Impact Factor ISSN: 1449-2288
Int J Biol Sci 2015; 11(3):295-303. doi:10.7150/ijbs.10586 This issue Cite
Research Paper
Hydroferrate Fluid, MRN-100, Provides Protection against Chemical-Induced Gastric and Esophageal Cancer in Wistar Rats
1. Department of Otolaryngology, Charles Drew University of Medicine and Science, 1731 E. 120th Street, Los Angeles, CA 90059, USA.
2. Department of Zoology, Faculty of Science, University of Mansoura, Mansoura 35516, Egypt.
3. Drug and Radiation Research Department, National Center for Radiation and Research Technology, P.O. Box 29 Nasr City, Cairo, Egypt.
4. Department of Internal Medicine, Charles Drew University of Medicine and Science, 1731 E. 120th Street, Los Angeles, CA 90059, USA.
5. Department of Pathology, Charles Drew University of Medicine and Science, 1731 E. 120th Street, Los Angeles, CA 90059, USA.
Received 2014-9-19; Accepted 2014-12-9; Published 2015-1-28
Abstract

In the current study, we examined the protective effect of hydroferrate fluid MRN-100 against the carcinogen methylnitronitrosoguanidine (MNNG)-induced gastric and esophageal cancer in rats. MRN-100 is an iron-based compound composed of bivalent and trivalent ferrates. At 33 weeks post treatment with MNNG, rats were killed and examined for the histopathology of esophagus and stomach; liver, spleen, and total body weight; and antioxidant levels in the blood and stomach tissues. Results showed that 17/20 (85%) gastroesophageal tissues from carcinogen MNNG-treated rats developed dysplasia and cancer, as compared to 8/20 (40%) rats treated with MNNG plus MRN-100. In addition, MRN-100 exerted an antioxidant effect in both the blood and stomach tissues by increasing levels of GSH, antioxidant enzymes SOD, CAT, and GPx, and total antioxidant capacity (TAC) level. This was accompanied by a reduction in the total free-radical and malondialdehyde levels. Furthermore, MRN-100 protected against body and organ weight loss. Thus, MRN-100 exhibited significant cancer chemopreventive activity by protecting tissues against oxidative damage in rats, which may suggest its effectiveness as an adjuvant for the treatment of gastric/esophageal carcinoma.
Keywords: hydroferrate, gastric cancer, dysplasia, anti-oxidant
Introduction
Gastric and esophageal cancers are two leading causes of cancer-related deaths throughout the world (1). In the United States, it was estimated that approximately 40,000 people would be diagnosed with esophageal and stomach cancer in 2014, and despite advancement in treatment options, the 5-year survival rates for these cancer patients remain low: 17% and 27%, respectively (1). Both cancers are thought to arise from chronic inflammation caused by Helicobacter pylori (H. pylori) (2) or gastroesophageal reflux disease (GERD). Inflammation associated with esophageal cancer is believed to be induced by GERD (3). An estimated 28% of the United States adult population suffers from GERD-like symptoms (4). This inflammation leads to atrophy and transformation, or metaplasia, of epithelial cells in the lining of the digestive tract, which will cause dysplasia and subsequently cancerous lesions (2).
The most effective treatment for gastric/eshophageal cancers is surgical removal of the cancerous lesions; however, this treatment is palliative for many advanced stages and does not address the causative chronic inflammation which could lead to the development of new lesions (5). Several potential preventative therapies have been examined for the treatment of gastric and esophageal cancers: chemoprevention, anti-inflammatory agents, and eradication of H. pylori. However, there is still a lack of evidence that these approaches will be effective in humans due to an insufficient number of clinical trials (6); novel preventative agents for treatment of esophageal/gastric cancers remain in high demand.
Hydroferrate fluid, MRN-100, is an iron-based compound composed of bivalent and trivalent ferrates isolated from phytosin. Previous research on MRN-100 has shown its potential as a protector against age-associated oxidative stress (7), γ-radiation (8), and HIV activity (9). The current study was a preliminary investigation of whether MRN-100 has the ability to restrict esophageal/gastric cancer in rats. Results show that MRN-100 decreases the extent of esophageal/gastric dysplasia and carcinoma by a mechanism that involves protection against oxidative stress damage to tissues.
Materials and Methods
N- methyl-N-nitro-N- nitrosoguanidine (MNNG)
The carcinogen MNNG (Sigma-Chemical, St. Louis, MO) was used at a concentration of 200mg/kg body weight, and was orally administered to the rats daily for 2 weeks.
