Impact Factor ISSN: 1449-2288
Int J Biol Sci 2010; 6(4):371-381. doi:10.7150/ijbs.6.371 This issue Cite
Research Paper
Proliferation of Myoblast Skeletal Cells on Three-Dimensional Supermacroporous Cryogels
1. Department of Biological Sciences and Bioengineering, Indian Institute of Technology Kanpur, 208016- Kanpur, India
2. Dr. MGR Educational and Research Institute, Madurayvol, Chennai- 95 and Birla Institute of Technology, Goa, India
Received 2010-4-7; Accepted 2010-6-28; Published 2010-7-3
Abstract
Cardiac and skeletal muscle tissue engineering provides a smart approach to overcome problems associated with organ transplantation and cardiac tissue and also lays a platform for superior alternative approaches in muscle regeneration. The aim of the study was to demonstrate cryogel scaffold potential in the field of skeletal muscle and cardiac tissue engineering. Poly-hydroxyethyl methacrylate (pHEMA)-gelatin cryogel scaffold was synthesized using cryogelation technique and such a designed material is being reported first time. Rheology study of the pHEMA-gelatin (HG) suggested that the cryogel scaffolds were stable at different temperatures and phase angle remained constant in both dry and wet state. HG cryogel was able to bear increased stress without leading to deformation. Monitoring the hydration of HG scaffold showed shift from a stiff to a more pliable material and upon continuing hydration, shear modulus remained constant with no further change observed. However, the change in phase angle <0.24º indicates a gradual increase in stiffness of the material over time. Scaffold synthesised using such polymer combinations gave cells a native environment for proliferation and surface stiffness have shown to help in differentiation of the cells. Myoskeletal cell lines were cultured on these scaffolds to check the biocompatibility and cell proliferation. Alamar blue assay performed over a period of 3 weeks analysed the metabolic activity of cells which showed more than 60% increase in the total cellular activity. DNA content of cells was found to be directly related to number of cells present at a given time point and this was found to have increased by more than 50% in 3 weeks. Since in 3-D scaffold the surface area is more in comparison to 2-D, hence better cell proliferation is observed. Hoechst and DAPI staining showed tubular structure and alignment of the cells during formation of the tubules shows promising cellular response to the cryogel matrix. The mechanical strength, stiffness and elastic measurements of the scaffold indicated potential application of these materials for skeletal and cardiac tissue engineering.
Keywords: Cryogel, pHEMA-gelatin, C2C12 myocardial cells, Skeletal tissue engineering, Rheology, Myotubes
Introduction
Tissue regeneration needs scaffold that balance temporary mechanical function with mass transport to aid biological delivery and tissue regeneration [1]. Development of the porous scaffold for sustainable three dimensional (3-D) growth of cells is of particular interest in the field of tissue engineering and regenerative medicine as these scaffolds are tailor-made to mimic the micro-environment or act similar to extracellular matrix which has defined role such as to provide suitable structural and chemical composition and the mechanical properties [2-6]. Due to limited ability of the cardiac cells to regenerate in-vivo, attempts are being made to restore the functionality of the heart affected from myocardial infarction by injecting myogenic cells into the affected myocardium. It has been proposed that the implantation of in-vitro cultured functional myocardial muscle could potentially enhance cell localization and efficiency of tissue repair [7, 8]. First material to be used for cardiac repair was combination of poly-lactic acid and poly-glycolic acid but these polymers and their derivatives failed to provide substantial mechanical strength to scaffold which is attributed along with elasticity as most important property while choosing materials for cardiac repair. For successful cardiac and skeletal repair a critical step involved is the creation of tailor made 3-D matrices that act host to the cells (defined as cardiac patches) and should help in maintaining cellular viability, proliferation, differentiation and support cell integration [9].
There are number of processes to obtain porous matrix with desired morphology like, salt leaching and phase emulsion being few such examples [6, 10-14]. Cell source can be primary cells, cell lines or pluripotent stem cells but the most important aspect is the cell-matrix interaction and the success of tissue implant mostly depends upon this connection. Three dimensional (3-D) porous scaffolds have to support cellular attachment, proliferation, unhindered transport of nutrients and also play a role in cell differentiation. The most common process like salt leaching and phase emulsion or hydrogel synthesis has demonstrated its validity for the preparation of scaffolds of different characteristics [15, 16]. Manufacturing of scaffold requires appropriate combination of polymers which is still considered to be a challenge for providing the desired results. There are several parameters that determine the final scaffold architecture like e.g., in electrospinning which include material concentration, solvent type, distance between collecting target from the spinning nuzzle, viscosity, the needle gauge and voltage to obtain uniform fibrous scaffolds [17]. Such complexities have lead for the development of new approaches for scaffold designing like “cryogels”. These are supermacroporous three dimensional (3-D) polymeric scaffolds synthesised at sub-zero temperature by cryogelation process [18-20]. During the process of cryogel formation the monomers or polymeric precursors are dissolved in a suitable solvent such as water and mixed with cross-linkers and are frozen instantly. In the process most of the solvent freezes and forms ice crystals. These ice crystals remain interconnected with each other whereas smaller microphase in which the liquid remains non-frozen, the solute precursors polymerize and crosslinkes to form an interconnected web like structure. After the reaction period is over and the gels are thawed at room temperature which allows melting of the ice crystals and a porous web like network is generated of the polymers leading to the formation of a versatile system called cryogels [21-23]. Depending upon the polymers used and cross linkers, the pore size of the synthesized cryogels can vary from few micrometers to about 200 µm. The pore size depends on the initial concentration of precursors in solution, their physicochemical properties and the freezing conditions [24].
