Impact Factor ISSN: 1449-2288
Int J Biol Sci 2023; 19(12):3640-3660. doi:10.7150/ijbs.85126 This issue Cite
Research Paper
Protection of c-Fos from autophagic degradation by PRMT1-mediated methylation fosters gastric tumorigenesis
1. Department of Integrative Biotechnology and Biomedical Institute for Convergence at SKKU (BICS), Sungkyunkwan University, Suwon 16419, Republic of Korea.
2. R&D Center, Yungjin Pharmaceutical Co, Suwon 16229, Republic of Korea.
3. Emergency Department, Hermina Hospital Tangkubanprahu, Malang 65119, Indonesia.
4. Pharmacy Program, Faculty of Science and Engineering, Universitas Ma Chung, Malang 65151, Indonesia.
5. Proteomic Analysis Team, Research Institute, National Cancer Center, Goyang 10408, Republic of Korea.
6. Division of Translational Science, Research Institute, National Cancer Center, Goyang 10408, Republic of Korea.
7. Department of Physiology and Division of Pathology, Michigan State University, East Lansing, Michigan 48824, USA.
8. Department of Molecular Cell Biology, Single Cell Network Research Center, Sungkyunkwan University School of Medicine, Suwon 16419, Republic of Korea.
9. Department of Surgery, Department of Biomedical Sciences, Ajou University School of Medicine, Suwon 16499, Republic of Korea.
*These authors contributed equally to this work.
Abstract
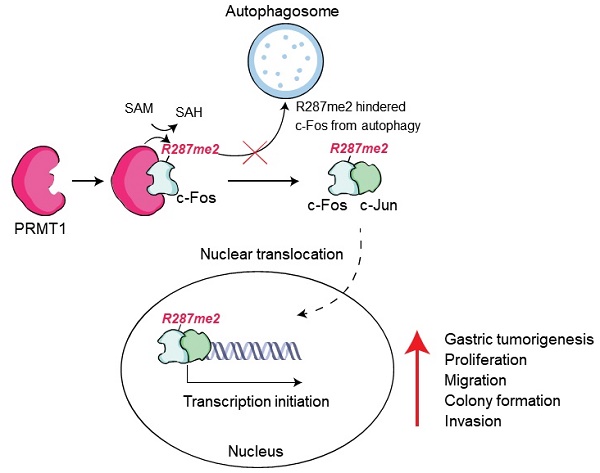
Both AP-1 and PRMT1 are vital molecules in variety of cellular progresssion, but the interaction between these proteins in the context of cellular functions is less clear. Gastric cancer (GC) is one of the pernicious diseases worldwide. An in-depth understanding of the molecular mode of action underlying gastric tumorigenesis is still elusive. In this study, we found that PRMT1 directly interacts with c-Fos and enhances AP-1 activation. PRMT1-mediated arginine methylation (mono- and dimethylation) of c-Fos synergistically enhances c-Fos-mediated AP-1 liveliness and consequently increases c-Fos protein stabilization. Consistent with this finding, PRMT1 knockdown decreases the protein level of c-Fos. We discovered that the c-Fos protein undergoes autophagic degradation and found that PRMT1-mediated methylation at R287 protects c-Fos from autophagosomal degradation and is linked to clinicopathologic variables as well as prognosis in stomach tumor. Together, our data demonstrate that PRMT1-mediated c-Fos protein stabilization promotes gastric tumorigenesis. We contend that targeting this modification could constitute a new therapeutic strategy in gastric cancer.
Keywords: autophagy, c-Fos, gastric cancer, PRMT1, protein methylation, tumorigenesis

 Global reach, higher impact
Global reach, higher impact