Hydroferrate fluid (MRN-100)
MRN-100 was prepared in distilled water (DW) with the concentration of Fe2+ and Fe3+ ions at about 2 × 10-12 mol/l. MRN-100 was obtained from a plant extract called phytosin. It contains iron and neutral lipid compounds and can be found in plants such as radish seeds, rice, and wheat. The extraction method of MRN-100 is as follows: Phytosin (1 unit) was dissolved in 100mL DW, and then FeCl3•6H2O was added. Subsequently, a liquid-liquid extraction technique was used to remove lipid compounds. This was followed by filtration of the remaining liquid using No. 5 filter paper. The filtrate was then evaporated and condensed in a water bath. In order to generate MRN-100, the iron compound obtained was subjected to fractional determination with respect to bivalent ferrate and trivalent ferrate. Hydroxylamine-HCl (10%) was added to the sample liquid to reduce Fe (III) to Fe (II). The o-phenanthrolin method was used to determine the quantity of Fe (II). Subsequently, all of the ferrate quantities were determined, as well as those of Fe (III). Finally, the obtained iron compounds were bivalent and trivalent ferrates (8). MRN-100 was provided by ACM Co., Ltd, Japan.
Animals
In the current study, we used male Wistar rats (4-months old, body weight~120 g). Rats were obtained from the Research Institute of Ophthalmology (Giza, Egypt), and were acclimated for one week before the start of the experiments. Rats were individually housed with light and temperature controls (20±2˚C) and were fed standard laboratory cube pellets (Misr Oil & Soap Company (Cairo, Egypt). The pellets consist of wheat flour (80%), bran (3.3%), casein (12.5%), olive oil (2.3%), fats (1.0%), vitamins and salt mixture (0.2%), dl-methionine (0.5%) and water (0.2%). The approximate ratio of total calories is protein (18%), carbohydrate (73%), and fat (9%). Animal protocols were in compliance with the Guide for the Care and Use of Laboratory Animals at the University of Mansoura, Egypt.
Experimental design
40 rats were randomly divided into 4 groups: Control (untreated with carcinogen or MRN-100); MRN-100 treated (MRN-100-treated only), MNNG treated (carcinogen-treated only), and MNNG plus MRN-100 treated (MRN-100 and carcinogen-treated). In order to induce gastric/esophageal cancer, rats were given carcinogen MNNG at dose 200 mg/kg body weight once daily by oral gavage for 2 weeks, followed by oral administration of NaCl (1ml/rat) once every 3 days for 4 weeks. Concomitantly with chemical induction, the rats were given MRN-100-free water (groups 1 and 3) or MRN-100 water (groups 2 and 4) for a total of 33 weeks. All animals were weighed at different time intervals. At the end of experimental period (33 weeks), animals were killed and examined for the following: histopathological changes in the esophageal and gastric tissues, changes in the weight of livers and spleens, and redox status in the blood and stomach tissues.
Sample collection and esophageal/gastric tissue preparation
After 33 weeks, animals were allowed to fast and then were killed by cervical dislocation. Blood samples were collected by puncturing the orbital venous plexus using heparinized capillary glass tubes. Blood was used for measurement of total free radicals. Hemolysates were used for measurement of the levels of the following parameters: malondialdehyde-MDA, Glutathione (GSH), and endogenous antioxidant enzymes including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx). In addition, plasma was used to determine total antioxidant capacity (TAC) level.
Regarding the gastric redox biomarkers and histopathology examination, the stomach was excised and divided into 2 parts symmetrically along the greater and lesser curves. Part 1 was used to evaluate the redox biomarkers and was washed and homogenized in ice-cold phosphate buffer (0.1 mol/l, pH 7.4) using a Potter-Elvehjem homogenizer to give a 10% w/v homogenate. Part 2 was used for histopathology examination and was fixed along with esophageal tissues in 10% formaldehyde.
Analytical procedures
Lipid peroxidation (LPx) level, GSH content, and SOD, CAT, and GPx activities were examined in erythrocytes and gastric tissues. LPx level was ascertained by the formation of MDA and measured as in (10), GSH content as in (11), SOD activity as in (12), CAT activity as in (13), GPx activity as in (14), TAC level in plasma and gastric tissue was measured using Randox total antioxidant status kit (UK) according to (15), and gastric protein levels as in (16).
Detection of blood total free radicals by Electron Spin Resonance (ESR)
The method previously described by Heckly in 1979 was followed to detect the levels of blood total free radicals (17). Samples were processed and measured as previously described (7, 17).
Analysis of ESR data
Earlier methods of ESR analysis were used as described (18). Intensities were measured as the distance between top and bottom points of the first derivative for monitoring variations in the peak height of ESR signals as a function of the magnetic field. Quantitative assessments of free-radical concentrations were made according to the following equation,
Nd=K[Ho(ΔH2) A/2]/[Hm×Ge √ PH ]
where Nd = number of radicals, K = 103/cm, Ho = peak magnetic field in gauss, ΔH = peak-to-peak width, Hm = modulation field, PH = 1.008mW, Ge = detector gain = 3.17×105, concentration = unpaired electrons/lyophilized blood (g) or spin/lyophilized blood (g), and A = peak height of signals/weight.