Cryogels have found application in various fields such as cell chromatography, affinity chromatographic separations, cell separation, tissue engineering and bioreactors for monoclonal antibody production [25-30]. Such broad range of application is because of its supermacroporous nature with highly inter-connected pores and these can be synthesised in various format like monoliths, beads or discs depending upon the application. The high mechanical strength and ability to bear a load or stretched without collapsing is also important features of these matrices. Cryogels are becoming an important scaffold designing tool for tissue engineering applications [31-33].
Skeletal muscles are primary responsible for the voluntary movement control and also in maintenance of structural contours of the body. Mature skeletal muscle is mostly compromised of multi-nucleated, post mitotic fibres which cannot be regenerated once damaged. A very small population of myogenic progenitor satellite cells which is capable of regeneration and are composed of 1-5% of total nuclei of a mature muscle [33]. Number of attempts have been made so far to reconstruct skeletal muscle in the lab conditions [34-43], and the results have been promising suggesting it is possible to engineer bio-artificial muscles with current technology. To engineer and characterise skeletal tissue construct we have cultured myogenic skeletal cell C2C12 on p(HEMA)-gelatin (HG) cryogel after characterizing the material by rheology, the cell-matrix interaction was observed for three weeks for achieving structural and functional construct that can be suitable graft for damaged skeletal muscles.
Materials and Methods
Materials
2-Hydroxyethyl methacrylate (HEMA) (MW: 141.09), gelatin (from cold water fish skin; MW: ~60,000), poly(ethylene glycol) diacrylate (PEGDA), Dulbecco's modified Eagle's medium (DMEM), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT, 98%) reagent, Hoechst 342, 4'-6-diamidino-2-phenylindole (DAPI) and nystatin was purchased from Sigma Chemical Co. (St. Louis, MO, USA). Glutaraldehyde was purchased from s.d. fine-chemicals limited (Mumbai, India). Fetal bovine serum (FBS) and streptomycin-penicillin antibiotic solutions were bought from HyClone (Utah, USA). Cell tracker- green fluorescent probe was purchased from Lonza (Walkersville, USA) and Alexa Phallodein 568 was obtained from Invitrogen (Paisley, UK). Alamar Blue was from AbD Serotec (Martinsried/Planegg, Germany) and ds-DNA quantification kit, F02962 was from Molecular probes (Eugene, OR, USA). All other chemicals used were of analytical grade.
Designing of HG cryogel for skeletal tissue engineering
Gelatin (200 mg) was weighed and dissolved in 9 ml degassed water with final concentration of 2% (w/v). To this solution HEMA (0.8 ml) with the final concentration of 8% (v/v) was added and to ensure thorough mixing, solution was vortexed. Ammonium persulphate (APS) (0.1 ml of 10% w/v) and TEMED (0.01ml) were added as initiator of free radical polymerization. Instantly polymer solution, poly (ethylene glycol) diacrylate (PEGDA) (ratio 1:2, PEGDA: pHEMA) and glutaraldehyde (25% v/v) (ratio 1:20, glutaraldehyde: Gelatin) were added as cross linkers. Cryogelation reaction was allowed to proceed overnight at -12°C in liquid cryostat (Julabo, Seelbach, Germany). The gels were removed after incubation and using de-ionized water were thawed at room temperature. After thawing, HG cryogels were immediately washed with deionized water and were vacuum-dried at -50°C and stored at room temperature.
Rheology analysis for material characterization
The rheology analysis helps in measuring the flow of the material and deformation at given conditions [44]. Device (MCR301 SN824057, Malvern instruments, UK) was used for characterization of polymeric material as these scaffolds have their distinctive flow rate and behaviour in different temperatures. Sections (10 mm thick) of the cryogel were cut and samples were placed on sample holder fitted with cone and plate geometry with gap width of 150 µm and cone diameter of 40 mm with 4oangle and applied force of 1N per sec for 15 min. The storage modulus (G`) and loss modulus (G``) was calculated using oscillatory logarithmic sweep at a frequency of 1Hz. Substance measurement points was kept at 1000, number of data points was set to 60 and all the conditions were kept constant for rest of the material analysis. Materials may act as liquid in short run but in longer run can transform to solid state and this dual nature of polymeric materials is known as visco-elastic behaviour. The visco-elastic property of cryogels can play a very important role in the skeletal muscle tissue engineering, HG cryogels were analysed at two different temperatures of 25 ºC and 37 ºC which are room temperature and body temperature, respectively and cryogels were also checked for phase difference at dry and wet state. The dry state defines the scaffold lyophilized overnight and stored in vacuum to be completely dry and wet state of the scaffold describes saturation of scaffold with water. Dry cryogels were first tested and same sample were saturated with water for further analysis.
C2C12 culture on HG scaffold
HG cryogels were sectioned in 2 mm thickness and were sterilized stepwise by gradient alcohol concentrations (20%, 40%, 60%, 80% and 100%). Scaffolds were calibrated with PBS for 3 h followed by calibration with serum free DMEM overnight. C2C12 cells were reviewed by spinning the cells down at 200 g for 10 min and the cell pellet was re-suspended in 1 ml of DMEM media containing the fetal bovine serum. Viability and cell count was performed before seeding cells on scaffold. The cell seeding density was 1×106 cells/ml/per scaffold/per well. The culture plates were incubated at 37 ºC with 5% CO2 for over three weeks and experiment was set-up in duplicates.
