Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(2):783-799. doi:10.7150/ijbs.65211 This issue Cite
Research Paper
The cross-talk between PARylation and SUMOylation in C/EBPβ at K134 site participates in pathological cardiac hypertrophy
1. Department of Pharmacology and Toxicology, School of Pharmaceutical Sciences, Sun Yat-Sen University, Guangdong, China.
2. Laboratory of Hematopathology & Drug Discovery, School of Medicine, South China University of Technology, Guangdong, China.
3. Department of Endocrinology, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Anhui, China.
4. Center for Phenomics of Traditional Chinese Medicine/the Affiliated Hospital of Traditional Chinese Medicine, Southwest Medical University, Sichuan, China.
5. National and Local United Engineering Lab of Druggability and New Drugs Evaluation, School of Pharmaceutical Sciences, Sun Yat-Sen University, Guangdong, China.
6. School of Pharmacy, Jinan University, 601 Huangpu Avenue West, Guangdong, China.
*These authors contributed equally: Luping Wang, Panxia Wang.
Abstract
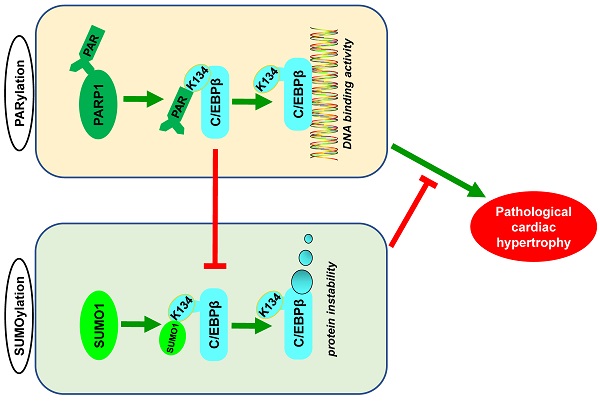
Poly(ADP-ribosyl)ation (PARylation) and SUMO modification (SUMOylation) are novel post-translational modifications (PTMs) mainly induced by PARP1 and SUMO1. Growing evidence has revealed that C/EBPβ plays multiple roles in biological processes and participates in cardiovascular diseases. However, the cross-talk between C/EBPβ PARylation and SUMOylation during cardiovascular diseases is unknown. This study aims to investigate the effects of C/EBPβ PTMs on cardiac hypertrophy and its underlying mechanism. Abdominal aortic constriction (AAC) and phenylephrine (PE) were conducted to induce cardiac hypertrophy. Intramyocardial delivery of recombinant adenovirus (Ad-PARP1) was taken to induce PARP1 overexpression. In this study, we found C/EBPβ participates in PARP1-induced cardiac hypertrophy. C/EBPβ K134 residue could be both PARylated and SUMOylated individually by PARP1 and SUMO1. Moreover, the accumulation of PARylation on C/EBPβ at K134 site exhibits downregulation of C/EBPβ SUMOylation at the same site. Importantly, C/EBPβ K134 site SUMOylation could decrease C/EBPβ protein stability and participates in PARP1-induced cardiac hypertrophy. Taken together, these findings highlight the importance of the cross-talk between C/EBPβ PTMs at K134 site in determining its protein level and function, suggesting that multi-target pharmacological strategies inhibiting PARP1 and activating C/EBPβ SUMOylation would be potential for treating pathological cardiac hypertrophy.
Keywords: poly(ADP-ribosyl)ation, C/EBPβ, SUMOylation, cardiac hypertrophy

 Global reach, higher impact
Global reach, higher impact