10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2018; 14(9):1122-1132. doi:10.7150/ijbs.25881 This issue Cite
Research Paper
KDM5B demethylates H3K4 to recruit XRCC1 and promote chemoresistance
1. Laboratory of Cancer Biology, Key Laboratory of Biotherapy in Zhejiang, Sir Run Run Shaw hospital, Medical School of Zhejiang University, China;
2. Department of Medical Oncology, Sir Run Run Shaw hospital, Medical School of Zhejiang University, China;
3. Department of Pathology, Sir Run Run Shaw hospital, Medical School of Zhejiang University, China;
4. Department of general surgery, Sir Run Run Shaw hospital, Medical School of Zhejiang University, China;
5. Department of Molecular Cell Biology and Toxicology, School of Public Health, Nanjing Medical University, Nanjing, China.
Abstract
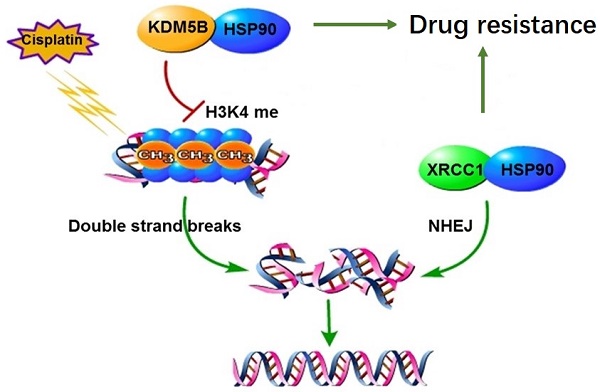
Chemotherapy is the main treatment for human cancers including gastric cancer. However, in response to chemotherapeutic drugs, tumor cells can develop drug resistance by reprogramming intracellular metabolic and epigenetic networks to maintain their intrinsic homeostasis. Previously, we have established cisplatin-resistant gastric cancer cells as a drug resistant model, and elucidated the XRCC1 as the core DNA repair mechanism of drug resistance. This study investigated the regulation of XRCC1 by lysine demethylase 5B (KDM5B) in drug resistance. We found that the methylation level of H3K4 decreased significantly in drug-resistant cells. The chemical inhibitor of H3K4 demethylases, JIB-04, restored the methylation of H3K4 and blocked the co-localization of XRCC1 and γH2AX, eventually improved drug sensitivity. We further found that the expression level of KDM5B increased significantly in drug-resistant cells. Knockdown of KDM5B increased the methylation level of H3K4 and blocked the localization of XRCC1 to the DNA damage site, leads to increased drug sensitivity. In the sensitive cells, overexpression of KDM5B suppressed H3K4 methylation levels, which resulted to resistance to cisplatin. Moreover, we found that the posttranslational modification of KDM5B is responsible for its high expression in drug-resistant cells. Through mass spectrometry screening and co-immunoprecipitation validation, we found that the molecular chaperone HSP90 forms a complex with KDM5B in drug resistance cells. Interestingly, HSP90 inhibitor 17-AAG induced KDM5B degradation in a time-and-dose-dependent manner, indicating that HSP90 protected KDM5B from protein degradation. Targeting inhibition of HSP90 and KDM5B reversed drug resistance both in vitro and in vivo. Taken together, molecular chaperon HSP90 interacted with KDM5B to protect it from ubiquitin-dependent proteasomal degradation. Increased KDM5B demethylated H3K4 and facilitated the recruitment of XRCC1 to repair damaged DNA. Therefore, inhibition of HSP90 or KDM5B represented a novel approach to reverse chemoresistance in human cancers.
Keywords: KDM5B, XRCC1, H3K4 demethylation, Chemoresistance, gastric cancer

 Global reach, higher impact
Global reach, higher impact