Total cellular metabolic activity assay using alamar blue
Alamar blue 10% working solution was prepared by mixing 1 ml alamar blue stock solution with 9 ml HBSS (without phenol red and serum) and filtered using sterilised 0.2 µm filter and wrapped in foil until further use. The sterile test scaffolds were transferred to 24 well tissue culture plates along with 1 ml complete cell culture media. The media was discarded and 1 ml of alamar blue was added to test well and incubated at 37 oC for 90 min. Alamar Blue (100 µl) working solution (post-incubation with scaffolds) was transferred to a 96 microtitre well plate and fluorescence was measured at 570 nm (Ex 560 nm/ Em 590 nm). The seeding density and media volume was kept constant in both cryogel group and in control group. Different cryogels sections were used at each time point to prevent alamar blue saturation problem.
DNA quantification by Hoechst 33258 assay
The Hoechst 33258 working solution was prepared by adding 1 ml of TNE buffer (100mM Tris; 2.0M NaCl; 10mM EDTA; pH 7.4) along with 9 ml of distilled water and the solution was filtered. To the filtrate 10 µl of Hoechst dye was added and covered with foil until further use. The test scaffolds were transferred into vials containing 250 µl of papin and were incubated at 60 ºC overnight. The vials were allowed to cool to room temperature and the shredded tubes were used to centrifuge scaffold to ensure complete lysis of cells and to obtain clear supernatant free from any scaffold digest. Supernatant (100 µl) was mixed with 100 µl of Hoechst working solution and the fluorescence was measured using excitation and emission filters centred at 360 nm and 460 nm, respectively.
Cell differeniation
Hoechst staining was performed by dissolving Hoechst 342 stock solution in distilled water at 1 mM concentration and final working concentration of 1µM from the stock used for 15 min to stain the adherent cells. Hoechst 342 was visualized at excitation wavelength of 360 nm and emission is recorded at 480 nm. 4'6-diamidino-2-phenylindole (DAPI) is known to form fluorescent complexes with double-stranded DNA showing fluorescence specificity for AT, AU and IC clusters and because of this property DAPI is a useful tool in various cytochemical investigations. C2C12 cells cultured on cryogel scaffold were used for microscopic observation of cell-matrix interaction. Cell seeded onto sections were fixed with 2.5 % glutaraldehyde for 2 h and were sectioned using microtome (Microm HM 560 CryoStar, Thermo) followed by 20 min incubation of sections in nuclear stain DAPI (200 ng/ml working solution prepared in PBS). After the incubation samples were gently washed with PBS. Sections were selected at different region of the scaffold (upper, middle and lower portion) to check penetration of cells in HG scaffold. The morphology of the cell nuclei was observed under fluorescence microscope (Nikon, TE-2000U) at excitation wavelength of 350 nm. Nuclei with normal phenotype were expected to appear glowing bright and homogenously distributed. Scaffold samples were fixed in 4% paraformaldheyde for 10 min, and were then permeabilized with 0.1% Triton-X 100 for 5 min and after each step thorough rinsing with 0.01M PBS at room temperature was performed. The working solution of cell tracker and phallodein stains were made up in 1:200 dilution with 1% BSA and scaffolds were incubated for 15 min before imaging.
Results and Discussion
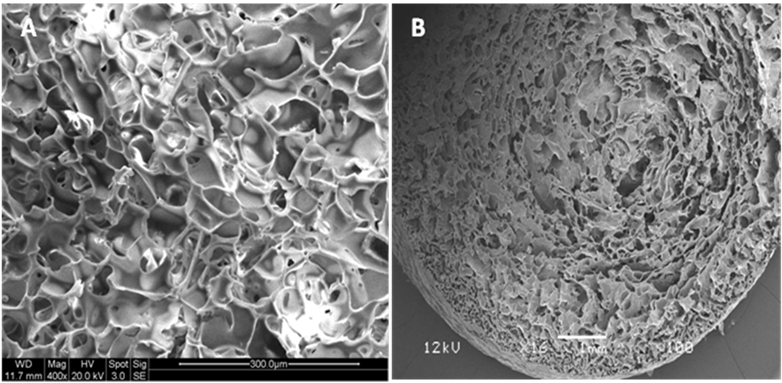
The designing of cryogel scaffold with high volume fraction porosity within soft, mechanically stable and high water absorbing capacity was obtained in HG cryogels. The cryogel synthesized were light yellow (Figure 1) in colour and have shown to retain 3-D architecture while air drying. The SEM was performed to check the pore distribution and porosity of the scaffold and the pore size of HG cryogel were found to be in the range of 30 to 120 µm, while the average pore diameter range was lying in between 50- 80 µm (Figure 2A). The overview of cryogel (Figure 2B) showed uniform pore distribution and interconnected pore network. The polymer combination of pHEMA-gelatin has been used before as an IPN using hydrogel techniques, but the combination of pHEMA-PEGda-gelatin for cryogel synthesis has not been reported before. pHEMA based slabs have been prepared and dipped in gelatin solution for supporting mouse embryonic stem cells [45], while photopolymerized PEGda-pHEMA hydrogels are used for drug delivery [46]. The interconnected pores allow convective flow of nutrients and facilitate gaseous exchange during cell culture conditions, which is considered to be a key characteristic for any tissue engineering scaffolds. These results suggest that the microstructure of HG cryogel can be a potential scaffold material for tissue engineering applications.
