Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(5):1395-1412. doi:10.7150/ijbs.57774 This issue Cite
Research Paper
Moderate mechanical stimulation rescues degenerative annulus fibrosus by suppressing caveolin-1 mediated pro-inflammatory signaling pathway
1. Department of Orthopaedic Surgery, Orthopaedic Institute, The First Affiliated Hospital, Soochow University, Suzhou, Jiangsu, China.
2. China Orthopaedic Regenerative Medicine Group (CORMed), Hangzhou, Zhejiang, China.
#These authors contributed equally to this study.
Abstract
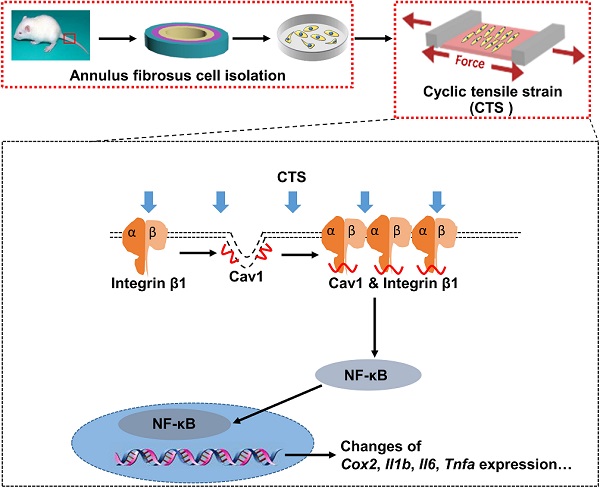
Mechanical loading can induce or antagonize the extracellular matrix (ECM) synthesis, proliferation, migration, and inflammatory responses of annulus fibrosus cells (AFCs), depending on the loading mode and level. Caveolin-1 (Cav1), the core protein of caveolae, plays an important role in cellular mechanotransduction and inflammatory responses. In the present study, we presented that AFCs demonstrated different behaviors when subjected to cyclic tensile strain (CTS) for 24 h at a magnitude of 0%, 2%, 5% and 12%, respectively. It was found that 5% CTS had positive effects on cell proliferation, migration and anabolism, while 12% CTS had the opposite effects. Besides, cells exposed to interleukin-1β stimulus exhibited an increase expression in inflammatory genes, and the expression of these genes decreased after exposure to moderate mechanical loading with 5% CTS. In addition, 5% CTS decreased the level of Cav1 and integrin β1 and exhibited anti-inflammatory effects. Moreover, the expression of integrin β1 and p-p65 increased in AFCs transfected with Cav1 plasmids. In vivo results revealed that moderate mechanical stimulation could recover the water content and morphology of the discs. In conclusion, moderate mechanical stimulation restrained Cav1-mediated signaling pathway and exhibited anti-inflammatory effects on AFCs. Together with in vivo results, this study expounds the underlying molecular mechanisms on the effect of moderate mechanical stimulation on intervertebral discs (IVDs) and may provide a new therapeutic strategy for the treatment of IVD degeneration.
Keywords: annulus fibrosus, moderate mechanical stimulation, annulus fibrosus regeneration, caveolin-1, integrin β1, NF-κB, anti-inflammation

 Global reach, higher impact
Global reach, higher impact