Histopathological analysis
The gastric and esophogeal tissues were examined for histopathological changes at 33 weeks post exposure to MNNG. One slide containing two tissue sections from each of the 36 rats were prepared (Table 1). Tissues were fixed in 10% formalin solution and fixed overnight in cassettes. Each tissue section measured 1.5 x 0.3 x 0.1 cm in average. The paraffin-embedded tissues were sectioned on a microtome to 4 μm. The tissue sections were stained with hematoxylin-eosin (H&E) and examined by light microscopy to check for dysplasia and carcinoma. In addition, the cancer incidence and cancerous lesions were calculated as percentage of rats per group.
Statistical analysis
Body-weight values were reported as mean ± SD, while other values were reported as mean ± SE, and significance of the differences between mean values was determined by one-way analysis of variance (ANOVA) coupled with the Newman-Keuls multiple comparison test. Different pathological lesions were evaluated by Fisher's exact test or Chi-square test whenever appropriate. P < 0.05 was considered statistically significant.
Results
Percentage of dysplasia and cancer
36 H&E-stained slides from rats under 4 different treatment conditions were examined under light microscopy to check for dysplasia and carcinoma (Table 1). No rats from the control or MRN-100-treated group developed dysplasia or carcinoma. On the other hand, rats from the carcinogen-treated group and the carcinogen plus MRN-100 group developed dysplasia and carcinoma. Rats treated with only carcinogen showed 90% (9/10 rats) development of either single or multiple (≥2) foci. In contrast, of rats treated with carcinogen plus MRN-100 only 40% (4/10) carried foci. Moreover, only 10% (1/10) of MRN-100-treated rats developed multiple foci as compared to 20% (2/10) in the carcinogen group (Table 2).
Histopathological slide details.
| GROUPS | CONTROL | MRN-100 | CARCINOGEN | CARCINOGEN + MRN-100 |
|---|---|---|---|---|
| NO. OF RATS | 7 | 9 | 10 | 10 |
| NO. OF SLIDES (ONE PER RAT) | 7 | 9 | 10 | 10 |
| TOTAL NO. OF TISSUE SECTIONS | 14 | 18 | 20 | 20 |
Esphogeal tissue
In the carcinogen-treated group, 9 of 10 rats (90%) developed squamous dysplasia of variable degree (mild to severe) and extent (involving long segments of epithelium). 1 of 10 rats (10%) developed squamous cell carcinoma involving a longer segment of esophagus. However, in the carcinogen plus MRN-100-treated group, a fewer number of rats, 4 of 10 (40%) developed squamous dysplasia, and the dysplasia was of lesser degree and extent than those treated with the carcinogen alone. Although 1 of 10 rats (10%) also developed squamous cell carcinoma, the extent of involvement was lesser than in the carcinogen group (Table 2).
Percentage of rats displaying squamous dysplasia or squamous cell carcinoma foci in the esophageal tissue
| Experimental Groups | Number of Foci Per Rat | % of Rats with Dysplasia/ Carcinoma | ||
|---|---|---|---|---|
| Mild Dysplasia | Severe Dysplasia | Squamous Cell Carcinoma | ||
| Control Group | 0 | 0 | 0 | 0% |
| MRN-100 Group | 0 | 0 | 0 | 0% |
| Carcinogen Group | ||||
| #1 Rat | 0 | 0 | 0 | |
| #3 Rat | 10 | 2 | 3 | 90% |
| #4 Rat | 8 | 5 | 0 | |
| #2,5,6,7,8,9,10 Rats | 1 | 0 | 0 | |
| Carcinogen Plus MRN-100 Group | ||||
| #5 Rat | 2 | 6 | 1 | 40% |
| #1,2,4 Rats | 1 | 0 | 0 | |
| #3,6,7,8,9,10 Rats | 0 | 0 | 0 | |
Gastric tissue
In the carcinogen-treated group, 2 of 10 rats (20%) developed glandular dysplasia and adenocarcinoma. In addition, mucous-gland hyperplasia was observed in 6 of 10 rats (60%). Mucous-gland hyperplasia is a benign physiologic change seen in chronic gastritis. In contrast, in the carcinogen plus MRN-100-treated group, mucous-gland hyperplasia, glandular dysplasia, and adenocarcinoma were not observed (Table 3).
Percentage of rats displaying glandular dysplasia and adenocarcinoma foci in the gastric tissue
| Experimental Groups | Number of Foci Per Rat | % of Rats with Dysplasia/Carcinoma | |
|---|---|---|---|
| Glandular Dysplasia | Adenocarcinoma | ||
| Control Group | 0 | 0 | 0% |
| MRN-100 Group | 0 | 0 | 0% |
| Carcinogen Group #3 Rat #4 Rat #1,2,5,6,7,8,9,10 Rats | 1 1 0 | 1 1 0 | 20% |
| Carcinogen Plus MRN-100 Group | 0 | 0 | 0% |
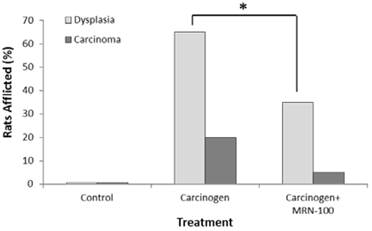
Figure 1 summarizes the results of histopathological examination of treatments with carcinogen and carcinogen plus MRN-100. Carcinogen-treated rats showed that 17/20 (85%) of the gastric and esophageal (foregut) tissues developed dysplasia or cancer: 13/20 (65%) showed dysplasia and 4/20 (20%) developed cancer. Conversely, rats treated with carcinogen in the presence of MRN-100 showed significantly (p<0.01) lower incidence of dysplasia (7/20 (35%)) and cancer (1/20 (5%))(Fig 1 & Table 2).