The digital image shows physical appearance of water saturated HG cryogel.

Scanning electron microscopy (SEM) image at magnification 400X of HG cryogel (A), showing the interconnected macroporous cryogel structure and an overview of the porous HG cryogel scaffold (B), showing even distribution of the pores.
Rheology study of HG cryogel
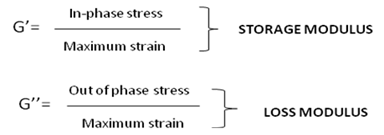
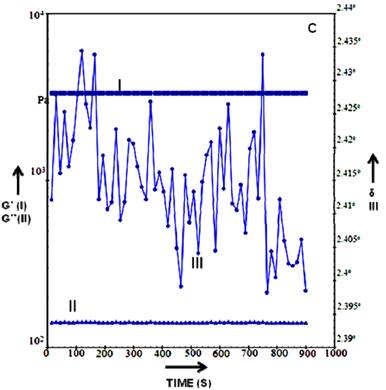
Rheology can be simply defined as a study of flow and deformation of materials. It is used in studying complex structure of substances including body fluid, suspensions, polymers etc. The flow of these materials cannot be characterized by a single value of viscosity (at a fixed temperature). More theoretically rheology is the relationship between the flow/deformation, performance of material and its internal structure that cannot be defined by classic fluid mechanics or elasticity. When material is subjected to a stress of different sorts (that is a force per area) [47], material responds in various ways. The two important ways materials can change is either its elasticity or viscosity and in-between lie the third property of the material termed as visco-elastic behaviour of the material. Depending upon the processing time of the polymer like if polymers are pumped or coated or sprayed the processing time can be different and with short processing time polymer may behave as a solid and in long processing times the material can behave as a fluid. This dual nature (fluid-solid) in rheology term is referred to as viscoelastic behaviour of materials. The solid behaviour of the material is depicted in rheology term as storage modulus (G′) since it reflects relaxation time and elastic modulus and the perfectly elastic nature will develop stress that is in-phase with strain. The liquid behaviour is represented by loss modulus (G′′) and it gives information about dissipation (viscous) of the flow. The equation to define storage and loss modulus is:
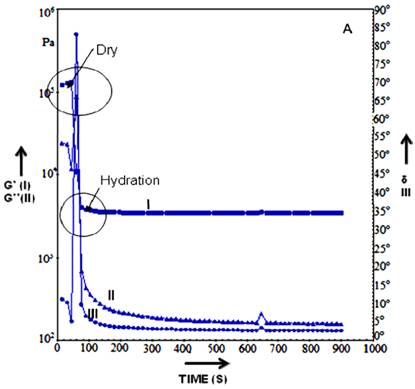
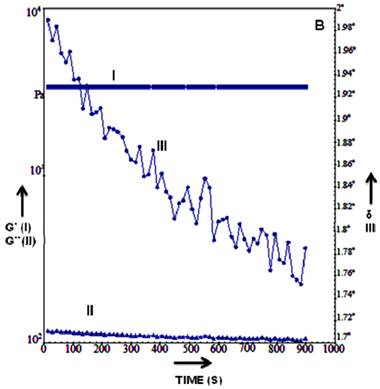
The rheological measurement (such as G', G”, elongation viscosity and viscosity) can be used as excellent tools in material characterization and input data for computational simulation etc [45]. Rheology study of the HG cryogel suggested that the scaffold were stable at different temperatures and phase angle remained constant in both dry and wet state (Figure 3A-C). HG cryogel were able to bear increased stress without leading to deformation. Upon hydration (addition of water to the dry scaffold tested previously) of HG scaffold showed the shift from a stiff to a more pliable material which is seen in graph were the dry >105 to wet <104Pa value shifts (Figure 3A). The hydration is quick as observed and due to this there is rapid change and equilibrium position is attained within 30 sec of hydration. The time gap measurement was done between additions and levelling off in G'. By continuing hydration G', G” (Shear modulus) value remain constant with no further change in modulus. However, the change in phase angle <0.24º indicates a gradual increase in stiffness of the material over the time which may be due to hydration influencing changes in bonding and loss modulus which could be resulting in possible molecular re-arrangements (Figure 3B). Upon further analysing HG cryogel shows steady state as there is no change in loss modulus and storage modulus, the scaling for the phase angle shows changes over 0.03º and from there on it is seen to be flat with no significant change in the phase angle indicating the equilibrium state of the cryogel as seen in Figure 3C. The phase angle reduction in Figure 3A shows the material exhibiting elastic characteristics. The rheology study of the HG cryogel showed the ability of the material to bear stress without leading to deformation and the mechanical strength along with elastic nature of scaffold. Therefore exploiting these material properties like stiffness of the scaffold, visco-elastic behaviour which are most important parameter for designing construct for cardiac tissue engineering as during the in-vivo conditions, the stress experienced by these tissue is probably the maximum as compared to other tissues or organs and the rheology studies of the HG scaffold illustrated the material properties that could be used in skeletal muscle regeneration.
Graph represents the rheology data of HG cryogels, the storage modulus, loss modulus and phase angle is plotted against the time at temperature 37 ºC in dry state (A) 37 ºC wet (B) and 25 ºC wet state (C). In the graph the storage modulus G' is represented by (I), the loss modulus G” is depicted by (II) and phase angle by δ (III).
