Impact Factor ISSN: 1449-2288
Int J Biol Sci 2026; 22(7):3509-3521. doi:10.7150/ijbs.130260 This issue Cite
Review
Natural Killer Cell Therapy in Ovarian Cancer: From Preclinical Potentials to Clinical Applications and Combination Strategies
1. Obstetrics & Gynecology Hospital of Fudan University, Shanghai Key Lab of Reproduction and Development, Shanghai Key Lab of Female Reproductive Endocrine Related Diseases, Shanghai, China.
2. Department of Gynecology, Obstetrics and Gynecology Hospital, Fudan University, Shanghai, China.
Received 2025-12-19; Accepted 2026-3-4; Published 2026-3-17
Abstract
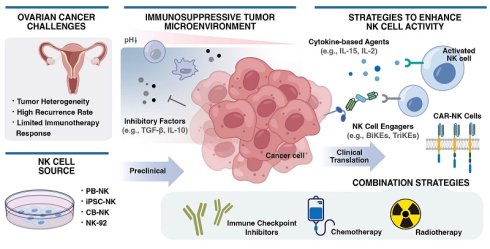
Ovarian cancer represents a leading cause of mortality among gynecologic cancers, with limited therapeutic options for advanced and recurrent disease, highlighting an urgent need for innovative immunotherapies. Natural Killer (NK) cells, functioning as crucial effector cells of the innate immune system, have been identified as promising candidates for ovarian cancer treatment. This review systematically examines the evolving landscape of NK cell-based therapies for ovarian cancer, spanning their preclinical rationale, clinical translation, and innovative combination strategies. Nevertheless, the highly immunosuppressive tumor microenvironment (TME) of ovarian cancer and barriers to tumor infiltration pose significant challenges to their clinical efficacy. Here, we discuss various therapeutic strategies (such as cytokine-based agents, NK cell engagers and chimeric antigen receptor NK cells) designed to enhance NK cell activity, which leverage the unique characteristics of NK cells to complement standard treatments and potentiate combination immunotherapies. Ongoing preclinical and clinical investigations are paramount to converting these advances into efficacious therapies, ultimately revolutionizing the therapeutic paradigm for ovarian cancer.
Keywords: ovarian cancer, natural killer cells, immunotherapy, tumor microenvironment, clinical trials, chimeric antigen receptor
1. Introduction
Ovarian cancer, as one of the common malignant tumors of the female reproductive system, presents a significant global clinical challenge due to its high mortality and recurrence rates [1, 2]. Currently, first-line treatment for ovarian cancer is based on cytoreductive surgery combined with platinum-taxane chemotherapy. However, as most patients are diagnosed at an advanced stage and frequently develop chemoresistance [3], the efficacy of these conventional therapies is significantly limited, resulting in an unfavorable prognosis. Therefore, innovative therapeutic strategies are urgently required to improve the outcomes in ovarian cancer patients. In recent years, immunotherapy has made breakthrough progress in ovarian cancer treatment, and with ongoing characterization of its intricate tumor microenvironment [4, 5], it has become clear that overcoming the highly immunosuppressive tumor microenvironment of ovarian cancer will greatly enhance the efficacy of immunotherapy.
Within this context, natural killer (NK) cells, as pivotal effector cells of the innate immune system, have attracted considerable interest owing to their distinct anti-tumor potential [6]. NK cells have the capability to recognize tumor cells without prior sensitization [7], and they play a dual role in synergistic tumor lysis and initiation of adaptive immunity by secreting various cytokines and chemokines. Preliminary evidence indicates a positive correlation between NK cell infiltration levels in various solid tumors and favorable patient prognoses [8, 9]. Nevertheless, in the highly complex ovarian cancer tumor microenvironment (TME), infiltrating NK cells are often dysfunctional. This phenomenon highlights the substantial and yet untapped potential of NK cells in ovarian cancer immunotherapy [10].
This review aims to explore the cytotoxic effects and immunomodulatory functions of NK cells in anti-tumor immunity. Focusing on preclinical studies and evaluating the prospects of NK cell-based immunotherapeutic approaches in combined therapies for ovarian cancer, we present a summary of the research advancements and future directions of NK cells in this field. This work aims to offer a conceptual foundation for clinical application, ultimately bettering patient outcomes.
2. The antitumor functions of NK cells
Natural killer cells were described about 50 years ago as a distinct lymphoid cell subset, characterized by their lack of antigen-specific receptors and ability to rapidly eliminate virus-infected and malignant cells. Traditionally, NK cell subsets are classified based on the surface expression of CD56 and CD16a, predominantly into CD56brightCD16lo NK cells and CD56dimCD16hi NK cells [11]. Recent advancements in high-dimensional single-cell analysis technologies have greatly deepened our comprehension of human NK cell heterogeneity [12-14]. Researchers have categorized peripheral blood NK cells into three core subsets (NK1, NK2 and NK3), which are respectively enriched with classical CD56dim NK cells, CD56bright NK cells, and human cytomegalovirus-driven adaptive NK cells. The gene signatures of these NK subsets are applicable not only for classifying peripheral blood NK cells in healthy individuals but also for describing NK cell heterogeneity across diverse tissues. Notably, NK2 cells are found to be enriched in numerous tumor types, including ovarian cancer. While the features of NK cell dysfunction within the TME have been well established [15], the specific phenotype of these dysfunctional NK cells remains uncleared. The impaired cytotoxic capacity of NK cells in the TME correlates with the lower expression of effector molecules characteristic of NK2 cells, suggesting that an increased proportion of NK2 cells may be related to tumor immune escape.
2.1 Mediating direct cytotoxicity
The precise regulation of NK cell function relies on the dynamic balance between its surface activating and inhibitory receptors, engaging with specific ligands on target cells [16]. Employing the classic "missing-self" mechanism, NK cells effectively distinguish "self" from "non-self." Their inhibitory receptors (e.g., KIRs, NKG2A heterodimer), recognize major histocompatibility complex class I (MHC-I) molecules on normal cells, thereby transmitting inhibitory signals that prevent attacks on normal cells (Figure 1). To evade T cell surveillance, malignant cells often downregulate MHC-I expression. This reduction inherently diminishes the inhibitory signals to NK cells, consequently leading to their activation. Ideally, this process enables NK cells to effectively identify tumor cells with absent or low MHC-I expression [17]. For example, certain poorly differentiated ovarian cancer cell lines (OVCAR8 and CaOV3) remain considerable susceptibility to NK cell-mediated cytotoxicity despite the diminished MHC-I expression and high invasiveness [18].
Natural killer cell anti-tumor activity and TME-mediated suppression in ovarian cancer. (A) Upon recognition, NK cells deploy multiplex cytotoxic mechanisms: 1) direct killing via perforin/granzyme release and death receptor pathways (e.g., FAS/FASL, TRAIL); 2) ADCC mediated by CD16a; and 3) immunomodulation via cytokine/chemokine secretion (e.g., IFN-γ, TNF-α, CCL5). Together, these functions coordinate a potent anti-tumor immune response. (B) The immunosuppressive TME compromises NK cell activity through three principal mechanisms: 1) dysregulated receptor-ligand interactions that favor inhibitory signaling and impair cancer cell recognition; 2) metabolic molecules (e.g., IDO, lactate) and soluble inhibitory factors (e.g., TGF-β, IL-10), leading to functional exhaustion; and 3) recruitment and activation of immunosuppressive cells, including TAMs, MDSCs, CAFs, Bregs, and Tregs.
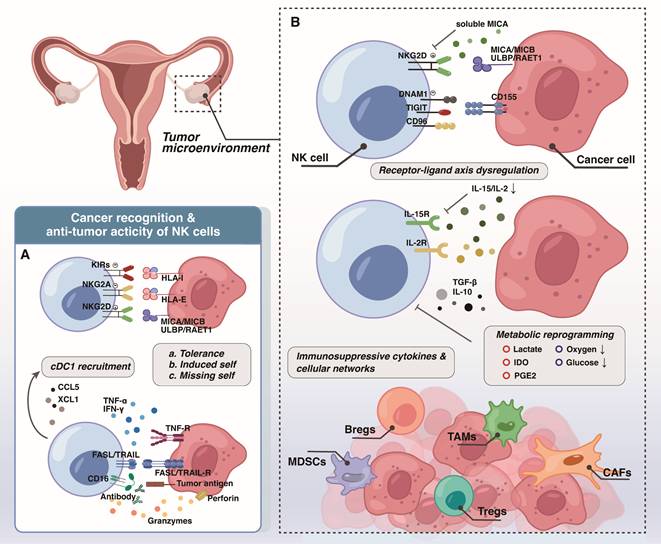
Furthermore, NK cells express a variety of activating receptors, including NKG2D, natural cytotoxicity receptors (NCRs), and CD16 (FcγRIIIA). These receptors recognize upregulated stress ligands (e.g., MICA/B, B7-H6) on tumor cells, transmitting strong activating signals. It is noteworthy that the binding affinity of CD16a to the Fc region of an antibody is a key determinant of antibody-dependent cellular cytotoxicity (ADCC) efficiency [19, 20], resulting in effective tumor lysis. When activating signals surpass inhibitory signals, NK cells achieve full activation, and the specific mechanisms by which their different subsets induce tumor cell apoptosis vary slightly. The NK1 subset, characterized by high cytotoxicity, relies on the classic perforin/granzyme pathway to directly lyse target cells, while also expressing FasL to enhance killing effects [14]. In contrast, NK2 cells highly express TRAIL (TNFSF10) to induce tumor cell apoptosis and predominantly engage in immunomodulation by secreting cytokines, consequently bolstering adaptive immune responses to synergistically achieve broader immune surveillance and defense [14].