Percentage of rats with dysplasia or cancer post administration of carcinogen MNNG and MRN-100. Rats were treated with MNNG alone or MNNG plus MRN-100 and the percentages of dysplasia and carcinoma were examined at 33 weeks post treatment. No squamous dysplasia or carcinoma were detected in the control rats. Each group contains 9-10 rats. *p<0.01 compared to MNNG plus MRN-100.
Histopathology examination of esophageal tissues
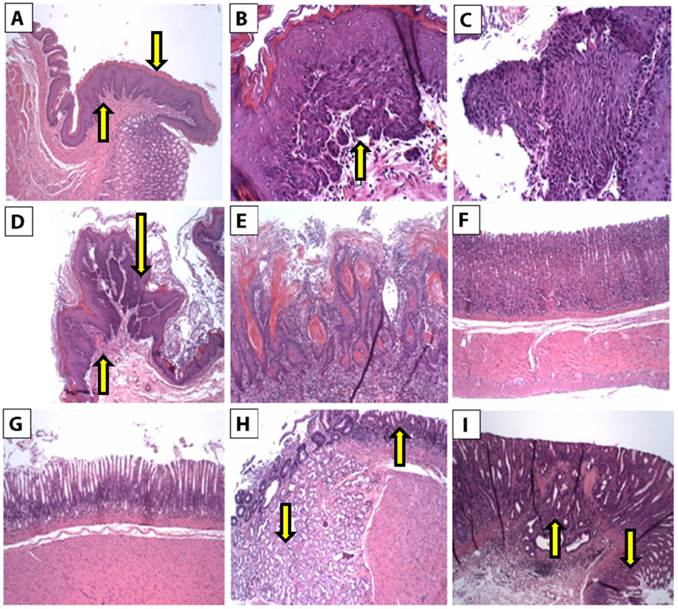
Histopathological changes of H&E-stained tissues of the esophageal mucosa were examined. Squamous epithelium of all control untreated rats showed esophageal mucosa with hyperkeratosis and squamous hyperplasia (Figure 2A). The squamous epithelium from all rats treated with carcinogen showed hyperkeratosis and patchy areas of mild squamous dysplasia (Figure 2B) and severe squamous dysplasia (Figure 2C). In addition, well-differentiated keratinizing squamous cell carcinoma was detected (Figures 2D & E).
Histopathology examination of gastric tissues
The gastric mucosa from the body and the antrum of all control untreated rats was within normal limits (Figures 2F & G, respectively). Squamous hyperplasia, dysplasia, or carcinoma was not observed in the control tissues. In contrast, gastric mucosa from carcinogen-treated rats showed hyperplastic mucinous glands and mild squamous dysplasia (Figure 2H). In addition, invasive adenocarcinoma was detected (Figure 2I). Conversely, the tissues from carcinogen plus MRN-100-treated rats showed patchy and small areas of mild squamous dysplasia in only 7/20 tissues. Thus, it appeared that MRN-100 decreased the extent of esophageal dysplasia and squamous cell carcinoma. Similar findings were also noted for gastric dysplasia and adenocarcinoma.
H&E histopathology staining from esophageal and gastric tissues. The foci of dysplasia and carcinoma are patchy and involved 1 to 2 mm of the tissue sections. (A) Section of a control rat's esophageal tissue, there is hyperkeratosis (down arrow) and squamous hyperplasia (up arrow) (2X). (B-E) Sections from esophageal mucosa from carcinogen-treated rats. (B) Section of esophagus showing a focus of mild squamous dysplasia (up arrow) (10X). (C) Section of esophagus showing severe squamous dysplasia (10X). (D) Section of esophagus showing a focus of mild, moderate, and severe squamous dysplasia (down arrow) and a focus of squamous cell carcinoma (up arrow) (4X). (E) Section of esophagus showing invasive well-differentiated keratinizing squamous cell carcinoma (10X). (F) Section of the stomach body of a control rat (4X). (G) Section of the antrum of the stomach of a control rat (4X). (H,I) Sections from gastric tissues of carcinogen-treated rats. (H) Section of antrum area showing mild dysplasia of glands (up arrow) and hyperplastic mucinous glands (down arrow) (4X). (I) Section of the body showing high-grade glandular dysplasia (down arrow) and invasive adenocarcinoma (up arrow) (4X).
