Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(15):4165-4175. doi:10.7150/ijbs.63889 This issue Cite
Research Paper
SOCS2 Suppresses Inflammation and Apoptosis during NASH Progression through Limiting NF-κB Activation in Macrophages
1. Department of Gastroenterology, the First Affiliated Hospital of Nanjing Medical University, Jiangsu, China.
2. Hepatobiliary Center, The First Affiliated Hospital of Nanjing Medical University.
3. Key Laboratory of Liver Transplantation, Chinese Academy of Medical Sciences.
4. NHC Key Laboratory of Living Donor Liver Transplantation (Nanjing Medical University), Nanjing, Jiangsu Province, China.
5. Department of General Surgery, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu, 210029, People's Republic of China.
6. The First School of Clinical Medicine, Nanjing Medical University, Nanjing, People's Republic of China.
#These authors contributed equally to this work.
Abstract
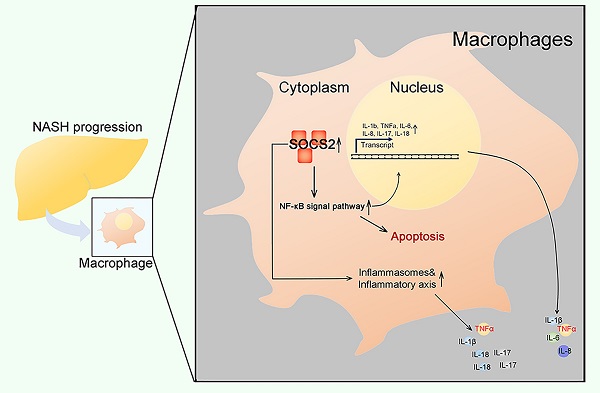
Background: Inflammation and apoptosis play a crucial role in the progression of nonalcoholic steatohepatitis (NASH). Suppressor of cytokine signaling 2 (SOCS2) is one of classic negative regulators of cytokine signaling, which has recently been described as anti-inflammatory mediators. However, the role of SOCS2 in macrophages during NASH progression and the relationship among SOCS2, inflammation, apoptosis and NASH is largely unknown. Herein, we aimed to study the function of SOCS2 in NASH progression.
Methods: We detected SOCS2 expression in macrophages in human subjects without steatosis, with simple steatosis and with NASH to confirm the relationship between SOCS2 and NASH. Free fatty acids was used to establish stress environment in RAW 264.7 cell lines stably overexpressing or knockdown SOCS2. In vitro and vivo assays also performed to study the molecular function of SOCS2 in NASH progression.
Findings: Our human samples illustrated that SOCS2 was decreased in macrophages during NASH progression and was negatively correlated to NASH level. Meanwhile, In vitro assays showed SOCS2 overexpression in macrophages suppressed inflammation and apoptosis via inhibiting NF-κB signaling pathway, while SOCS2 knock-down in macrophages caused an increased activation of NF-κB, which could be blocked by ammonium 1-pyrrolidinedithiocarbamate (PDTC). In addition, SOCS2 in macrophages also suppressed inflammation via limiting the activation of inflammasomes. Consistent with these, our BMT model also confirmed the SOCS2 function in macrophages during NASH.
Interpretation: Our data strongly indicate that SOCS2 plays a role in inhibiting inflammation and apoptosis via NF-κB and inflammasome signaling pathway in macrophages during NASH. Further studies are required to explore the potential preventive and therapeutic strategies of SOCS2 for this common liver disease.
Keywords: Nonalcoholic steatohepatitis, Suppressor of cytokine signaling 2, inflammation, apoptosis, NF-κB

 Global reach, higher impact
Global reach, higher impact