2.2 Orchestrating immunomodulation
Besides direct cytotoxicity, NK cells play a significant immunomodulatory role in anti-tumor immunity. Their crosstalk with conventional type 1 dendritic cells (cDC1) cells exemplifies this interaction. Latest research highlights that the NK-cDC1 axis plays a crucial role in early immune surveillance of cells with chromosomal instability (CIN) or TP53 mutations, during the very early stages of high-grade serous ovarian carcinoma (HGSOC) development [21]. It is known that activated NK cells efficiently recruit cDC1s into tumors by secreting chemokine (C-C motif) ligand 5 (CCL5) and XCL1 [22], and continuously provide FLT3LG to maintain cDC1 proliferation and survival [23, 24]. Studies demonstrate that NK cell-derived FLT3L is vital for maintaining cDC1 cell abundance, which correlates with improved overall survival (OS) and enhanced response to anti-PD-1 immunotherapy in melanoma patients [23]. Moreover, NK cells secrete cytokines such as interferon-γ (IFN-γ), tumor necrosis factor-α (TNF-α), and granulocyte-macrophage colony-stimulating factor (GM-CSF), which together with CD40/ CD40 ligand (CD40L) interactions promote cDC1 maturation [25, 26], thereby enhancing their capacity to cross-present tumor antigens and activate CD8⁺ T cells.
Research further indicates that CD103+ Batf3-dependent cDC1 cells secrete interleukin-12 (IL-12) to stimulate NK cell production of IFN-γ, an interaction critical for suppressing tumor metastasis [27]. One reported case of stage IV metastatic ovarian cancer showed positive outcomes, including tumor reduction, ascites regression, and declining tumor markers, after treatment with chemotherapy combined with a Wilms' tumor antigen 1 (WT1) dendritic cell vaccine, highly activated NK cell therapy, and Nivolumab [28]. This indicates that combining multiple immunotherapies can elicit synergistic anti-tumor effects via a bidirectional activation mechanism between NK cells and dendritic cells, thus offering novel therapeutic avenues for patients with advanced cancer.
3. Impaired NK cell function in the ovarian cancer TME
Even with increasing clarity regarding the heterogeneity of NK cell subsets within the ovarian cancer TME, their intrinsic anti-tumor activity remains significantly constrained [9]. This prevalent NK cell dysfunction is attributable to multiple convergent factors within the complex microenvironment. Firstly, dysregulation of NK cell receptor-ligand interactions directly impairs tumor cell recognition by NK cells. In addition, immunosuppressive cytokines and cellular networks, along with metabolic reprogramming within the TME, collectively impose significant constraints on NK cell survival and function.
3.1 Dysregulation of NK cell receptor-ligand interactions
Studies on the immunosuppressive state in individuals with primary and recurrent epithelial ovarian cancer highlight a downregulation of NKG2D [29], the crucial activating receptor on CD56bright NK cells, primarily driven by abnormally elevated levels of soluble MICA (sMICA). MICA/B, which are MHC class I-related stress-induced antigens, typically activate NK cell anti-tumor immunity by engaging NKG2D [30]. Research indicates that elevated concentration of sMICA in the serum competitively bind to NKG2D receptors on NK cells, thereby impeding NKG2D from effectively recognizing MICA/B ligands on tumor cells [29]. Moreover, continuous exposure to such high sMICA levels can diminish NKG2D+ immune cell subsets and induce NK cell anergy [9, 29], facilitating immune escape.
NK cells extracted from the ascites of ovarian cancer patients show reduced expression of DNAM-1 (CD226), while inhibitory receptors TIGIT and CD96 are highly expressed [31]. CD155 acts as a ligand for DNAM-1, TIGIT, and CD96, and in ovarian cancer, CD155 exhibits substantially higher affinity for TIGIT than for DNAM-1, thereby tilting the functional balance toward inhibition. Investigations have found that focal adhesion kinase (FAK) activity in high-grade serous ovarian cancer affects the expression of CD155 on tumor cells, and FAK inhibitors can mitigate the immunosuppressive effects of the CD155/TIGIT axis [32].
Therefore, upregulating activating receptors on NK cells [33, 34] and blocking inhibitory receptor signaling represent crucial avenues for developing innovative therapeutic strategies against ovarian cancer. Given the high proportion of PD-1+ NK cells also found in the peripheral blood and ascites of ovarian cancer patients [35], this provides a theoretical basis for further combining immune checkpoint inhibitors [36] to restore NK cell cytotoxicity, with related phase I/II clinical trials (NCT02671435, NCT02459301) have already been initiated in solid tumors.
3.2 Immunosuppressive cytokines and cellular networks
Malignant ascites (MA) develops in up to 90% of stage III/IV ovarian cancer patients, representing a critical pathological feature of advanced ovarian cancer [37]. This intricate peritoneal fluid, rich in both cellular and acellular components [38], serves as a vital source for characterizing the ovarian cancer TME. Its fluctuating cytokine levels and heterogeneity of immune cell subsets are strongly correlated with adverse patient outcomes [37].
It is noteworthy that among multiple anatomical sites in advanced ovarian cancer patients, NK cells in the ascites represent the largest proportion of CD45+ leukocytes [39]. Despite this numerical dominance, it fails to confer effective anti-tumor activity and are instead characterized by severely impaired function. This dysfunction results from the concerted action of various factors, including the abnormally elevated levels of cytokines such as TGF-β, certain subset of interleukins (IL), prostaglandin E2 (PGE2), and vascular endothelial growth factor (VEGF) [40], as well as the infiltration of immunosuppressive cell populations such as myeloid-derived suppressor cells (MDSCs) and regulatory T cells (Tregs) along with their inhibitory secretions [40, 41].
Among these factors, TGF-β has been widely documented for its inhibitory effect on NK cells [42, 43]. Elevated TGF-β1 levels in ovarian cancer ascites correlate with NK cell dysfunction and reduced patient progression-free survival (PFS) and OS [44]. Additionally, NK cells differentiated from umbilical cord blood CD34+ hematopoietic progenitor cells (HPC-NK) exhibit greater resistance to ascites-mediated suppression than peripheral blood NK cells (PB-NK) from healthy donors, suggesting the therapeutic potential of HPC-NK in adoptive cell therapy. Building on these observations, researchers confirmed that knockout of SMAD4, a key transcriptional regulator in the TGFβ signaling pathway, enables NK cells to maintain potent anti-tumor activity in TGFβ-enriched TME [45], and is applicable to various NK cell-based platforms such as anti-CD19-CAR-NK, HPC-NK (GTA002), and adaptive NK cell products (ADAPT-NK). Moreover, the bifunctional fusion protein Bintrafusp Alfa (BA), which simultaneously targets TGF-β and PD-L1, effectively clears TGF-β and VEGF from ascites and enhances cytolytic NK cell activity in ovarian cancer preclinical models [43], supporting BA as a promising new immunotherapeutic strategy for ovarian cancer.
PGE2 is identified as an immunomodulatory molecule synthesized by tumor cells via COX-1/COX-2, binds to EP2 and EP4 receptors on NK cells and directly impairing NK cell early tumor infiltration, intercellular contact, and IFN-γ production. This disruption hinders NK cell-driven polarization of tumor-associated macrophages (TAMs) toward classical inflammatory phenotype and CD8+ T cell activation [46]. Crucially, the accumulation of immunosuppressive cells such as TAMs, CAFs, and MDSCs [47] within the TME establishes intricate crosstalk networks that synergistically impact NK cells, ultimately leading to persistent NK cell dysfunction [48, 49]. Recent research has focused on a 'decoy' mechanism between CAFs and NK cells, in which CAFs continuously engage NK cells by upregulating multiple ligands for activating receptors NKG2D and DNAM-1, such as NECTIN2, PVR/CD155, and RAE-1. This interaction triggers the internalization and downregulation of activating receptors on NK cells, markedly impairing their capacity to recognize and eliminate cancer cells [50]. Another recent study revealed that in a pancreatic cancer model, nociceptor neurons release calcitonin gene-related peptide (CGRP), which binds to receptor activity-modifying protein 1 (RAMP1) on CAFs, inhibiting IL-15 secretion by CAFs. This suppression impairs NK cell infiltration and cytotoxicity, thereby promoting tumor progression and cancer pain [51]. The FDA-approved CGRP receptor antagonist Rimegepant reversed this effect [51], offering a paradigm for similar mechanistic investigations in ovarian cancer [52].
Collectively, these findings emphasize that a comprehensive understanding of how immunosuppressive cytokines and related cellular networks synergistically suppress NK cells in ovarian cancer TME, along with elucidation of the underlying mechanisms, holds significant translational value for developing novel therapeutic strategies.
3.3 Metabolic reprogramming
Tumor cells undergo significant metabolic reprogramming to meet their rapid proliferation and survival needs, and among all identified cancer metabolic reprogramming patterns, the Warburg effect is undoubtedly the most representative [53]. One of the most direct consequences of the Warburg effect is the accumulation of large amounts of lactate in the TME. Elevated lactate levels in the TME directly impede NK cell cytotoxicity by inducing lysine lactylation (Kla) or indirectly by increasing the abundance of MDSCs [54]. Furthermore, lactate accumulation is a critical factor that transcriptionally suppresses NAMPT expression in NK cells, leading to NAD⁺ metabolic imbalance and ultimately inhibiting their anti-tumor efficacy [55].