Total cellular metabolic activity by alamar blue
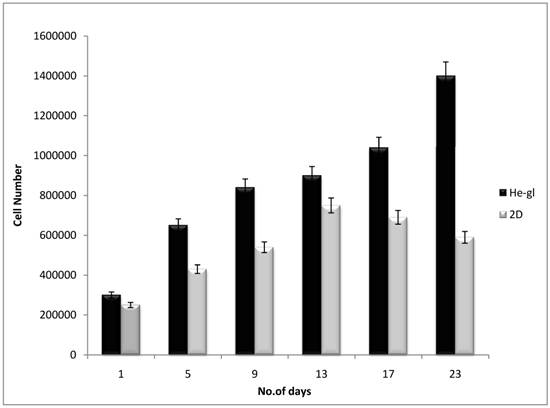
To check the potentiality of the synthesised scaffold, C2C12 cells were seeded on the HG cryogels. The total metabolic activity of the cells was checked by using alamar blue. Alamar blue works on redox system which fluoresces and changes colour in response to chemical reduction in the media resulting from the cell growth. The cellular growth has direct effect on the media provided for cell proliferation and alamar blue uses this chemical change in the media by either reduction or oxidation. The reduction which is related to cellular growth causes the redox indicator in the alamar blue to change from oxidised to reduced state. The normal colour of alamar blue changes to the fluorescent red that depends upon the cellular growth and total metabolic activity. Figure 4 shows the rate of proliferation of C2C12 cells on HG cryogel scaffold and is compared with 2-D control. The cell proliferation and growth on the scaffold showed an increasing trend with 60% increase in the total cellular activity for over 3 weeks whereas in 2-D the diminished cell growth was noticed from the day 13th of cell culture. Cell-polymer construct has been used for transplantation of myoblasts using biodegradable polymer strands to avoid transplantation buffer [48] showing importance of matrix which acts as delivery system and also supports cellular proliferation. Three dimensional engineered construct of cardiac muscle can provide a basic model system for cardiovascular research [49, 50]. Increase in the cellular viability and the total metabolic rate substantiates that these scaffolds provided good support for the cell growth and the positive effect of the cryogel matrix on the proliferation of the C2C12 cells.
The total metabolic activity of the cells was checked by alamar blue at regular time intervals. The graph describes C2C12 growth on HG cryogel (Black bar) which shows increasing trend in the metabolic activity but the 2-D culture (grey bar) starts declining from 13th day which could be due to lesser surface area available for cells after attaining confluency. The total cellular metabolic activity assay indicates the cryogels supporting the proliferation of C2C12 cells for over 3 weeks.
DNA quantification by Hoechst 33258 assay
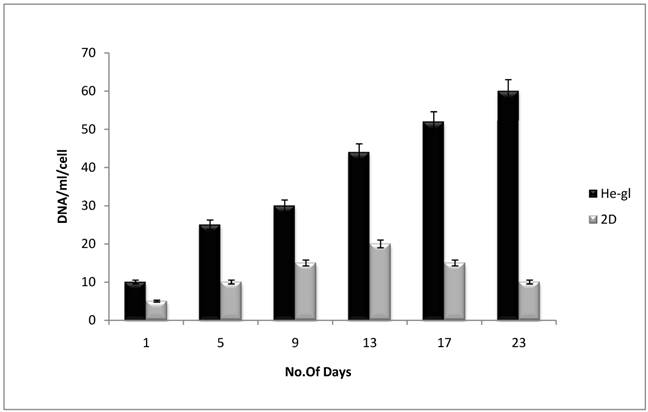
To validate the alamar blue results and to check the metabolic activity being related to cellular proliferation, DNA quantification was performed. The bis-benzimidazole derivatives of Hoechst 33258 exhibits fluorescence enhancement upon binding to A-T rich regions of double stranded DNA. The Hoechst assay results were in accordance to alamar blue. On HG scaffold the proliferation of C2C12 was observed to be better when compared to the 2-D system which results in higher DNA content being observed in C2C12 on HG scaffold (figure 5). From the 1st to 23rd day, at regular intervals the scaffolds were weighed and subjected to papin digestion. Since in 3-D scaffold surface area is larger in comparison to 2-D better cell proliferation is noted and due to increased metabolic activity of cells resulting from enhanced cell proliferation led to amplified DNA content. The increase in the DNA content shows the HG cryogels have provided the C2C12 cells with native environment that supported better cellular growth and proliferation.
The DNA quantification was done by using the Hoechst assay. The proliferative cells are analyzed for total DNA content. The standard curve was used to derive the DNA per ml of cells and further used for calculating DNA content in the test samples. Graph indicates C2C12 on the HG cryogels (Black bar) which showed improved proliferation as compared to 2-D (grey bar) which showed total DNA content increasing and these results are in accordance with the alamar blue data which shows similar trend. Since in 3-D scaffold surface area is larger in comparison to 2-D hence the better cell proliferation is noted due to increased metabolic activity of cells resulting in enhanced cell proliferation and increased DNA content.
