Impact Factor ISSN: 1449-2288
Int J Biol Sci 2018; 14(7):726-735. doi:10.7150/ijbs.24505 This issue Cite
Research Paper
FBW7 suppresses metastasis of colorectal cancer by inhibiting HIF1α/CEACAM5 functional axis
1. Department of Colorectal Surgery, Fudan University Shanghai Cancer Center, Shanghai 200032, China;
2. Department of Oncology, Shanghai Medical College, Fudan University, Shanghai 200032, China;
3. Departments of CyberKnife, Huashan Hospital, Fudan University, Shanghai 200032, China.
*Qingguo Li and Yaqi Li contributed equally to this work.
Abstract
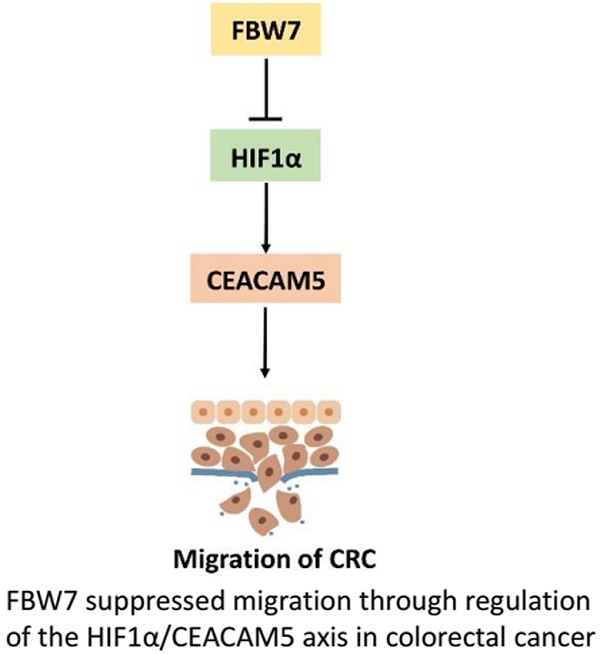
F-box and WD repeat domain-containing 7 (FBW7) functions as a major tumor suppressor by targeting oncoproteins for degradations. FBW7 has been reported to be one of the most frequently mutated genes in colorectal cancer (CRC). However, its roles and possible mechanisms in the development of CRC are still unclear. In the present study, we adopted immunohistochemistry staining in tissue microarray (TMA), consisting of 276 samples from stage I-IV CRC patients, and analyzed the correlation between FBW7 expression and clinicopathological parameters, as well as overall survival (OS) and disease-free survival (DFS). The impact of FBW7 on migration was further validated in vitro. Whole-genome expression microarray (GEO,accession numbers GSE76443), was then analyzed to find the possible target of FBW7. The results were verified by functional experiments in vitro and IHC staining of TMA. Finally, luciferase and chromatin immunoprecipitation (ChIP) assays were carried out to identify the possible mechanisms. The expression level of FBW7 in TMA was negatively correlated with serum CEA level, venous invasion, N stage and M stage, and positively associated with the survival of CRC patients(P<0.05). Ectopic FBW7 expression significantly suppressed migration of colon cancer cells in vitro. GEO analysis revealed that decreased FBW7 significantly correlated with increased level of CEACAM5, which encoded CEA. The correlation was verified by IHC of TMA and silencing CEACAM5 inhibited migration in vitro. Mechanistically, we demonstrated that CEACAM5 was a HIF1α target gene and that FBW7 regulated CEACAM5 in a HIF1α-dependent manner. In conclusion, our results revealed that FBW7 suppressed migration through regulation of the HIF1α/CEACAM5 axis in colorectal cancer. Therefore, our study sheds novel lights on the impact of FBW7 on HIF1α/CEACAM5 signaling axis and constitutes potential prognostic predictors and therapeutic targets for CRC.
Keywords: FBW7, Colorectal cancer, HIF1α, CEACAM5

 Global reach, higher impact
Global reach, higher impact