A common metabolic alteration in the TME is lipid accumulation, which is strongly associated with immune dysfunction. Recent investigations have revealed that NK cells massively uptake polar lipids, particularly phosphatidylcholine PC(36:1), from ovarian cancer ascites through the scavenger receptor SR-B1 [39]. This aberrant lipid accumulation compromises the integrity of NK cell plasma membranes and interferes with the reduction of neutral lipid droplet levels, subsequently damages mitochondrial function, resulted in a comprehensive impairment of cytotoxic function. Pharmacological blockade of SR-B1 or lipid depletion approaches can partially ameliorate these metabolic deficiencies [39], underscoring the potential therapeutic utility of targeting lipid metabolism in NK cell immunotherapy.
Indoleamine 2,3-dioxygenase (IDO), the rate-limiting enzyme in tryptophan metabolism, produces kynurenine (KYN), which is often elevated in the TME of various tumors. Research suggests that Kyn activates aryl hydrocarbon receptor (AhR), a transcription factor that binds to the xenobiotic-responsive element (XRE) site on the ADAM10 promoter, thereby upregulating ADAM10 transcription and expression. This process enhances the shedding of NKG2D ligands (NKG2DLs) from tumor cell, reducing membrane-bound NKG2DLs while increasing soluble NKG2DL release [56]. Consequently, this impairs NK cell recognition and binding of tumor cells via NKG2D receptors [56]. Kyn can also induce ferroptosis in NK cells through AhR-independent pathways [57], substantially aggravating both the depletion of NK cells and their functional impairment within the TME. While several IDO1 inhibitors have advanced to clinical trials, results from a phase III trial (ECHO-301/KEYNOTE-252) showed no additional clinical benefit when combined with anti-PD-1 therapy. However, researchers have since developed NTRC 3883-0, an IDO1 inhibitor capable of inhibiting tryptophan metabolism in interferon-γ (IFN-γ)-stimulated primary ovarian cancer cells [58].
Recent proteomic analyses of ovarian cancer ascites have revealed that approximately 50% of patients exhibit higher total iron levels in ascites than in healthy human serum [59], suggesting that ascites may serve as a localized iron source for iron-dependent ovarian cancer cells. Further research has confirmed that the FDA-approved iron chelator deferoxamine alters mitochondrial integrity, leading to the release of mtDNA into cytoplasm, activation of the cGAS-STING-IRF3 pathway, and increased type I interferon (IFN-I) release. This cascade subsequently promotes IL-15 secretion by tumor-associated dendritic cells (tDCs), thereby enhancing NK cell infiltration and cytotoxic function [59]. These findings strongly demonstrate that elucidating and targeting the metabolic dysregulation features of ovarian cancer represents a viable strategy to reverse immunosuppression and unleash the potential of NK cell-based immunity.
Researchers have implemented targeted metabolic reprogramming of NK cells during ex vivo expansion to foster intrinsic resilience against metabolic suppression within the TME. A primary exemplar of this strategy is METR-NK, a metabolically programmed NK cell product that integrates specific metabolic modulators during manufacturing to delay cellular senescence and bolster cytotoxicity. Currently, METR-NK is being evaluated for advanced epithelial ovarian cancer both as a neoadjuvant intraperitoneal therapy (NCT06395844) and in combination with Solantinib (NCT06884345), to leverage the therapeutic synergy between metabolic fitness and anti-angiogenic targeting.
4. Therapeutic strategies to enhance NK cell function
Adoptive cell therapy (ACT), which involves ex-vivo selection, activation, and large-scale expansion of immune effector cells with anti-tumor activity followed by reinfusion into the patient, has emerged as a major strategy in oncology. Among these, NK cells are regarded as highly promising effector cells due to their antigen-independent recognition, broad target range, and low risk of graft-versus-host disease (GVHD), showing potential in both hematologic malignancies and solid tumors. Several clinical trials have recently assessed the safety and efficacy of NK cell-based ACT in ovarian cancer. (Table 1).
Novel NK cell-based immunotherapy clinical trials for ovarian cancer.
| NCT Number | Study Title | Phase (status) | Conditions | Interventions | Cell Source |
|---|---|---|---|---|---|
| Non-genetically modified NK cells | |||||
| NCT06321484 | Intraperitoneal Cytokine-Induced Memory Like (CIML) Natural Killer (NK) Cells in Recurrent Ovarian Cancer | Phase I (Recruiting) | Platinum-resistant Ovarian Cancer Recurrent Ovary Cancer Ovarian Cancer | BIOLOGICAL: CIML NK Cells DRUG: IL-2 | Peripheral Blood |
| NCT06710288 | A Phase 2, Open-label, Single-arm Study of Autologous M-CENK Adoptive Cell Therapy And N-803 (IL-15 Superagonist) In Combination with Gemcitabine in Participants with Recurrent Platinum-Resistant High-Grade Ovarian Cancer | Phase II (Recruiting) | Platinum-resistant Ovarian Cancer | DRUG: Gemcitabine BIOLOGICAL: N-803 BIOLOGICAL: M-CENK | Peripheral Blood |
| NCT06395844 | Safety and Efficacy of Intraperitoneal Injection of METR-NK Cells as Neoadjuvant Therapy for Advanced Epithelial Ovarian Cancer | Phase I /II (Recruiting) | Ovarian Cancer | DRUG: METR-NK Cells | Peripheral Blood |
| NCT06884345 | METR-NK Cells in Combination with Anti-angiogenic Neoadjuvant Therapy for Advanced Epithelial Ovarian Cancer | Phase I /II (Active, not recruiting) | Ovarian Cancer | DRUG: METR-NK Cells combined with Solvatinib | Peripheral Blood |
| NCT03213964 | Intraperitoneal Delivery of Adaptive Natural Killer (NK) Cells (FATE-NK100) With Intraperitoneal Int | Phase I (Completed) | Epithelial Ovarian Cancer Fallopian Tube Cancer Primary Peritoneal Cancer | BIOLOGICAL: FATE-NK100 DRUG: IL-2 | Peripheral Blood |
| Engineered NK cells | |||||
| NCT06342986 | Intraperitoneal FT536 in Recurrent Ovarian, Fallopian Tube, and Primary Peritoneal Cancer | Phase I (Recruiting) | Gynecologic Cancer Ovarian Cancer Fallopian Tube Cancer Primary Peritoneal Cavity Cancer | DRUG: FT536 DRUG: Fludarabine DRUG: CY | iPSC |
| NCT03692637 | Study of Anti-Mesothelin Car NK Cells in Epithelial Ovarian Cancer | Early Phase I (Unknown) | Epithelial Ovarian Cancer | BIOLOGICAL: anti-Mesothelin CAR-NK Cells | Peripheral Blood |
| NCT05410717 | CLDN6/GPC3/Mesothelin/AXL-CAR-NK Cell Therapy for Advanced Solid Tumors | Phase I (Recruiting) | Stage IV Ovarian Cancer Refractory Testis Cancer Recurrent Endometrial Cancer | BIOLOGICAL: Claudin6 CAR-NK cells BIOLOGICAL: GPC3 CAR-NK cells BIOLOGICAL: Mesothelin CAR-NK cells BIOLOGICAL: AXL CAR-NK cells | Peripheral Blood |
| NCT05856643 | Single-arm, Open-label Clinical Study of SZ011 in the Treatment of Ovarian Epithelial Carcinoma | Early Phase I (Recruiting) | Epithelial Ovarian Cancer | DRUG: SZ011 CAR-NK | Unknown |
| NCT05776355 | NKG2D CAR-NK & Ovarian Cancer | Unknown | Ovarian Cancer | Biological: NKG2D CAR-NK | Unknown |
| NCT05922930 | Study of TROP2 CAR Engineered IL15-transduced Cord Blood-derived NK Cells Delivered Intraperitoneally for the Management of Platinum Resistant Ovarian Cancer, Mesonephric-like Adenocarcinoma, and Pancreatic Cancer | Phase I /II (Recruiting) | Ovarian Cancer Pancreatic Cancer Adenocarcinoma | DRUG: TROP2-CAR-NK DRUG: Cyclophosphamide DRUG: Fludarabine | Cord Blood |
Abbreviations: CAR: Chimeric Antigen Receptor, CIML: Cytokine-Induced Memory Like, CLDN6: Claudin 6, CY: Cyclophosphamide, GPC3: Glypican-3, IL-15: Interleukin-15, IL-2: Interleukin-2, iPSC: Induced Pluripotent Stem Cells, METR: Metabolic Remodeling, NK: Natural Killer, NKG2D: Natural Killer Group 2D, TROP2: Trophoblast Cell-Surface Antigen 2.
