Impact Factor ISSN: 1449-2288
Int J Biol Sci 2020; 16(16):3184-3199. doi:10.7150/ijbs.45505 This issue Cite
Research Paper
Histone deacetylase inhibitor MPT0B291 suppresses Glioma Growth in vitro and in vivo partially through acetylation of p53
1. Graduate Institute of Medical Sciences, College of Medicine, Taipei Medical University, 110 Taipei, Taiwan.
2. Department of Neurology, Mongolian National University of Medical Sciences, 14210 Ulaanbaatar, Mongolia.
3. Department of Pathology and Neurosurgery, University of Maryland School of Medicine, 21201 Baltimore, MD, USA.
4. Department of Neurosurgery, Wan Fang Hospital, Taipei Medical University, 116 Taipei, Taiwan.
5. Department of Neurosurgery, Taipei Medical University Hospital, Taipei Medical University, 110 Taipei, Taiwan.
6. Neuroscience Research Center, Taipei Medical University, 110 Taipei, Taiwan.
7. School of Pharmacy, College of Pharmacy, Taipei Medical University, 110 Taipei, Taiwan.
Abstract
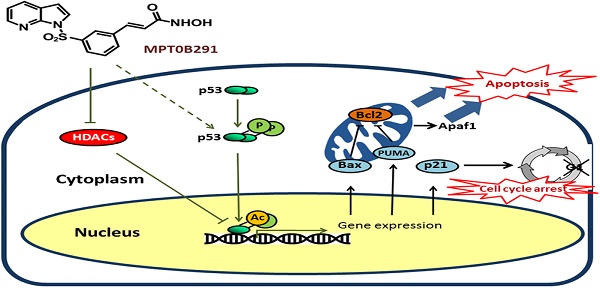
Background: Histone deacetylase (HDAC) inhibitors have emerged as a new class of anti-tumor agents for various types of tumors, including glioblastoma.
Methods and results: We found that a novel HDAC inhibitor, MPT0B291, significantly reduced the cell viability and increased cell death of human and rat glioma cell lines, but not in normal astrocytes. We also demonstrated that MPT0B291 suppressed proliferation by inducing G1 phase cell cycle arrest and increased apoptosis in human and rat glioma cell lines by flow cytometry and immunocytochemistry. We further investigated the anti-tumor effects of MPT0B291 in xenograft (mouse) and allograft (rat) models. The IVIS200 images and histological analysis indicated MPT0B291 (25 mg/kg, p. o.) reduced tumor volume. Mechanistically, MPT0B291 increased phosphorylation and acetylation/activation of p53 and increased mRNA levels of the apoptosis related genes PUMA, Bax, and Apaf1 as well as increased protein level of PUMA, Apaf1 in C6 cell line. The expression of cell cycle related gene p21 was also increased and Cdk2, Cdk4 were decreased by MPT0B291.
Conclusion: Our study highlights the anti-tumor efficacy of a novel compound MPT0B291 on glioma growth.
Keywords: glioma, HDAC6 inhibition, cell death, cell cycle arrest, xenograft, allograft

 Global reach, higher impact
Global reach, higher impact