Cell differentiation
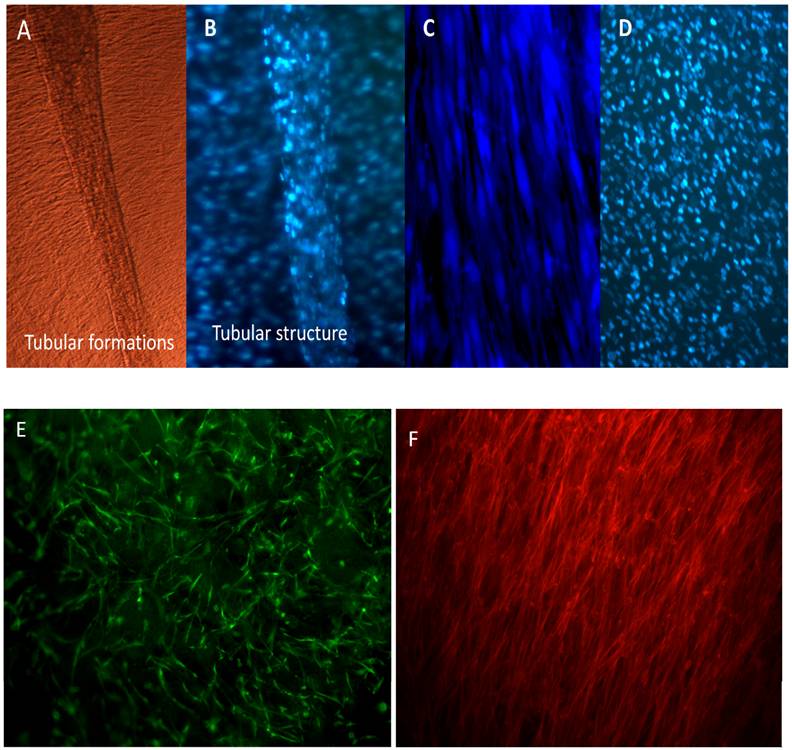
To check the distribution and differentiation of C2C12 on HG scaffold, these were observed under fluorescent microscopy. Cells were seen to adhere and proliferate throughout the scaffold and tubular structure which were elongated and multinucleated was seen to penetrate and fill the void pores of the scaffold and were found to be highly aligned along channels of the scaffold pores as visualized under phase contrast, DAPI imaging and Hoechst staining (Figure 6A-B) where 2-D cells were seen proliferating as monolayer (Figure 6C-D) with delayed tubular formation. Cell tracker was used to check the cells on the scaffold (Figure 6E) and the F-actin stained with rhodamine-phalloidin shows the myotubes aligning along the preferential directions on the HG cryogels (Figure 6F), which show 3-D growth pattern of the cells on the scaffolds. The myoblast nuclei was stained using Hoechst staining and DAPI on 7th day and14th days (Figure 7A, B). It was clearly seen that the cells have been evenly distributed within the pores of the HG cryogel. The interconnected pores of the scaffold allows unhindered transportation of media and nutrient leading to enhanced proliferation of cells on HG cryogels which can be noticed by DAPI staining showing increased population in about 2 weeks time.
C2C12 cells observed by phase contrast microscopy (A) shows formation of tubular structure from day 6th of culture. On further analysis by fluorescent microscopy using DAPI multinucleated tubular structure was seen to be formed by group of cells (B) along with background of cells spreading homogenously on the HG matrix. On 2-D culture the 6th day cells are seen after Hoechst staining (C) and DAPI staining (D). Cell tracker showing the proliferation of cells on the scaffold (E), and the alignment of the C2C12 in preferential direction on the scaffolds (F).
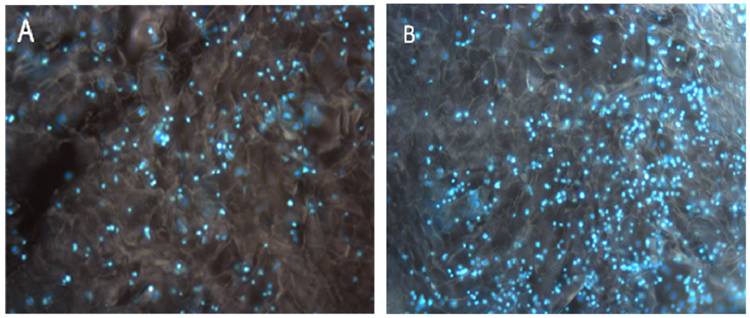
Cell attachment was visualized under fluorescent microscopy by using nuclear stain DAPI (A). The image was recorded after 48 h of cell seeding and (B) after 2 weeks. The fluorescent microscopy images show significant increase in cell number and proliferation of C2C12 on HG cryogel.
In conclusion this work investigates the potential use of supermacroporous cryogel scaffold for skeletal muscle tissue engineering, and on the basis of these findings we believe there is suitable utility of cryogel scaffold for this application. For specific application scaffold was characterized mainly for degradation, elasticity and mechanical property and also cell attachment and proliferation was checked by measuring total metabolic activity of C2C12 on HG cryogels. We describe a simple inexpensive technique of designing scaffold for complex tissue engineering and conclude that the cryogel have shown promising cellular response by enhanced attachment and proliferation and can be potential scaffold for skeletal or cardiac tissue engineering. We aim to further tune the scaffold and characterize the engineered cardiac muscle construct by gene-level and cell-level electrophysiological studies. The ultimate aim will be to develop neo-cardiac tissue in the three-dimensional architecture.
Acknowledgements
Authors would like to duly acknowledge Department of Biotechnology (DBT), Ministry of Science and Technology, Govt. of India and Indo-UK (DST-UKIERI) award project for funding the research. We thank Lloyd Hamilton for helping with rheology analysis.
Conflict of Interest
The authors have declared that no conflict of interest exists.