4.1 Cytokine-based agents
As a direct and efficient strategy, cytokine-based agents can bypass the complex immunosuppressive network within the TME and rapidly activate NK cell cytotoxicity through exogenous delivery of high-concentration cytokines. This approach is especially suitable for the CD56bright NK cell subset, which expresses high levels of cytokine receptors [14]. A primary objective of current research is to generate CIML NK cells through in-vitro activation with IL-2, IL-15, and IL-18 [60, 61]. These cells exhibit enhanced IFN-γ production and cytotoxicity against ovarian cancer [61]. A study combining assay for transposase-accessible chromatin sequencing (ATAC-seq), cellular indexing of transcriptomes and epitopes by sequencing (CITE-seq), and single-cell RNA sequencing (scRNA-seq) revealed that IL-12/15/18 rapidly induces epigenetic remodeling in human NK cells, forming enriched memory-like (eML) NK cells and effector conventional NK (effcNK) cells [62], with significantly increased chromatin accessibility in the IFNG promoter region of eML NK cells. This work advances our understanding of CIML NK cell heterogeneity and suggests that personalized immunotherapeutic strategies could be developed based on their distinct phenotypes in the future. Preliminary findings from an investigator-initiated Phase 1b trial (NCT06321484) evaluated the safety and tolerability of intraperitoneally (i.p.) delivered autologous CIML-NK cells in patients with platinum-resistant ovarian cancer (PROC) [63]. Initial results from the first two patients demonstrated that autologous CIML-NK products of sufficient cell number were successfully generated following a 6-day culture in IL-2-supplemented media, and their i.p. administration was well-tolerated with no reported adverse events.
Adoptive therapies based on allogeneic cytokine-activated NK cells are being investigated clinically (Figure 2). The IL-15 superagonist N-803, which combines IL-15 with an activating mutation, an IL-15Rα sushi domain for trans-presentation, and an IgG1-Fc fragment to extend its half-life, overcomes the short half-life limitation of conventional cytokines [64, 65]. It stimulates HPC-NK cell expansion, IFN-γ secretion, and cytotoxicity against leukemia and ovarian cancer cells. Preliminary results from a phase II clinical trial (NCT03054909) have been reported for N-803 as maintenance therapy in advanced ovarian cancers. Preclinical investigations also suggest that combining N-803 with Avelumab significantly reduces ovarian cancer tumor burden [66].
Potential strategies for enhancing NK cell therapy in ovarian cancer. (A) Cytokine Modulation for NK Cell Enhancement: This strategy leverages specific cytokines and chemokine receptor engineering (e.g., N-803, CIML-NK cell) to boost NK cell activity, proliferation, and tumor targeting. Engineering NK cells with chemokine receptors like CXCR2/CXCR4 improves their migration and infiltration into tumor sites. (B) NKCEs are engineered to guide NK cells to tumor cells, establishing an immune complex by engaging both tumor-specific antigens and NK cell activating receptors (e.g., CD16, NKG2D, NKp46). (C) Chimeric antigen receptor NK cells: 1) CAR design has evolved significantly: first-generation CARs feature an extracellular antigen-binding domain (scFv) linked to an intracellular CD3ζ signaling domain; second-generation CARs incorporate an additional costimulatory domain (e.g., CD28, 4-1BB) to CD3ζ for improved activation and persistence; third-generation CARs further enhance signaling by including two costimulatory domains. 2) To boost their fitness and longevity in the tumor microenvironment, NK cells can be armored with cytokines. 3) Other strategies like TCR-complex engineering can enable recognition of intracellular tumor peptides or HLA-independent antigens, respectively. 4) Advanced CAR designs facilitate multi-antigen targeting to enhance tumor coverage and mitigate antigen escape.
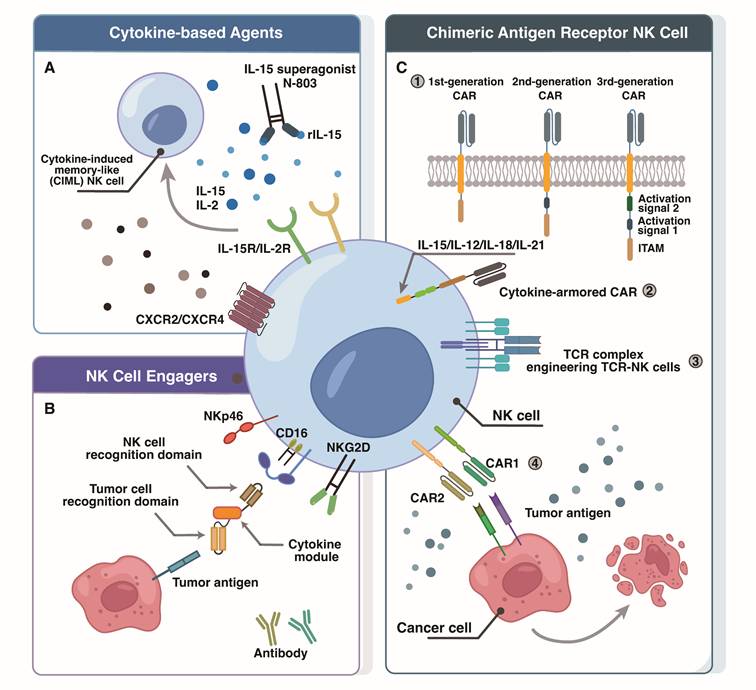
Moreover, compared to IL-2, N-803 enhances NK cell and CD8⁺ T-cell function while avoiding the common IL-2-mediated expansion of Tregs. Another novel engineered IL-2 variant, Nemvaleukin alfa, was evaluated in the clinical trial ARTISTRY-1 (NCT02799095). It selectively binds to intermediate-affinity IL-2 receptors, preferentially activating and expanding NK cells and CD8⁺ T cells while minimizing Treg proliferation. With a low rate of treatment discontinuation due to treatment-related adverse events (TRAEs), it demonstrates a manageable safety profile [67]. An exploratory analysis in the ARTISTRY-1 study included 14 evaluable patients with platinum-resistant ovarian cancer who received nemvaleukin alfa combined with pembrolizumab, resulting in an objective response rate (ORR) of 21% [67]. These findings prompted the launch of the phase III multicenter trial (ARTISTRY-7, NCT05092360), which will evaluate the efficacy of this combination compared to chemotherapy in patients with ovarian cancer.
Beyond standard cytokine priming, FATE-NK100 represents a novel adaptive NK cell therapy modulated with IL-15 and a glycogen synthase kinase 3β (GSK3β) inhibitor. In the phase I trial, nine patients were treated, and no dose-limiting toxicities (DLTs) were observed [68]. Given the clinical benefit, three patients (33%) underwent retreatment, including one who achieved a partial remission (48% tumor reduction). Functional assays of in vivo samples confirmed that these FATE-NK100 cells persisted in the peritoneal cavity for up to 21 days, exhibiting superior in vivo function compared to the patient's endogenous NK cells (NCT03213964) [69].
4.2 NK cell engagers
From a long-term treatment perspective, cytokine-based agents may inevitably induce cytokine release syndrome (CRS) in the complex TME and have limited target specificity. In contrast, antibody-dependent NK Cell Engagers (NKCEs) can specifically recognize and bind to tumor-associated antigens (TAAs) on tumor cells and NK cell activating receptors, displaying activity only in the presence of tumor cells [70-72]. Many NKCEs target CD16a on the NK cell, a receptor naturally responsible for recognizes antibody Fc fragments (Fcγ) and mediates ADCC. These NKCEs are therefore engineered to actively and efficiently bridge NK cells with tumor cells, achieving enhanced, redirected ADCC [73].
The conventional human epidermal growth factor receptor 2 (HER2)-targeting monoclonal antibody Trastuzumab has low affinity between its Fc region and the CD16a receptor on NK cells, and it readily cross-reacts with other Fcγ receptors (e.g., CD16b, CD32b), resulting in suboptimal ADCC effects. To overcome this limitation, researchers developed a novel bispecific killer cell engager, BiKE:HER2/CD16a (BiKE:E5C1), which uses high-affinity anti-CD16a and anti-HER2 VHH to precisely bridge NK cells to HER2+ ovarian cancer cells [74, 75]. In vitro, BiKE:E5C1 enhanced NK cell activation, increasing the release of perforin, granzyme B (GZMB), TNF-α, and IFN-, and demonstrated approximately 100-fold greater potency than trastuzumab. In a humanized mouse model of ovarian cancer, BiKE-E5C1 combined with laNK92 cells eradicated metastatic HER2+ tumors [76]. Another Tribody, [(HER2) ₂xCD16], which contains two HER2-binding domains, is capable of induce the lysis of HER2+ ovarian tumor cells even with minimal infiltration of NK and γδ T cells [77]. Such a framework can be extended to the design of trispecific NK cell engagers (TriKEs), for example by co-targeting multiple activating receptors on NK cells or by fusing them with cytokines to achieve synergistic signal activation and functional support [78, 79].
A recent phase I study (NCT04259450) reported results for AFM24 that among 35 patients with advanced solid tumors, 10 (approximately 28.6%) achieved stable disease [80], which is a positive signal for advanced patients who have received multi-line therapy.
4.3 CAR-NK cells
While NKCEs efficiently augment ADCC by bridging TAAs to NK cell receptors, their overall efficacy is largely limited by tumor antigen density, heterogeneity, and the persistence of infused NK cells in vivo. Therefore, chimeric antigen receptor (CAR)-NK cells, which are genetically engineered to directly recognize tumor antigens, have emerged as a highly promising advancement in NK cell-based immunotherapy [81]. CAR-NK therapy can utilize MHC-mismatched allogeneic NK cells, thereby reducing the risks of CRS and GVHD [82]. Although most CAR-NK research remains in preclinical or early-phase clinical trials, their favorable safety and initial efficacy profiles have been widely recognized.