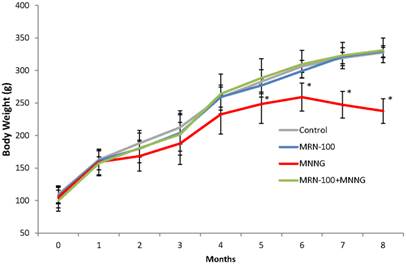
Changes in body weight under different treatment conditions. Rats were given carcinogen MNNG in the presence or absence of MRN-100. Animals in the 4 groups were examined for the changes in their body weight every month for 33 weeks. *p<0.01 compared to control and MRN-100 plus MNNG group. Each bar represents the mean ± SD of 10 rats/group.
Body weight changes
Figure 3 shows changes in body weight. Treatment with carcinogen alone resulted in early weight loss that was detected at 2 months and became significant at 5 months (p<0.01). In contrast, rats treated with carcinogen plus MRN-100 did not experience loss in body weight due to carcinogen treatment.
Organ weight changes
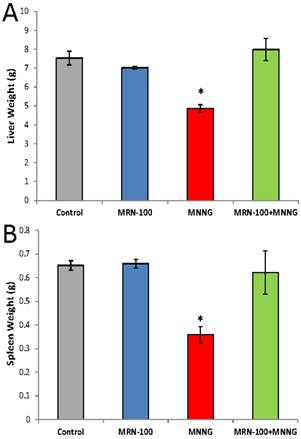
Results of changes in the weight of the livers and spleens at 33 weeks are shown in Figure 4. Rats bearing tumors had a significant decrease in the weight of liver (35%) and spleen (45%) as compared to the control group. While the liver-weight-loss proportion was approximately equal to the body-weight-loss proportion, the spleen showed a significantly larger decrease in weight. In contrast, rats with MRN-100 showed organ weight similar to control rats.
Antioxidant Effect
Antioxidant effects were examined in the blood. The levels of MDA, GSH, and antioxidant enzymes were determined in RBCs, while the level of TAC was measured in plasma. These parameters were also examined in the gastric tissues.
MDA level
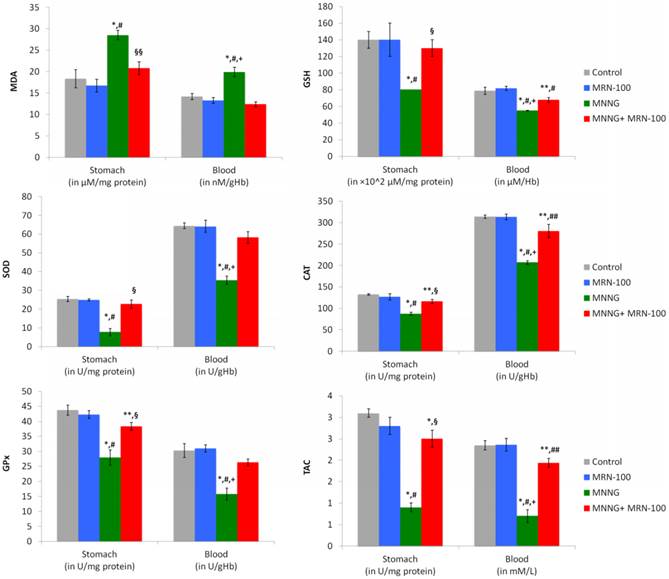
Data in Figure 5 shows that the carcinogen-treated group displayed a remarkable increase in the levels of blood and gastric MDA by 40.8% and 55.8%, respectively (p<0.01), as compared to control rats. In contrast, treatment with MRN-100 provided protection against MNNG-induced elevation of MDA values in both tissues.
Levels of GSH
Data show that rats treated with MNNG displayed a significant decrease in the levels of blood GSH (-30.1%) and gastric GSH (-39.9%) (p<0.01) when compared with control group. On the other hand, the decrease in GSH content in these tissues was nearly prevented post treatment with MRN-100 (Figure 5).
Changes in the liver and spleen weight. Rats were given carcinogen MNNG in the presence or absence of MRN-100 for 33 weeks and were examined for changes in the weight of livers (A) and spleens (B). *p<0.01 compared to control and other groups. Each bar represents the mean ± SE of 10 rats/group.
Effect of carcinogen MNNG alone and MNNG+MRN-100 treatments for 33 weeks on stomach and blood MDA, GSH, SOD, CAT, GSH-Px, and TAC. Each value represents the mean ± SE of 6 rats/group. ** & *Significantly different from control group at 0.05, 0.01 level respectively. ## & #Significantly different from MRN-100 group at 0.05, 0.01 level respectively. §§ & §Significantly different from MNNG group at 0.05, 0.01 level respectively. + Significantly different from MNNG+MRN-100 group at 0.01 level.
