reveal the regulatory effects of Rg1 on cytoskeletal and morphological lesion in oxidative stress-induced neuronal apoptosis. The results demonstrated that pre-treatment with Rg1 (0.1-10 μM) attenuated hydrogen peroxide (H2O2)-induced neuronal apoptosis and oxidative stress through reducing the intracellular reactive oxygen species (ROS) production and methane dicarboxylic aldehyde (MDA) level. The Rg1 treatment also abolished H2O2-induced morphological changes, including cell rounding, membrane blebbing, neurite retraction and nuclei condensation, which were generated by myosin IIA-actin interaction. These effects were mediated via the down-regulation of caspase-3, ROCK1 (Rho-associated kinase1) activation and myosin light chain (MLC, Ser-19) phosphorylation. Furthermore, inhibiting myosin II activity with blebbistatin partly blocked the neuroprotective effects of Rg1. The computer-aided homology modelling revealed that Rg1 preferentially positioned in the actin binding cleft of myosin IIA and might block the binding of myosin IIA to actin filaments. Accordingly, the neuroprotective mechanism of Rg1 is related to the activity that inhibits myosin IIA-actin interaction and the caspase-3/ROCK1/MLC signaling pathway. These findings put some insights into the unique neuroprotective properties of Rg1 associated with the regulation of myosin IIA-actin cytoskeletal structure under oxidative stress and provide experimental evidence for Rg1 in CNS diseases.
Keywords: Ginsenoside Rg1, oxidative stress, neuronal apoptosis, cytoskeleton, myosin IIA-actin interaction

 Global reach, higher impact
Global reach, higher impact