CAR-NK cell products derive from diverse sources, such as NK92 cell lines, induced pluripotent stem cell (iPSC)-derived NK cells, CD34⁺ hematopoietic stem and progenitor cells (HSPCs) from umbilical cord blood (CB), and peripheral blood-derived NK (PB-NK) cells. To facilitate "off-the-shelf" therapy, iPSC-derived NK cells are a major focus of current research [83-86]. Beyond addressing the extended preparation times, high costs of autologous therapies, and the donor-to-donor variability of peripheral blood NK cells, the absence of DLTs in the first patient treated with GPC3-targeted iPSC-derived CAR-NK cells (jRCT2033200431) further supports the clinical feasibility of this standardized, mass-producible platform [87].
The design of CAR-NK cells builds on the framework established for CAR-T cells. Currently, second and third-generation CAR-NK cells, building on the first-generation CAR-NK cells that only contained a CD3ζ activation signal, additionally integrate costimulatory domains [88, 89], such as the CD28 costimulatory domain. This domain recruits the intrinsically expressed Src family kinases (e.g., LCK) via its PYAP motif, subsequently phosphorylating CD3ζ and activating ZAP70 to enhance NK activation signals [90]. In ovarian cancer, an NK-CAR construct comprising NKG2D-2B4ζ expressed in iPSC-NK cells was shown to markedly improve cytotoxicity against antigen-positive ovarian cancer targets [91]. Similarly, a third-generation CAR containing costimulatory domains (CD28 and 4-1BB) along with a CD3ζ signaling domain was introduced into NK-92 cells. In vitro assays confirmed that these engineered NK-92 cells exhibited significantly enhanced killing of CD24high SKOV3 and OVCAR3 ovarian cancer cells [92].
Selection of appropriate tumor-associated antigens (TAAs) is critical to achieving precise targeting and minimizing off-tumor toxicity. Several TAAs are currently under investigation as CAR-NK targets in ovarian cancer, including mesothelin (MSLN) [93, 94], CD24 [92], HER2 [95], α-folate receptor (αFR) [96], Claudin-6 [97], CD44 [98], and glypican-3 (GPC3) [99].
Nevertheless, inadequate cytokine and chemokine support within the ovarian TME severely limits CAR-NK cell tumor infiltration and in vivo persistence, posing a major translational challenge for solid tumor therapy. Recent studies have explored strategies such as cytokine armoring and chemokine receptor engineering. A study revealed that in CAR-NK cells armed with engineered expression of the IL-2Rβγ agonist Neo-2/15 [100], Neo-2/15 can continuously and stably activate the STAT5/Akt signaling pathway, driving persistent high expression of the transcription factor c-Myc. c-Myc directly upregulates glucose and glutamine transporter expression, enhancing the metabolic competitiveness of cells in nutrient-deprived TME, while activating the endoplasmic reticulum (ER) stress sensor IRE1α to promote XBP1s splicing and nuclear translocation. Nuclear XBP1s subsequently upregulates nuclear respiratory factor 1 (NRF1), maintaining mitochondrial integrity, upregulating anti-apoptotic proteins, and suppressing ER stress-induced apoptosis. Although the enhancement mechanism in Neo-2/15-armed CAR-NK cells differs from that in cytokine-pretreated MSLN-CAR CIML NK cells [93], both approaches similarly improve in vivo persistence and anti-tumor efficacy [93, 100]. Another study focused on chemokine receptor engineering. Observing that IFN-γ and TNF-α produced during NK-92-αFR-CAR-mediated killing of ovarian cancer cells induce CXCL10 via JAK1/2-STAT1 and NF-κB pathways [96], researchers engineered NK-92 cells to co-express CXCR3A and αFR-CAR (termed NK-92-αFR-CAR-CXCR3A). This modification successfully enhanced the migration and tumor-infiltrating abilities of the CAR-NK cells [96].
The optimization of CAR-NK adoptive therapy has advanced through diverse strategies [81], including multi-antigen targeting, chemokine receptor engineering, cytokine armoring, transcriptional remodeling, and the development of TCR-engineered NK cells. Subsequent research will focus on identifying rational combinations and optimal timing for incorporating CAR-NK cell therapy with established or emerging treatments (e.g., oncolytic viruses [101, 102], chemotherapy, immune checkpoint inhibitors, or immunomodulatory drugs [103]), to advance CAR-NK cell therapies for solid tumors into clinical practice.
5. Concluding remarks
NK cell therapy has demonstrated remarkable preclinical potential in ovarian cancer. As a central effector cell of innate immunity, NK cells can directly eliminate tumor cells and broadly enhance adaptive immune responses [21, 22]. Therefore, therapeutic approaches that efficiently leverage these innate functions have emerged as crucial complements to conventional treatments and are vital for boosting the efficacy of combination immunotherapies. By combining NK cell-based strategies with chemotherapy, immune checkpoint inhibitors, or oncolytic viruses [104], thereby enhancing anti-tumor activity and in vivo persistence, they represent a transformative avenue for improving outcomes in ovarian cancer patients.
However, the clinical application of ovarian cancer NK cell therapies remains hindered by TME-mediated suppression of infiltration and activity, coupled with the donor heterogeneity and prohibitive costs of conventional cell sources. In light of this, the development of next-generation NK cell platforms is pivoting toward industrialization and precision, centered on iPSC technology and targeted gene editing. Through the establishment of master cell banks from a single, rigorously characterized clone, ensuring a uniform genetic background and consistent functional potency across production lots, the iPSC-derived NK cell platform provides the unparalleled scalability and clonal consistency required for standardized, off-the-shelf immunotherapy [105]. Furthermore, leveraging the high gene-editing efficiency of iPSCs, researchers can precisely and stably engineer multiple functional enhancements. For instance, through the co-expressing of a high-affinity non-cleavable CD16a and a membrane-bound IL-15/IL-15R fusion protein, combined with the knockout of the CD38, triple gene-edited iPSC-derived NK cells (termed iADAPT NK) can persist and function in vivo in the absence of exogenous cytokine support [86]. Alternatively, the deletion of the negative regulator CISH significantly enhances the sensitivity of iPSC-NK cells to low-dose IL-15 and promotes mTOR-mediated metabolic fitness, thereby substantially improving their in vivo persistence and anti-tumor activity [106].
In conclusion, future research should prioritize a comprehensive elucidation of the complex interactions between NK cells and the TME, employing multi-omics approaches such as single-cell transcriptomics, spatial transcriptomics, and metabolomics. This foundational insight will be critical for refining therapeutic target selection, optimizing in vitro expansion and in vivo delivery platforms, and advancing personalized combination approaches that integrate NK cell-based therapy with other modalities. By synthesizing current advances and delineating future directions, this review seeks to support and advance the refinement of more effective NK cell-based immunotherapies for ovarian cancer.
Abbreviations
ACT: Adoptive cell therapy; ADAPT-NK: adaptive natural killer cell products; ADCC: antibody-dependent cellular cytotoxicity; AhR: aryl hydrocarbon receptor; ATAC-seq: assay for transposase-accessible chromatin sequencing; BA: Bintrafusp Alfa; BiKE: bispecific killer cell engager; CAR: chimeric antigen receptor; CAR-NK: chimeric antigen receptor natural killer cells; CB: cord blood; CCL5: chemokine (C-C motif) ligand 5; CIML: cytokine-induced memory-like; CIN: chromosomal instability; CITE-seq: cellular indexing of transcriptomes and epitopes by sequencing; COX-1/COX-2: cyclooxygenase-1/-2; CRS: cytokine release syndrome; DLTs: dose-limiting toxicities; eML: enriched memory-like; effcNK: effector conventional natural killer cells; EGFR: epidermal growth factor receptor; FAK: focal adhesion kinase; FDA: Food and Drug Administration; GM-CSF: granulocyte-macrophage colony-stimulating factor; GPC3: glypican-3; GSK3β: glycogen synthase kinase 3β; GVHD: graft-versus-host disease; HCMV: human cytomegalovirus; HER2: human epidermal growth factor receptor 2; HGSOC: high-grade serous ovarian carcinoma; HPC-NK: hematopoietic progenitor cell-derived natural killer cells; HSPCs: hematopoietic stem and progenitor cells; IDO: indoleamine 2,3-dioxygenase; IFN-I: type I interferon; IFN-γ: interferon-γ; IL: interleukins; IL-12: interleukin-12; IL-15: interleukin-15; IL-18: interleukin-18; IL-2: interleukin-2; iPSC: induced pluripotent stem cell; KIRs: killer cell immunoglobulin-like receptors; Kla: lysine lactylation; KYN: kynurenine; MA: malignant ascites; MDSCs: myeloid-derived suppressor cells; MHC-I: major histocompatibility complex class I; MICA/B: MHC class I-related stress-induced antigens; MSLN: mesothelin; NCRs: natural cytotoxicity receptors; NKG2A: NK group 2 member A; NKG2D: NK group 2 member D; NKG2DLs: NKG2D ligands; NKCEs: NK Cell Engagers; NK cells: Natural Killer cells; ORR: objective response rate; OS: overall survival; PB-NK: peripheral blood natural killer; PD-L1: programmed cell death-ligand 1; PFS: progression-free survival; PGE2: prostaglandin E2; PROC: platinum-resistant ovarian cancer; RAMP1: receptor activity-modifying protein 1; scRNA-seq: single-cell RNA sequencing; SR-B1: scavenger receptor B1; TAAs: tumor-associated antigens; TAMs: tumor-associated macrophages; TGF-β: transforming growth factor-beta; TME: tumor microenvironment; TNF-α: tumor necrosis factor-α; TRAIL: TNF-related apoptosis-inducing ligand; TRAEs: treatment-related adverse events; Tregs: regulatory T cells; TriKEs: trispecific NK cell engagers; tDCs: tumor-associated dendritic cells; VEGF: vascular endothelial growth factor; WT1: Wilms' tumor antigen 1; XRE: xenobiotic-responsive element; αFR: α-folate receptor.