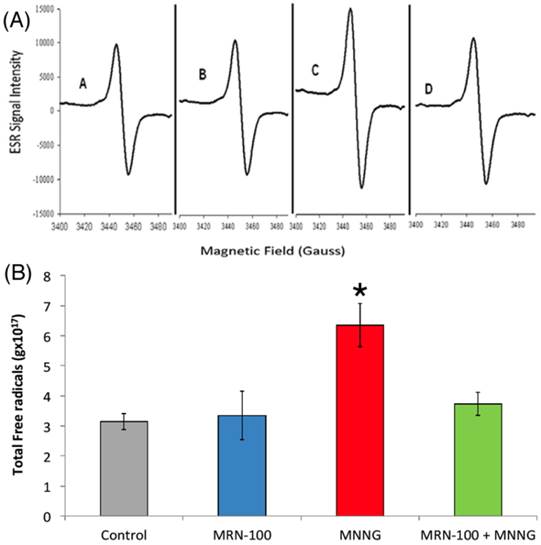
Blood total free radicals in rats treated with MNNG and MNNG+MRN-100 for 33 weeks. (A) Total free radical levels were analyzed by ESR spectra of lyophilized blood samples. (B) Levels of total free radicals in rats under different treatment conditions. * p<0.01 compared to control and other groups. Each bar represents the mean ± SE of 6 rats/group.
Levels of antioxidant enzymes
Carcinogen MNNG depleted the levels of antioxidant enzymes in the blood (SOD: -45.1%, CAT:- 34.0%, and GPx: -48.1%), and gastric tissues (SOD: -69.8%, CAT:-34.0%, and GPx: -36.2%) (p<0.01), as compared to control untreated rats. In contrast, MRN-100 supplementation markedly enhanced the levels of antioxidant enzymes of blood and gastric tissues within reach of normal values (Figure 5).
Level of the total antioxidant capacity (TAC)
Results of TAC levels post treatment with MNNG and MRN-100 are depicted in (Figure 5). Treatment with MNNG resulted in a decrease in the TAC level in the blood (-70.4%-p<0.01) and in the gastric tissues (-70.7%) (p<0.01) as compared to control rats. However, MRN-100 treatment reduced such decline in TAC level in both tissues.
Total free radical (TFR) level
The total levels of free radicals were measured in whole blood by ESR (Figure 6A & B). Quantitative assessments of free-radical concentrations were made and the significance of the differences between mean values was determined. MNNG-treated rats demonstrated a remarkable increase in TFR as compared to control untreated rats. However, treatment with MRN-100 brought the levels to within the normal values.
Discussion
Preventative and protective treatment options for gastric and esophageal cancers are limited, and novel products that effectively combat this disease are in demand. The current study reveals the effectiveness of MRN-100 in suppressing the growth of gastric and esophageal cancers in rats as manifested by the significant reduction in the percentages of rats bearing dysplasia and cancer. MRN-100 treatment resulted in decreased incidents of dysplasia (35%) and cancer (5%) as compared to animals treated with carcinogen alone, (65%) and (20%), respectively (Fig 1). This was associated with the absence of long segments of epithelial involvement and a remarkable decrease in the number of foci in each dysplastic stage.
Results of this study also showed significant loss of body weight in rats bearing dysplasia or gastric/esophageal cancer. These data are in accordance with recent findings on patients with gastrointestinal cancers and lung cancer showing significant weight loss (19, 20). It is of interest to note that supplementation with MRN-100 provided significant protection against the body-weight loss in carcinogen-treated animals.
Results of this study demonstrated that growth of gastric carcinoma is associated with the accumulation of oxygen-derived free radicals, markedly elevated MDA levels, and significant depletion in GSH content and antioxidant enzymes. This observation is in accordance with other studies in tumor-bearing animals (21, 22). During cancer growth, glutathione redox (GSH/GSSG) decreases in the blood of Ehrlich ascites tumor-bearing mice which was attributed to an increase in blood GSSG. This is due to an increase in peroxide production by tumor cells leading to GSH oxidation within the RBCs and the subsequent increase of GSSG release from different tissues into the blood (22). Similar results were found in patients with gastric cancer (23) and laryngeal carcinoma (24).
The generation of reactive oxygen species (ROS) results in lipid peroxidation, DNA degradation, and protein denaturation. Increased levels of ROS has been attributed to the initiation of many diseases, such as cancer (25, 26), aging (27; 28), and diabetes (29). Our earlier studies show MRN-100 may protect against age-induced ROS in rats through modulation of protein oxidation, antioxidant status, and lipid peroxidation in the blood, liver, and brain tissues (7). In this study, MRN-100 also acts as a potent antioxidant agent in carcinogen-induced gastric and esophageal cancers. It protects against carcinogen-induced disturbances in the antioxidant levels of the blood and gastric tissues. This was simplified by the elevation of GSH and antioxidant enzyme levels which was accompanied by reducing the total free radical and malondialdehyde levels. The reduction in ROS by MRN-100 may represent a mechanism by which this agent suppresses gastric and esophageal tumor growth in rats.
