Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(5):1217-1233. doi:10.7150/ijbs.56833 This issue Cite
Research Paper
3,3'-Diindolylmethane induces gastric cancer cells death via STIM1 mediated store-operated calcium entry
1. Department of Preventive Medicine and Public Health Laboratory Science, School of Medicine, Jiangsu University, Zhenjiang, China.
2. Department of Gastroenterology, Affiliated Hospital of Jiangsu University, Jiangsu University, Zhenjiang, China.
3. Department of Clinical Laboratory Center, Shaoxing People's Hospital (Shaoxing Hospital, Zhejiang University School of Medicine), Shaoxing, China.
4. Key Laboratory of Medical Science and Laboratory Medicine of Jiangsu Province, School of Medicine, Jiangsu University, Zhenjiang, China.
5. Center for Experimental Research, Affiliated Kunshan Hospital to Jiangsu University School of Medicine, Kunshan, Suzhou, China.
6. Department of Health Care, Zhenjiang Fourth Peoples Hospital, Zhenjiang, China.
* These authors contributed equally.
Abstract
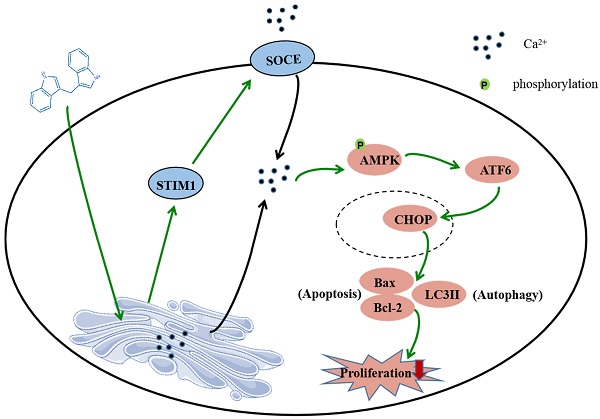
3,3'-Diindolylmethane (DIM), a natural phytochemicals isolated from cruciferous vegetables, has been reported to inhibit human gastric cancer cells proliferation and induce cells apoptosis as well as autophagy, but its mechanisms are still unclear. Store-operated calcium entry (SOCE) is a main Ca2+ influx pathway in various of cancers, which is activated by the depletion of endoplasmic reticulum (ER) Ca2+ store. Stromal interaction molecular 1 (STIM1) is the necessary component of SOCE. In this study, we focus on to examine the regulatory mechanism of SOCE on DIM-induced death in gastric cancer. After treating the human BGC-823 and SGC-7901 gastric cancer cells with DIM, cellular proliferation was determined by MTT, apoptosis and autophagy were detected by flow cytometry or Hoechst 33342 staining. The expression levels of related proteins were evaluated by Western blotting. Free cytosolilc Ca2+ level was assessed by fluorescence monitoring under a laser scanning confocal microscope. The data have shown that DIM could significantly inhibit proliferation and induce apoptosis as well as autophagy in two gastric cancer cell lines. After DIM treatment, the STIM1-mediated SOCE was activated by upregulating STIM1 and decreasing ER Ca2+ level. Knockdown STIM1 with siRNA or pharmacological inhibition of SOCE attenuated DIM induced apoptosis and autophagy by inhibiting p-AMPK mediated ER stress pathway. Our data highlighted that the potential of SOCE as a promising target for treating cancers. Developing effective and selective activators targeting STIM1-mediated SOCE pathway will facilitate better therapeutic sensitivity of phytochemicals acting on SOCE in gastric cancer. Moreover, more research should be performed to validate the efficacy of combination chemotherapy of anti-cancer drugs targeting SOCE for clinical application.
Keywords: 3,3'-Diindolylmethane, stromal interaction molecular 1, store-operated calcium entry, p-AMPK, endoplasmic reticulum stress, gastric cancer.

 Global reach, higher impact
Global reach, higher impact