Funding
This work was supported by the National Natural Science Foundation of China (82273205, 82203665, 82473274, 82503116, 82503386), Natural Science Foundation of Shanghai (23ZR1408300) and Shanghai Science and Technology Innovation Action Plan (23Y11909500, 23Y11901800).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Webb PM, Jordan SJ. Global epidemiology of epithelial ovarian cancer. Nat Rev Clin Oncol. 2024;21:389-400
2. Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10-45
3. Konstantinopoulos PA, Matulonis UA. Clinical and translational advances in ovarian cancer therapy. Nat Cancer. 2023;4:1239-57
4. Ghisoni E, Morotti M, Sarivalasis A, Grimm AJ, Kandalaft L, Laniti DD. et al. Immunotherapy for ovarian cancer: towards a tailored immunophenotype-based approach. Nat Rev Clin Oncol. 2024;21:801-17
5. Kandalaft LE, Dangaj Laniti D, Coukos G. Immunobiology of high-grade serous ovarian cancer: lessons for clinical translation. Nat Rev Cancer. 2022;22:640-56
6. Wu SY, Fu T, Jiang YZ, Shao ZM. Natural killer cells in cancer biology and therapy. Mol Cancer. 2020;19:120
7. Jacquelot N, Seillet C, Vivier E, Belz GT. Innate lymphoid cells and cancer. Nat Immunol. 2022;23:371-9
8. Zorko NA, Makovec A, Elliott A, Kellen S, Lozada JR, Arafa AT. et al. Natural killer cell infiltration in prostate cancers predicts improved patient outcomes. Prostate Cancer Prostatic Dis. 2025;28:129-37
9. Cozar B, Greppi M, Carpentier S, Narni-Mancinelli E, Chiossone L, Vivier E. Tumor-infiltrating natural killer cells. Cancer Discov. 2021;11:34-44
10. Myers JA, Miller JS. Exploring the NK cell platform for cancer immunotherapy. Nat Rev Clin Oncol. 2021;18:85-100
11. Seymour F, Cavenagh JD, Mathews J, Gribben JG. NK cells CD56bright and CD56dim subset cytokine loss and exhaustion is associated with impaired survival in myeloma. Blood Adv. 2022;6:5152-9
12. Tang F, Li J, Qi L, Liu D, Bo Y, Qin S. et al. A pan-cancer single-cell panorama of human natural killer cells. Cell. 2023;186:4235-51.e20
13. Netskar H, Pfefferle A, Goodridge JP, Sohlberg E, Dufva O, Teichmann SA. et al. Pan-cancer profiling of tumor-infiltrating natural killer cells through transcriptional reference mapping. Nat Immunol. 2024;25:1445-59
14. Rebuffet L, Melsen JE, Escalière B, Basurto-Lozada D, Bhandoola A, Björkström NK. et al. High-dimensional single-cell analysis of human natural killer cell heterogeneity. Nat Immunol. 2024;25:1474-88
15. Dean I, Lee CYC, Tuong ZK, Li Z, Tibbitt CA, Willis C. et al. Rapid functional impairment of natural killer cells following tumor entry limits anti-tumor immunity. Nat Commun. 2024;15:683
16. Mariuzza RA, Singh P, Karade SS, Shahid S, Sharma VK. Recognition of self and viral ligands by NK cell receptors. Immunol Rev. 2025;329:e13435
17. Li Y, Li Z, Tang Y, Zhuang X, Feng W, Boor PPC. et al. Unlocking the therapeutic potential of the NKG2A-HLA-E immune checkpoint pathway in T cells and NK cells for cancer immunotherapy. J Immunother Cancer. 2024;12:e009934
18. Chovatiya N, Kaur K, Huerta-Yepez S, Chen PC, Neal A, DiBernardo G. et al. Inability of ovarian cancers to upregulate their MHC-class I surface expression marks their aggressiveness and increased susceptibility to NK cell-mediated cytotoxicity. Cancer Immunol Immunother. 2022;71:2929-41
19. Zhu H, Blum RH, Bjordahl R, Gaidarova S, Rogers P, Lee TT. et al. Pluripotent stem cell-derived NK cells with high-affinity noncleavable CD16a mediate improved antitumor activity. Blood. 2020;135:399-410
20. Maskalenko NA, Zahroun S, Tsygankova O, Anikeeva N, Sykulev Y, Campbell KS. The FcγRIIIA (CD16) L48-H/R polymorphism enhances NK cell-mediated antibody-dependent cellular cytotoxicity by promoting serial killing. Cancer Immunol Res. 2025;13:417-29
21. Kader T, Lin JR, Hug CB, Coy S, Chen YA, de Bruijn I. et al. Multimodal spatial profiling reveals immune suppression and microenvironment remodeling in fallopian tube precursors to high-grade serous ovarian carcinoma. Cancer Discov. 2025;15:1180-202
22. Böttcher JP, Bonavita E, Chakravarty P, Blees H, Cabeza-Cabrerizo M, Sammicheli S. et al. NK cells stimulate recruitment of cDC1 into the tumor microenvironment promoting cancer immune control. Cell. 2018;172:1022-37.e14
23. Barry KC, Hsu J, Broz ML, Cueto FJ, Binnewies M, Combes AJ. et al. A natural killer-dendritic cell axis defines checkpoint therapy-responsive tumor microenvironments. Nat Med. 2018;24:1178-91
24. Avanessian SC, van den Bijgaart RJE, Chew NW, Supper VM, Tang TT, Zhang Y. et al. IL-2/IL-15 signaling induces NK cell production of FLT3LG augmenting anti-PD-1 immunotherapy. Cancer Immunol Res. 2026;14:122-38
25. Maskalenko NA, Zhigarev D, Campbell KS. Harnessing natural killer cells for cancer immunotherapy: dispatching the first responders. Nat Rev Drug Discov. 2022;21:559-77
26. Hou X, Wang C, Zhong Y, Wang L, Kang DD, Lubitz G. et al. Enhancing antitumor immunity through chemotherapeutic-derived lipid nanoparticle-induced immunogenic cell death and CD40L/Flt3L mRNA-mediated dendritic cell activation. J Control Release. 2025;382:113684
27. Mittal D, Vijayan D, Putz EM, Aguilera AR, Markey KA, Straube J. et al. Interleukin-12 from CD103⁺ Batf3-dependent dendritic cells required for NK cell suppression of metastasis. Cancer Immunol Res. 2017;5:1098-108
28. Nagai H, Karube R. Late-stage ovarian cancer with systemic multiple metastases shows marked shrinkage using a combination of Wilms' tumor antigen 1 (WT1) dendritic cell vaccine, natural killer (NK) cell therapy, and nivolumab. Cureus. 2024;16:e56685
29. Kumar P, Ranmale S, Mehta S, Tongaonkar H, Patel V, Singh AK. et al. Immune profile of primary and recurrent epithelial ovarian cancer cases indicates immune suppression, a major cause of progression and relapse of ovarian cancer. J Ovarian Res. 2023;16:114
30. Ferrari de Andrade L, Tay RE, Pan D, Luoma AM, Ito Y, Badrinath S. et al. Antibody-mediated inhibition of MICA and MICB shedding promotes NK cell-driven tumor immunity. Science. 2018;359:1537-42
31. Maas RJ, Hoogstad-van Evert JS, Van der Meer JM, Mekers V, Rezaeifard S, Korman AJ. et al. TIGIT blockade enhances functionality of peritoneal NK cells with altered expression of DNAM-1/TIGIT/CD96 checkpoint molecules in ovarian cancer. Oncoimmunology. 2020;9:1843247
32. Ozmadenci D, Shankara Narayanan JS, Andrew J, Ojalill M, Barrie AM, Jiang S. et al. Tumor FAK orchestrates immunosuppression in ovarian cancer via the CD155/TIGIT axis. Proc Natl Acad Sci U S A. 2022;119:e2117065119
33. Ferrari de Andrade L, Kumar S, Luoma AM, Ito Y, Alves da Silva PH, Pan D. et al. Inhibition of MICA and MICB shedding elicits NK cell-mediated immunity against tumors resistant to cytotoxic T cells. Cancer Immunol Res. 2020;8:769-80
34. Whalen KA, Henry CC, Mehta NK, Rakhra K, Yalcin S, Meetze K. et al. CLN-619, a MICA/B monoclonal antibody that promotes innate immune cell-mediated antitumor activity. J Immunother Cancer. 2025;13:e008987
35. Wan C, Keany MP, Dong H, Al-Alem LF, Pandya UM, Lazo S. et al. Enhanced efficacy of simultaneous PD-1 and PD-L1 immune checkpoint blockade in high-grade serous ovarian cancer. Cancer Res. 2021;81:158-73
36. Liu WN, Harden SL, Tan SLW, Tan RJR, Fong SY, Tan SY. et al. Single-cell RNA sequencing reveals anti-tumor potency of CD56⁺ NK cells and CD8⁺ T cells in humanized mice via PD-1 and TIGIT co-targeting. Mol Ther. 2024;32:3895-914
37. Ford CE, Werner B, Hacker NF, Warton K. The untapped potential of ascites in ovarian cancer research and treatment. Br J Cancer. 2020;123:9-16