The ability of MRN-100 to protect tissues against oxidative stress damage may involve regulating cellular free-iron levels (7), since this metal is known to protect against oxidative stress (30, 31). The increased levels of iron-binding compounds, such as ferritin and transferrin, by MRN-100 may prevent excess iron from taking part in the Fenton reaction which results in the prevention of reactive radical accumulation (7). In this study, we observed MRN-100 prevented a decline in GSH levels in the blood and gastric tissues due to carcinogen treatment. This is particularly interesting because GSH is a major contributor to the endogenous antioxidant system which inhibits the neoplastic process (32). In addition, MRN-100 prevented the decrease of antioxidant enzymes SOD, CAT, and GPx in the blood and gastric tissues. The clearance of superoxide and hydrogen peroxide require the presence of these antioxidant enzymes (33).
The immune modulatory effect of MRN-100 may represent an additional mechanism by which it suppresses the growth of gastric and esophageal cancers induced by carcinogen treatment. Earlier studies showed that oral administration of MRN-100 to healthy subjects and cancer patients resulted in an enhancement of their natural killer (NK) cell activity for up to 12 months (34-36). NK cells have been shown to play an important role in the primary host defense against cancer and virally infected cells (37-39).
In conclusion, MRN-100 exhibited a significant cancer chemo-preventive effect as demonstrated by the significant protection against dysplasia and gastric or esophageal cancer in rats. Our study suggests MRN-100 may be an effective adjuvant for the treatment of gastric or esophageal cancers.
Acknowledgements
This work was supported by Grant # C0030300 from ACM Co., Ltd, Tokyo, Japan (Ghoneum M) and by NIH-NIMHD grant U54MD007598 (formerly U54RR026138) (Pan D). The sponsors had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the manuscript; or in the decision to submit the manuscript for publication.
Competing Interests
The authors have declared that no competing interest exists.
References
1. SEER Cancer Statistics Review, 1975-2010, National Cancer Institute; Bethesda, MD; based on November 2012 SEER data submission, posted to the SEER web site April 2013. Howlader N, Noone AM, Krapcho M, Garshell J, Neyman N, et al. http://seer.cancer.gov/csr/1975_2010/
2. Yakirevich E, Resnick MB. Pathology of gastric cancer and its precursor lesions. Gastroenterol Clin North Am. 2013;42:261-84
3. Lagergren J, Bergström R, Lindgren A, Nyrén O. Symptomatic gastroesophageal reflux disease as a risk factor for esophageal adenocarcinoma. N Engl J Med. 1999;340:825-831
4. Camilleri M, Dubois D, Coulie B, Jones M, Kahrilas PJ. et al. Prevalence and socioeconomic impact of upper gastrointestinal disorders in the United States: results of the US Upper Gastrointestinal Study. Clin Gastroenterol Hepatol. 2005;3:543-552
5. Blakely AM, Miner TJ. Surgical considerations in the treatment of gastric cancer. Gastroenterol Clin North Am. 2013;42:337-357
6. Tan VP, Wong BC. Gastric cancer chemoprevention: the current evidence. Gastroenterol Clin North Am. 2013;42:299-316
7. Badr El-Din NK, Noaman E, Fattah SM, Ghoneum M. Reversal of age-associated oxidative stress in rats by MRN-100, a hydro-ferrate fluid. In Vivo. 2010;24:525-534
8. Ghoneum M, Elbaghdady HA, El-Shebly AA, Pan D, Assanah E. et al. Protective effect of hydroferrate fluid, MRN-100, against lethality and hematopoietic tissue damage in γ-radiated Nile tilapia, Oreochromis niloticus. J Radiat Res. 2013;54:852-862
9. Ghoneum M, Shaheen M. MRN-100, an iron-based compound, possesses anti-HIV activity. In Vitro Evid Based Complement Alternat Med. 2010;7:427-432
10. Yoshioka T, Kawada K, Shimada T, Mori M. Lipid peroxidation in maternal and cord blood and protective mechanism against activated oxygen toxicity in the blood. Am J Obstet Gynecol. 1979;135:372-376
11. Beutler E, Duron O, Kelly BM. Improved method of the determination of blood glutathione. J Lab Clin Med. 1963;61:882-888
12. Minami M, Yoshikawa HA. Simplified assay method of superoxide dismutase activity for clinical use. Clin Chim Acta. 1979;92:337-342
13. Luck H. Catalase. In: (ed.) Bergmeyer HW. Methods of Enzymatic Analysis. New York: Academic Press. 1963:885-894
14. Lawrence RA, Sunde RA, Schwartz GL, Hoekstra WG. Glutathione peroxidase activity in rat lens and other tissues in relation to dietary selenium intake. Exp Eye Res. 1974;18:563-569
