Impact Factor ISSN: 1449-2288
Int J Biol Sci 2023; 19(3):852-864. doi:10.7150/ijbs.77434 This issue Cite
Research Paper
Regulating Neutrophil PAD4/NOX-Dependent Cerebrovasular Thromboinflammation
1. Department of Neurology, Louisiana State University Health Sciences Center-Shreveport, Shreveport, LA, 71130, USA.
2. Department of Molecular & Cellular Physiology, Louisiana State University Health Sciences Center-Shreveport, Shreveport, LA, 71130, USA.
3. Department of Life Sciences, Centre of Inflammation Research and Translational Medicine (CIRTM), Brunel University London, London. UB8 3PH, UK.
Abstract
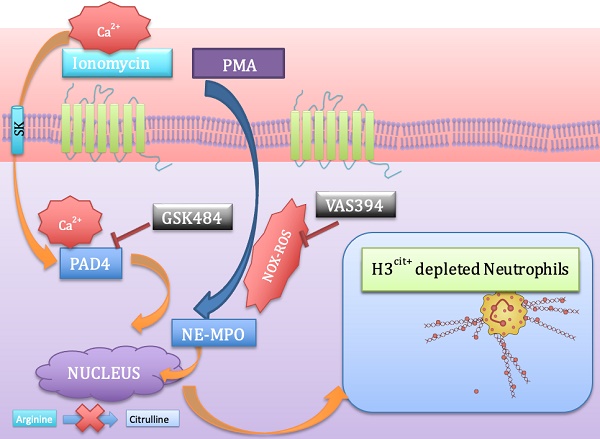
Background: Neutrophil extracellular trap (NET) production has been implicated in the pathogenesis of thromboinflammatory conditions such as Sickle Cell Disease (SCD), contributing to heightened risk for ischemic stroke. NETs are catalyzed by the enzyme Peptidyl Arginine Deiminase 4 (PAD4) and neutrophil derived reactive oxygen species (ROS), especially NADPH oxidase (NOX) which interacts with PAD4 and is therefore critical for neutrophil function. However, the role that NOX-dependent ROS and NETs play in the accelerated cerebral microvascular thrombosis associated with thromboinflammatory conditions, such as SCD, has not been fully elucidated and is the aim of this study.
Methods: The in-vitro effects of targeting PAD4 and NOX were examined using physiologically relevant NET assays with neutrophils isolated from healthy volunteers (control) and SCD patients. In addition, in-vivo intravascular effects of targeting PAD4 and NOX in the cerebral microcirculation of C57BL/6 and sickle transgenic mice (STM) were assessed using a photoactivation thrombosis model (light/dye) coupled with real-time fluorescence intravital microscopy.
Results: We found that targeting PAD4 and NOX in human neutrophils significantly inhibited ionomycin dependent H3cit+ neutrophils. Targeting PAD4 and NOX in-vivo resulted in prolonged blood flow cessation in cerebrovascular arterioles as well as venules. Moreover, we were able to replicate the effects of PAD4 and NOX targeting in a clinical model of accelerated thromboinflammation by increasing blood flow cessation times in cerebral microvessels in STM. These findings concurred with the clinical setting i.e. neutrophils isolated from SCD patients, which possessed an attenuation of H3cit+ neutrophil production on targeting PAD4 and NOX.
Conclusions: Taken together, our compelling data suggests that PAD4 and NOX play a significant role in neutrophil driven thromboinflammation. Targeting PAD4 and NOX limits pathological H3cit+ neutrophils, which may further explain attenuation of cerebral thrombosis. Overall, this study presents a viable pre-clinical model of prevention and management of thromboinflammatory complications such as ischemic stroke.
Keywords: Thromboinflammation, neutrophils, thrombosis, neutrophil extracellular traps, peptidyl arginine deiminase 4, NADPH oxidase

 Global reach, higher impact
Global reach, higher impact