38. Izar B, Tirosh I, Stover EH, Wakiro I, Cuoco MS, Alter I. et al. A single-cell landscape of high-grade serous ovarian cancer. Nat Med. 2020;26:1271-9
39. Slattery K, Yao CH, Mylod E, Scanlan J, Scott B, Crowley JP. et al. Uptake of lipids from ascites drives NK cell metabolic dysfunction in ovarian cancer. Sci Immunol. 2025;10:eadr4795
40. Dogra S, Adhikari L, Benbrook DM, Bohn JA, Burgett A, Chandra V. et al. Harnessing ovarian cancer ascites for translational science: models, biomarkers, and therapeutics. Mol Cancer. 2025;24:257
41. Hrvat A, Schmidt M, Wagner B, Zwanziger D, Kimmig R, Volbracht L. et al. Electrolyte imbalance causes suppression of NK and T cell effector function in malignant ascites. J Exp Clin Cancer Res. 2023;42:235
42. Shin SH, Lee YE, Yoon HN, Yuk CM, An JY, Seo M. et al. An innovative strategy harnessing self-activating CAR-NK cells to mitigate TGF-beta1-driven immune suppression. Biomaterials. 2025;314:122888
43. Kment J, Newsted D, Young S, Vermeulen MC, Laight BJ, Greer PA. et al. Blockade of TGF-β and PD-L1 by bintrafusp alfa promotes survival in preclinical ovarian cancer models by promoting T effector and NK cell responses. Br J Cancer. 2024;130:2003-15
44. Maas RJA, Hoogstad-van Evert JS, Hagemans IM, Brummelman J, van Ens D, de Jonge P. et al. Increased peritoneal TGF-β1 is associated with ascites-induced NK-cell dysfunction and reduced survival in high-grade epithelial ovarian cancer. Front Immunol. 2024;15:1448041
45. Rea A, Santana-Hernandez S, Villanueva J, Sanvicente-Garcia M, Cabo M, Suarez-Olmos J. et al. Enhancing human NK cell antitumor function by knocking out SMAD4 to counteract TGF-β and activin A suppression. Nat Immunol. 2025;26:582-94
46. Bonavita E, Bromley CP, Jonsson G, Pelly VS, Sahoo S, Walwyn-Brown K. et al. Antagonistic inflammatory phenotypes dictate tumor fate and response to immune checkpoint blockade. Immunity. 2020;53:1215-29 e8
47. Joshi S, Sharabi A. Targeting myeloid-derived suppressor cells to enhance natural killer cell-based immunotherapy. Pharmacol Ther. 2022;235:108114
48. Zhou Y, Cheng L, Liu L, Li X. NK cells are never alone: crosstalk and communication in tumour microenvironments. Mol Cancer. 2023;22:34
49. Wang Y, Geller AE, Yan J. Unexpected Breg-NK crosstalk in STING agonist therapy. Cell Mol Immunol. 2022;19:1330-2
50. Ben-Shmuel A, Gruper Y, Halperin C, Levi-Galibov O, Rosenberg-Fogler H, Barki D. et al. Cancer-associated fibroblasts serve as decoys to suppress NK cell znticancer cytotoxicity in breast cancer. Cancer Discov. 2025;15:1247-69
51. Wang K, Ni B, Xie Y, Li Z, Yuan L, Meng C. et al. Nociceptor neurons promote PDAC progression and cancer pain by interaction with cancer-associated fibroblasts and suppression of natural killer cells. Cell Res. 2025;35:362-80
52. Amit M, Eichwald T, Roger A, Anderson J, Chang A, Vermeer PD. et al. Neuro-immune cross-talk in cancer. Nat Rev Cancer. 2025;25:573-89
53. Wang ZH, Peng WB, Zhang P, Yang XP, Zhou Q. Lactate in the tumour microenvironment: from immune modulation to therapy. EBioMedicine. 2021;73:103627
54. Jin J, Yan P, Wang D, Bai L, Liang H, Zhu X. et al. Targeting lactylation reinforces NK cell cytotoxicity within the tumor microenvironment. Nat Immunol. 2025;26:1099-112
55. Guo X, Tan S, Wang T, Sun R, Li S, Tian P. et al. NAD⁺ salvage governs mitochondrial metabolism, invigorating natural killer cell antitumor immunity. Hepatology. 2023;78:468-85
56. Fang X, Guo L, Xing Z, Shi L, Liang H, Li A. et al. IDO1 can impair NK cell function against non-small cell lung cancer by downregulation of NKG2D ligand via ADAM10. Pharmacol Res. 2022;177:106132
57. Cui JX, Xu XH, He T, Liu JJ, Xie TY, Tian W. et al. L-kynurenine induces NK cell loss in gastric cancer microenvironment via promoting ferroptosis. J Exp Clin Cancer Res. 2023;42:52
58. Grobben Y, de Man J, van Doornmalen AM, Muller M, Willemsen-Seegers N, Vu-Pham D. et al. Targeting Indoleamine 2,3-Dioxygenase in Cancer Models Using the Novel Small Molecule Inhibitor NTRC 3883-0. Front Immunol. 2020;11:609490
59. Sandoval TA, Salvagno C, Chae CS, Awasthi D, Giovanelli P, Marin Falco M. et al. Iron chelation therapy elicits innate immune control of metastatic ovarian cancer. Cancer Discov. 2024;14:1901-21
60. Shi Y, Chen Y, Wang Y, Mo D, Ai H, Zhang J. et al. Therapeutic effect of small extracellular vesicles from cytokine-induced memory-like natural killer cells on solid tumors. J Nanobiotechnology. 2024;22:447
61. Uppendahl LD, Felices M, Bendzick L, Ryan C, Kodal B, Hinderlie P. et al. Cytokine-induced memory-like natural killer cells have enhanced function, proliferation, and in vivo expansion against ovarian cancer cells. Gynecol Oncol. 2019;153:149-57
62. Foltz JA, Tran J, Wong P, Fan C, Schmidt E, Fisk B. et al. Cytokines drive the formation of memory-like NK cell subsets via epigenetic rewiring and transcriptional regulation. Sci Immunol. 2024;9:eadk4893
63. Porter RL, Tayob N, Tarannum M, Birch G, Nguyen M, Welch C. et al. A phase 1b study of intraperitoneal cytokine-induced memory-like (CIML) natural killer (NK) cell therapy in platinum resistant ovarian cancer. J Immunother Cancer. 2025;13:547
64. Berrien-Elliott MM, Becker-Hapak M, Cashen AF, Jacobs M, Wong P, Foster M. et al. Systemic IL-15 promotes allogeneic cell rejection in patients treated with natural killer cell adoptive therapy. Blood. 2022;139:1177-83
65. Van der Meer JMR, Maas RJA, Guldevall K, Klarenaar K, de Jonge P, Evert JSH. et al. IL-15 superagonist N-803 improves IFNγ production and killing of leukemia and ovarian cancer cells by CD34(+) progenitor-derived NK cells. Cancer Immunol Immunother. 2021;70:1305-21
66. Felices M, Wesley E, Bendzick LE, Kodal B, Hopps R, Grzywacz B. et al. Reverse translation identifies the synergistic role of immune checkpoint blockade and IL15 to enhance immunotherapy of ovarian cancer. Cancer Immunol Res. 2023;11:674-86
67. Vaishampayan UN, Muzaffar J, Winer I, Rosen SD, Hoimes CJ, Chauhan A. et al. Nemvaleukin alfa, a modified interleukin-2 cytokine, as monotherapy and with pembrolizumab in patients with advanced solid tumors (ARTISTRY-1). J Immunother Cancer. 2024;12:e010143
68. Geller M, Cooley S, Wallet M, Valamehr B, Teoh D, DeFor T. et al. APOLLO: A phase I study of adaptive memory natural killer (NK) cells in recurrent ovarian cancer. J Clin Oncol. 2020;38:6044
69. Cooley S, Geller M, Cichocki F, Curtsinger J, McKenna DH, Storgard C. et al. In vivo persistence and function of adaptive NK cell infusions (FATE-NK100) from CMV seropositive haploidentical related donors. Biol Blood Marrow Transplant. 2019;25:S338
70. Bordoloi D, Kulkarni AJ, Adeniji OS, Pampena MB, Bhojnagarwala PS, Zhao S. et al. Siglec-7 glyco-immune binding mAbs or NK cell engager biologics induce potent antitumor immunity against ovarian cancers. Sci Adv. 2023;9:eadh4379
71. Gauthier L, Virone-Oddos A, Beninga J, Rossi B, Nicolazzi C, Amara C. et al. Control of acute myeloid leukemia by a trifunctional NKp46-CD16a-NK cell engager targeting CD123. Nat Biotechnol. 2023;41:1296-306
72. Boje AS, Pekar L, Koep K, Lipinski B, Rabinovich B, Evers A. et al. Impact of antibody architecture and paratope valency on effector functions of bispecific NKp30 x EGFR natural killer cell engagers. MAbs. 2024;16:2315640
73. Liao B, Tumanut C, Li L, Corper A, Challa D, Chang A. et al. Identification of novel anti-CD16a antibody clones for the development of effective natural killer cell engagers. MAbs. 2024;16:2381261
74. Hong SD, Katuwal NB, Kang MS, Ghosh M, Park SM, Kim TH. et al. Trastuzumab-mediated antibody-dependent cell-mediated cytotoxicity (ADCC) enhances natural killer cell cytotoxicity in HER2-overexpressing ovarian cancer. Int J Mol Sci. 2024 25
