Impact Factor ISSN: 1449-2288
Int J Biol Sci 2019; 15(6):1161-1176. doi:10.7150/ijbs.32004 This issue Cite
Research Paper
Hypoxia preconditioned renal tubular epithelial cell-derived extracellular vesicles alleviate renal ischaemia-reperfusion injury mediated by the HIF-1α/Rab22 pathway and potentially affected by microRNAs
1. Department of Urology, Zhongda Hospital, Southeast University, Nanjing, Jiangsu 210009, P.R. China;
2. Institute of Urology, Surgical Research Center, School of Medicine, Southeast University, Nanjing, Jiangsu 210009, P.R. China;
3. Department of Respiratory Medicine, The First Hospital of Jilin University, Changchun, Jilin 130000, P.R. China;
4. Department of Urology, The First Affiliated Hospital of Soochow University, Suzhou, Jiangsu 215006, P.R. China.
5. Department of Urology, The Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China.
* Equal contributors
Abstract
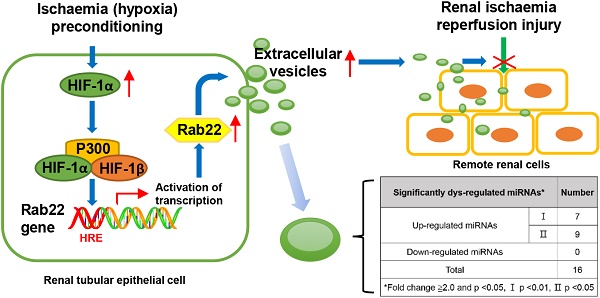
We previously found that hypoxia induced renal tubular epithelial cells (RTECs) release functional extracellular vesicles (EVs), which mediate the protection of remote ischaemic preconditioning (RIPC) for kidney ischaemia-reperfusion (I/R) injury. We intend to investigate whether the EVs were regulated by hypoxia-inducible factor 1α (HIF-1α) and Rab22 during RIPC. We also attempted to determine the potentially protective cargo of the EVs and reveal their underlying mechanism. Hypoxia preconditioning (HPC) of human kidney 2 (HK2) cells was conducted at 1% oxygen (O2) for different amounts of time to simulate IPC in vitro. EVs were isolated and then quantified. HIF-1α- and Rab22-inhibited HK2 cells were used to investigate the role of the HIF-1α/Rab22 pathway in HPC-induced EV production. Both normoxic and HPC EVs were treated in vivo to assess the protective effect of I/R injury. Moreover, microRNA (miRNA) sequencing analysis and bioinformatics analysis was performed. We revealed that the optimal conditions for simulating IPC in vitro was no more than 12 h under the 1% O2 culture circumstance. HPC enhanced the production of EVs, and the production of EVs was regulated by the HIF-1α/Rab22 pathway during HPC. Moreover, HPC EVs were found to be more effective at attenuating mice renal I/R injury. Furthermore, 16 miRNAs were upregulated in HPC EVs. Functional and pathway analysis indicated that the miRNAs may participate in multiple processes and pathways by binding their targets to influence the biochemical results during RIPC. We demonstrated that HIF-1α/Rab22 pathway mediated RTEC-derived EVs during RIPC. The HPC EVs protected renal I/R injury potentially through differentially expressed miRNAs. Further study is needed to verify the effective EV-miRNAs and their underlying mechanism.
Keywords: extracellular vesicles, ischaemic preconditioning, ischaemia-reperfusion injury, hypoxia, microRNA

 Global reach, higher impact
Global reach, higher impact