References
1. Hollister JS. Porous scaffold design for tissue engineering. Nature Materials. 2005;4:518-24
2. Harrision BS, Atala A. Carbon nanotube applications for tissue engineering. Biomaterials. 2007;28:344-64
3. Shea LD, Smiley E, Bonadio J, Mooney DJ. DNA delivery from polymer matrices for tissue engineering. Nat Biotechonol. 1999;17:551-54
4. Wang F, Weaver VM, Peterson OW, Larabell CA, Dedhar S, Briand P, Lupu R, Bissell MJ. Reciprocal interactions between beta1-integrin and epidermal growth factor receptor in three-dimensional basement membrane breast cultures: a different perspective in epithelial biology. Proc Natl Acad Sci USA. 1998;95:14821-6
5. Lutolf MP, Raeber GP, Zisch AH, Tirelli N, Hubbell JA. Cell-responsive synthetic hydrogels. Adv Mater. 2003;15:888-92
6. Partap S, Rehman I, Jones JR, Darr JA. Supercritical carbon-dioxide in water emulsion-templated synthesis of porous calcium alginate hydrogels. Adv Mater. 2006;18:501-04
7. Bursac N, Papadaki M, Cohen RJ, Schoen FJ, Eisenberg SR, Carrier RL, Vunjak-Novakovic G, Freed LE. Cardiac muscle tissue engineering: toward an in vitro model for electrophysiological studies. Am J Physiol Heart Circ Physiol. 1999;277:H433-44
8. Carrier RL, Papadaki M Rupnick M, Schoen FJ Bursac N, Langer R Freed LE, Vunjak-Novakovic G. Cardiac tissue engineering: cell seeding, cultivation parameters, and tissue construct characterization. Biotechnol Bioeng. 1999;64:580-89
9. Zammaretti P, Jaconi M. Cardiac tissue engineering: regeneration of the wounded heart. Current Opinion in Biotechnology. 2004;15:430-34
10. Mann S. The chemistry of form. Angew Chem Int Ed. 2000;39(19):3392-06
11. Sanchez C, Arribart H, Giraud-Guille MM. Biomimetics and bioinspiration as tools for design of innovative materials and systems. Nat Mater. 2005;4(4):277-88
12. Kotov NA, Liu YF, Wang SP, Cumming C, Eghtedari M, Vargas G, Motamedi M, Nichols J, Cortiella J. Inverted colloidal crystals as three-dimensional cell scaffolds. Langmuir. 2004;20(19):7887-92
13. Stachowaik AN, Bershteyn A, Tzatzalos E, Irvine DJ. Bioactive hydrogels with an ordered cellular structure combine interconnected macroporosity and robust mechanical properties. Adv Mater. 2004;17(4):399-03
14. Barbetta A, Dentini M, DeVecchis MS, Filippini P, Formisano G, Chiazza S. Scaffolds based on biopolymeric foams. Adv Funct Mater. 2005;15(1):118-24
15. Drury JL, Mooney DJ. Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials. 2003;24(24):4337-51
16. Wan ACA, Tai BCU, Leck KJ, Ying JY. Silica-incorporated polyelectrolyte-complex fibres as tissue- engineering scaffolds. Adv Mater. 2006;18(5):641-44
17. Hagvall S, Layland KS, Dhanasopon AP, Rofail F, Smith H, Wu BM, Shemin R. Three-dimensional electrospun ECM-based hybrid scaffolds for cardiovascular tissue engineering. Biomaterials. 2008;29(19):2907-14
18. Ahlqvist J, Kumar A, Sundström H, Ledung E, Hörnsten HG, Enfors SO, Mattiasson Bo. Affinity binding of inclusion bodies on supermacroporous monolithic cryogels using labelling with specific antibodies. J Biotechnology. 2006;122(2):216-25
19. Dainiak MB, Galaev IYu, Kumar A, Fatima P, Mattiasson Bo. Integrated isolation of antibody fragments from microbial cell culture fluids using supermacroporous cryogels. J Chromatography A. 2004;1045:93-98
20. Dainiak MB, Kumar A, Galaev IYu, Mattiasson Bo. Detachment of affinity captured bioparticles by elastic deformation of macroporous hydrogel. Proc. Nat. Acad. Sci USA. 2006;103:849-54
21. Jain E, Kumar A. Synthesis and characterization of polyacrylonitrile and interpenetrating network of polyacrylamide and chitosan supermacroporous cryogels as scaffolds for cell immobilization. J Biomaterial Science, Polymer Ed. 2009;20:877-02
22. Kumar A, Bhardwaj A. Methods in Cell Separation for Biomedical Application: Cryogels as a New Tool. Biomedical Materials. 2008;3(034008):11pp
23. Kumar A, Bansal V, Andersson J, Roychoudhury PK, Mattiasson Bo. Supermacroporous cryogel matrix for integrated protein isolation: IMAC purification of urokinase from cell culture broth of a human kidney cell line. J Chromatography A. 2006;1103:35-42
24. Kumar A, Bansal V, Nandakumar KS, Galaev IYu, Roychoudhury PK, Holmdahl R, Mattiasson Bo. Integrated Bioprocess for the production and isolation of urokinase from animal cell culture using supermacroporous cryogels. Biotechnology and Bioengineering. 2006;93(4):636-46
25. Kumar A, Caballero AR, Fatima P, Galaev IYu, Nandakumar KS, Kamihira M, Holmdahl R, Orfao T, Mattiasson Bo. Affinity binding of cells to cryogel adsorbents with immobilized specific ligands: Effect of ligand coupling and matrix architecture. J Molecular Recognition. 2005;18:84-93
26. Kumar A, Fatima P, Galaev IYu, Mattiasson Bo. Affinity fractionation of lymphocytes using a monolithic cryogel. J Immunol. Methods. 2003;283:185-94
27. Lozinsky VI, Galaev IYu, Plieva FM, Savina IN, Jungvid H, Mattiasson B. Polymeric cryogels as promising materials of biotechnological interest. Trends Biotechnol. 2003;21:445-51
28. Mattiasson Bo, Galaev IYu, Kumar A, Dainiak MB. Process for adsorption- based separation of bioparticles from an aqueous suspension. PCT/SE. 2006: 000556.