75. Nikkhoi SK, Li G, Eleya S, Yang G, Vandavasi VG, Hatefi A. Bispecific killer cell engager with high affinity and specificity toward CD16a on NK cells for cancer immunotherapy. Front Immunol. 2022;13:1039969
76. Khoshtinat Nikkhoi S, Yang G, Owji H, Grizotte-Lake M, Cohen RI, Gil Gonzalez L. et al. Bispecific immune cell engager enhances the anticancer activity of CD16+ NK cells and macrophages in vitro, and eliminates cancer metastasis in NK humanized NOG mice. J Immunother Cancer. 2024;12:e008295
77. Oberg HH, Kellner C, Gonnermann D, Sebens S, Bauerschlag D, Gramatzki M. et al. Tribody [(HER2)₂xCD16] is more effective than trastuzumab in enhancing γδ T cell and natural killer cell cytotoxicity against HER2-expressing cancer cells. Front Immunol. 2018;9:814
78. Gauthier L, Morel A, Anceriz N, Rossi B, Blanchard-Alvarez A, Grondin G. et al. Multifunctional natural killer cell engagers targeting NKp46 trigger protective tumor immunity. Cell. 2019;177:1701-13 e16
79. Lin H, Case R, Wei KY, Eres I, Alba BM, Kaner J. et al. Characterization and comparative analysis of multifunctional natural killer cell engagers during antitumor responses. Cell Rep Med. 2025;6:102117
80. El-Khoueiry A, Saavedra O, Thomas J, Livings C, Garralda E, Hintzen G. et al. First-in-human phase I study of a CD16A bispecific innate cell engager, AFM24, targeting EGFR-expressing solid tumors. Clin Cancer Res. 2025;31:1257-67
81. Biederstadt A, Rezvani K. Engineered natural killer cells for cancer therapy. Cancer Cell. 2025;43:1987-2013
82. Marin D, Li Y, Basar R, Rafei H, Daher M, Dou J. et al. Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19(+) B cell tumors: a phase 1/2 trial. Nat Med. 2024;30:772-84
83. Ghobadi A, Bachanova V, Patel K, Park JH, Flinn I, Riedell PA. et al. Induced pluripotent stem-cell-derived CD19-directed chimeric antigen receptor natural killer cells in B-cell lymphoma: a phase 1, first-in-human trial. Lancet. 2025;405:127-36
84. Wang X, Zhang Y, Jin Y, Dai L, Yue Y, Hu J. et al. An iPSC-derived CD19/BCMA CAR-NK therapy in a patient with systemic sclerosis. Cell. 2025;188:4225-38 e12
85. Thangaraj JL, Coffey M, Lopez E, Kaufman DS. Disruption of TGF-β signaling pathway is required to mediate effective killing of hepatocellular carcinoma by human iPSC-derived NK cells. Cell Stem Cell. 2024;31:1327-43 e5
86. Woan KV, Kim H, Bjordahl R, Davis ZB, Gaidarova S, Goulding J. et al. Harnessing features of adaptive NK cells to generate iPSC-derived NK cells for enhanced immunotherapy. Cell Stem Cell. 2021;28:2062-75.e5
87. Harano K, Kaneko S, Nakatsura T, Yuda J, Fuse N, Sato A. et al. First in human trial of off-the shelf iPS derived anti-GPC3 NK cells for recurrent ovarian clear cell carcinoma with peritoneal dissemination. Cancer Res. 2022;82:5185
88. Yi E, Lee E, Park HJ, Lee HH, Yun SH, Kim HS. A chimeric antigen receptor tailored to integrate complementary activation signals potentiates the antitumor activity of NK cells. J Exp Clin Cancer Res. 2025;44:86
89. Pan C, Zhai Y, Cui Z, Yin Y, Xu M, Wang D. et al. Natural killer cell-specific chimeric antigen receptor enhances CAR NK cell functions and anti-tumor activity. Theranostics. 2025;15:9344-58
90. Acharya S, Basar R, Daher M, Rafei H, Li P, Uprety N. et al. CD28 costimulation augments CAR signaling in NK cells via the LCK/CD3ζ/ZAP70 signaling axis. Cancer Discov. 2024;14:1879-900
91. Li Y, Hermanson DL, Moriarity BS, Kaufman DS. Human iPSC-derived natural killer cells engineered with chimeric antigen receptors enhance anti-tumor activity. Cell Stem Cell. 2018;23:181-92 e5
92. Klapdor R, Wang S, Morgan M, Dork T, Hacker U, Hillemanns P. et al. Characterization of a novel third-generation anti-CD24-CAR against ovarian cancer. Int J Mol Sci. 2019;20:660
93. Tarannum M, Dinh K, Vergara J, Birch G, Abdulhamid YZ, Kaplan IE. et al. CAR memory-like NK cells targeting the membrane proximal domain of mesothelin demonstrate promising activity in ovarian cancer. Sci Adv. 2024;10:eadn0881
94. Hung CF, Xu X, Li L, Ma Y, Jin Q, Viley A. et al. Development of anti-human mesothelin-targeted chimeric antigen receptor messenger RNA-transfected peripheral blood lymphocytes for ovarian cancer therapy. Hum Gene Ther. 2018;29:614-25
95. Shalaby N, Xia Y, Kelly JJ, Sanchez-Pupo R, Martinez F, Fox MS. et al. Imaging CAR-NK cells targeted to HER2 ovarian cancer with human sodium-iodide symporter-based positron emission tomography. Eur J Nucl Med Mol Imaging. 2024;51:3176-90
96. He M, Ao X, Yang Y, Xu Y, Liu T, Ao L. et al. Construction of self-driving anti-αFR CAR-engineered NK cells based on IFN-γ and TNF-α synergistically induced high expression of CXCL10. Neoplasia. 2024;58:101065
97. Li J, Hu H, Lian H, Yang S, Liu M, He J. et al. NK-92MI cells engineered with anti-claudin-6 chimeric antigen receptors in immunotherapy for ovarian cancer. Int J Biol Sci. 2024;20:1578-601
98. Klapdor R, Wang S, Morgan MA, Zimmermann K, Hachenberg J, Büning H. et al. NK cell-mediated eradication of ovarian cancer cells with a novel chimeric antigen receptor directed against CD44. Biomedicines. 2021;9:1339
99. Ueda T, Kumagai A, Iriguchi S, Yasui Y, Miyasaka T, Nakagoshi K. et al. Non-clinical efficacy, safety and stable clinical cell processing of induced pluripotent stem cell-derived anti-glypican-3 chimeric antigen receptor-expressing natural killer/innate lymphoid cells. Cancer Sci. 2020;111:1478-90
100. Luo J, Guo M, Huang M, Liu Y, Qian Y, Liu Q. et al. Neoleukin-2/15-armored CAR-NK cells sustain superior therapeutic efficacy in solid tumors via c-Myc/NRF1 activation. Signal Transduct Target Ther. 2025;10:78
101. Li F, Sheng Y, Hou W, Sampath P, Byrd D, Thorne S. et al. CCL5-armed oncolytic virus augments CCR5-engineered NK cell infiltration and antitumor efficiency. J Immunother Cancer. 2020;8:e000131
102. Park AK, Fong Y, Kim SI, Yang J, Murad JP, Lu J. et al. Effective combination immunotherapy using oncolytic viruses to deliver CAR targets to solid tumors. Sci Transl Med. 2020;12:eaaz1863
103. Jo DH, Kaczmarek S, Khan AUH, Pervin J, Clark DM, Gadde S. et al. Entinostat, a histone deacetylase inhibitor, enhances CAR-NK cell anti-tumor activity by sustaining CAR expression. Front Immunol. 2025;16:1533044
104. Quixabeira DCA, Pakola S, Jirovec E, Havunen R, Basnet S, Santos JM. et al. Boosting cytotoxicity of adoptive allogeneic NK cell therapy with an oncolytic adenovirus encoding a human vIL-2 cytokine for the treatment of human ovarian cancer. Cancer Gene Ther. 2023;30:1679-90
105. Lyu L, Feng Y, Huang B, Xu RH, Hu Y. Mapping the global landscape for induced pluripotent stem cells from patents and clinical trials. Nat Biotechnol. 2024;42:563-9
106. Zhu H, Blum RH, Bernareggi D, Ask EH, Wu Z, Hoel HJ. et al. Metabolic reprograming via deletion of CISH in human iPSC-derived NK cells promotes in vivo persistence and enhances anti-tumor activity. Cell Stem Cell. 2020;27:224-37.e6
Author contact
Corresponding authors: Hai-ou Liu, PhD, Shanghai Key Laboratory of Female Reproductive Endocrine Related Diseases, Obstetrics and Gynecology Hospital, Fudan University, No. 128, Shen-yang Road, Yangpu District, Shanghai 200011, China; Tel: +86-021-33189900; E-mail: liuhaiouedu.cn. Jia-qi Lu, MD, PhD, Department of Gynecology, Obstetrics and Gynecology Hospital, Fudan University, No. 128, Shen-yang Road, Yangpu District, Shanghai 200011, China; Tel: +86-021-33189900 ext. 2020; Email: lujiaqidoccom.

 Global reach, higher impact
Global reach, higher impact