15. Miller NJ, Rice-Evans C, Davies MJ, Gopinathan V, Milner A. A novel method for measuring antioxidant capacity and its application for monitoring the antioxidant status in premature neonates. Clin Sci. 1993;84:407-412
16. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with Folin phenol reagent. J Biol Chem. 1951;193:265-275
17. Heckly RJ. Free Radicals in Dry Biological Systems. In: (ed.) Pryor WA. Free Radicals in Biology, vol. 2. New York: Academic Press. 1976:135-158
18. Pascual EC, Goodman BA, Yeretzian C. Characterization of free radicals in soluble coffee by ESR. J Agric Food Chem. 2002;50:6114-6122
19. Huggett MT, Ghaneh P, Pereira SP. Drainage and bypass procedures for palliation of malignant diseases of the upper gastrointestinal tract. Clin Oncol (R Coll Radiol). 2010;22:755-763
20. Spina R, Chu SY, Chatfield M, Chen J, Tin MM. et al. Outcomes of chemoradiation for patients with locally advanced non-small-cell lung cancer. Intern Med J. 2013;43:790-797
21. Badr El-Din NK. Protective role of sanumgerman against γ-irradiation-induced oxidative stress in Ehrlich carcinoma-bearing mice. Nutr Res. 2004;24:271-291
22. Navarro J, Obrador E, Carretero J, Petschen I, Avino J. et al. Changes in glutathione status and the antioxidant system in blood and in cancer cells associate with tumour growth in vivo. Free Radic Biol Med. 1999;26:410-418
23. Bakan E, Taysi S, Polat MF, Dalga S, Umudum Z. et al. Nitric oxide levels and lipid peroxidation in plasma of patients with gastric cancer. Jpn J Clin Oncol. 2002;32:162-166
24. Seven A, Civelek S, Inci E, Inci F, Korkut N. et al. Evaluation of oxidative stress parameters in blood of patients with laryngeal carcinoma. Clin Biochem. 1999;32:369-373
25. Giannoni E, Parri M, Chiarugi P. EMT and oxidative stress: a bidirectional interplay affecting tumor malignancy. Antioxid Redox Signal. 2012;16:1248-63
26. Nikitovic D, Corsini E, Kouretas D, Tsatsakis A, Tzanakakis G. ROS-major mediators of extracellular matrix remodeling during tumor progression. Food Chem Toxicol. 2013;61:178-86
27. Hayakawa M, Sugiyama S, Hattori K, Takasawa M, Ozawa T. Age-associated damage in mitochondrial DNA in humans hearts. Mol Cell Biochem. 1993;119:95-103
28. Finkel T, Holbrook NJ. Oxidants, oxidative stress and the biology of aging. Nature. 2000;408:239-47
29. Fridlyand LE, Philipson LH. Reactive species, cellular repair and risk factors in the onset of type 2 diabetes mellitus: review and hypothesis. Curr Diabetes Rev. 2006;2:241-59
30. Latifi A, Jeanjean R, Lemeille S, Havaux M, Zhang CC. Iron starvation leads to oxidative stress in Anabaena sp. strain PCC 7120. J Bacteriol. 2005;187:6596-6598
31. Balla G, Jacob HS, Balla J, Rosenberg M, Nath K. et al. Vercellotti, Ferritin: a cytoprotective antioxidant strategem of endothelium. J Biol Chem. 1992;267:18148-18153
32. Sinclair AJ, Barnett AH, Lunie J. Free radical and antioxidant systems in health and disease. Br J Hosp Med. 1990;43:334-344
33. Rushmore TH, Picket CD. Glutathione-S-transferase, structure, regulation, and therapeutic implication. J Biol Chem. 1993;268:11475-11478
34. Ghoneum M, Kijima Y. Induction of human natural killer (NK) cell activity by pi-water (MRN-100). Proceedings of the Annual Conference on Clinical Immunology, New Orleans, LA. 1996
35. Ghoneum M, Namatalla G, Kijima Y. Phenotypic analysis of human lymphocyte sub-populations post treatment with pi-water (MRN-100). Proceedings of the Eighty-eighth Annual Meeting of the American Association for Cancer Research, San Diego, CA. 1997
36. Ghoneum M. NK immunorestoration in cancer patients by MRN-100, an iron based compound derived from bivalent and trivalent ferrate. Proceedings of the Fourth International Symposium on Predictive Oncology and Therapy, Nice, France. 1998
37. Whiteside TL, Herberman RB. The role of natural killer cells in immune surveillance of cancer. Curr Opin Immunol. 1995;7:704-710
38. Badr El-Din NK, Noaman E, Ghoneum M. In vivo tumor inhibitory effects of nutritional rice bran supplement MGN-3/Biobran on Ehrlich carcinoma-bearing mice. Nutr Cancer. 2008;60:235-244
39. Ames E, Murphy WJ. Advantages and clinical applications of natural killer cells in cancer immunotherapy. Cancer Immunol Immunother. 2014;63:21-28
Author contact
Corresponding author: Mamdooh H. Ghoneum, Ph. D. Address: 1731 E. 120th Street, Los Angeles, CA 90059. Email: mghoneumedu. Phone: (310) 474-6724/(323) 562-5953. Fax: (310)474-6724

 Global reach, higher impact
Global reach, higher impact