29. Nilsang S, Nandakumar KS, Galaev IYu, Rakshit SK, Holmdahl R, Mattiasson Bo, Kumar A. Monoclonal antibody production using a new supermacroporous cryogel perfusion reactor. Biotechnol. Prog. 2008;23:932-39
30. Nilsang S, Nehru V, Plieva FM, Nandakumar K.S, Rakshit SK, Holmdahl R. Three-dimensional culture for monoclonal antibody production by hybridoma cells immobilized in macroporous gel particles. Biotechnol Prog. 2008;24:1122-31
31. Srivastava A, Jain E, Kumar A. The physical characterization of supermacroporous poly (N-isopropylacrylamide) cryogel: mechanical strength and swelling/de-swelling kinetics. Materials Science & Engineering A. 2007;464:93-100
32. Tripathi A, Kathuria N, Kumar A. Elastic and macroporous agarose-gelatin cryogels with isotropic and anistropic porosity for tissue engineering. J Biomed mater Res A. 2009;90(3):680-94
33. Alameddine HS, Dehaupas M, Fardeau M. Regeneration of skeletal muscle fibres from autologus satalite cells multiplied in vitro. An experimental model for testing cultured cell myogenicity. Muscle Nerve. 1989;12(7):544-55
34. Dennis RG, Kosnik PE. Excitability and isometric contractile properties of mammalian skeletal muscle constructs engineered invitro. Invitro Cell Dev Biol Anim. 2000;36(5):327-35
35. Dennis RG, Kosnik PE, Gilbert ME, Faulkner JA. Excitability and contractile properties of mammalian skeletal muscle engineered from primary cultures and cell lines. Am J Physio Cell Physiol. 2001;280:C288-95
36. Kosnik PE, Faulkner JA, Dennis RG. Functional development of engineered skeletal muscle from adult and neonatal rats. Tissue Eng. 2001;7(5):573-84
37. Powell CA, Smiley BL, Mills J, Vandenburgh HH. Mechanical stimulation improves tissue-engineered human skeletal muscle. Am J Physiol Cell Physiol. 2002;283(5):C1557-65
38. Okano T, Satoh S, Oka T, Matsuda T. Tissue engineering of skeletal muscle: highly dense, highly oriented hybrid muscular tissue biomimicking native tissues. ASAIO J. 1997;43(5):M749-53
39. Cronin E, Thurmond F, Bassel DR, Williams RS. Cardio-myocytes grow and establish a differentiated state on poly glycolic acid fibres. Biomedical Engineering society annual fall meeting. 2001
40. Saxena AK, Marler J, Benvenuto M, Willital GH, Vacanti JP. Skeletal muscle tissue engineering using isolated myoblasts on synthetic biodegradable polymers: preliminary studies. Tissue Eng. 1999;5(6):525-32
41. Saxena AK, Willital GH, Vacanti JP. Vascularized three dimensional skeletal muscle tissue-engineering. Biomed Mater Eng. 2001;11(4):275-81
42. Acarturk TO, Peel MM, Petrosko P, LaFramboise W, Johnson PC, DiMilla PA. Control of attachment, morphology, and proliferation of skeletal myoblasts on silanized glass. J Biomed Mater Res. 1999;44(4):355-70
43. Mulder MM, Hitchcock RW, Tresco PA. Skeletal myogenesis on elastomeric substrates: implications of tissue engineering. J Biomater Sci: Polym Edition. 1998;9(7):731-48
44. Vlachopoulos J, Wagner JR. The SPE Guide on Extrusion Technology and Troubleshooting. Brookfield CT: Society of Plastics Engineers. 2001
45. Hora D, Kroupova J. Poly (2-hydroxyethyl methacrylate)-based slabs as a mouse embryonic stem cell support. Biomaterials. 2004;25(22):5249-60
46. Ayhan F, Ozkan S. Gentamicin Release from photopolymerized PEG Diacrylate and PHEMA hydrogel discs and their in-vitro antimicrobial activities. Drug Delivery. 2007;14(7):433- 39
47. Macosko CW. Rheology: Principles, Measurements and Applications. New York: VCH Publishers. 1994
48. Saxena AK, Willital GH, Vacanti JP. Vascularized three-dimensional skeletal muscle tissue-engineering. Biomed Mater Eng. 2001;11(4):275-81
49. Eschenhagen T, Fink C Remmers U, Scholz H Wattchow J, Weil J Zimmermann W. Three-dimensional reconstitution of embryonic cardiomyocytes in a collagen matrix: a new heart muscle model system. FASEB J. 1997;11:683-94
50. Folkman J, Haudenschild C. Self-regulation of growth in three dimensions. J Exp Med. 1973;138:745-53
Author contact
Corresponding author: Tel.: +91-512-2594051, Fax: +91-512-2594010; Email: ashokkumac.in

 Global reach, higher impact
Global reach, higher impact