Impact Factor ISSN: 1449-2288
Int J Biol Sci 2010; 6(2):116-128. doi:10.7150/ijbs.6.116 This issue Cite
Research Paper
E2F-1 has dual roles depending on the cell cycle
1. Department of Microbiology and Immunology, Finch University of Health Sciences/Chicago Medical School (now Rosalind Franklin University), 3333 Green Bay Road, North Chicago, Illinois 60064-3095, USA
2. Current address: Microbiology Department, School of Medicine, Ankara University, Sihhiye-Ankara, Turkey
3. Current address: 3802 Sagamore Drive, Greensboro, NC 27410, USA
Received 2009-12-24; Accepted 2010-2-28; Published 2010-3-3
Abstract
The E2F family of transcription factors play a critical role in the control of cell proliferation. E2F-1 is the major cellular target of pRB and is regulated by pRB during cell proliferation. E2F-1-mediated activation and repression of target genes occurs in different settings. The role of E2F-1 and E2F-1/pRB complexes in regulation of different target genes, and in cycling versus quiescent cells, is unclear. In this study, effects of free E2F-1 (doesn't complex with pRb) and E2F-1/pRb complex, on E2F-1 target gene expression were compared in different cell growth conditions. Findings suggest that E2F-1 acts in different ways, not only depending on the target gene but also depending on different stages of the cell cycle. For example, E2F-1 acts as part of the repression complex with pRB in the expression of DHFR, b-myb, TK and cdc2 in asynchronously growing cells; on the other hand, E2F-1 acts as an activator in the expression of the same genes in cells that are re-entering the cycle.
Keywords: E2F-1, Rb, Cell cycle, cmyc, dhfr, TK, cdc2
1. Introduction
The loss of pRB function either by mutation or by association with viral oncoproteins is linked to uncontrolled cell proliferation and is accompanied by the release of transcription factors such as E2F-1 [1-5]. Over the past decade, the E2F family of transcription factors has emerged as central components of the regulatory machinery of cell growth and proliferation [6]. E2F DNA binding sites in the adenovirus E2 promoter were the first E2F binding sites to be identified [7].
Thereafter, E2F DNA binding sites have been identified in promoters of many genes intimately involved in the regulation of cell cycle progression. It was shown that around 1,000 genes are regulated by E2F-1 [8-10]. E2F proteins are encoded by at least eight genes, E2F-1 through E2F-8. While specific roles for individual E2Fs in mediating the effects of RB loss are emerging, it is also becoming clear that there are no simple divisions of labor among the E2F family. Instead, an individual E2F can function to activate or repress transcription, promote or impede cell cycle progression and enhance or inhibit cell death, dependent on the cellular context [6].
In the initial model of E2F-1 action, activation of E2F-1 dependent transcription was proposed to be caused by the unmasking of an activation domain after release of pRB in the E2F complex [2, 3]. Regarding the mechanism of E2F-mediated transactivation, it is believed that “free” E2F binds to its binding sites in the promoters of the target gene. So-called “free” E2F is the heterodimer formed between E2F family members and DP family members without making a complex with pRB or related proteins [11]. This view was supplemented by the discovery that E2F-1/pRB complexes can actively repress transcription [12] [13]. Therefore, there appears to be three generic types of E2F complexes: activator E2F complexes (“free” E2F), in which the E2F activation domain promotes transcription, inhibited E2F complexes, in which the activation domain is masked by pRB-family proteins to give a complex that is essentially inert, and repressor E2F complexes, in which RB family proteins that are recruited to the DNA by E2F assemble a repressor activity [5].
The discovery of the E2F-1/pRB repressor has raised a debate that is still not completely settled. Should E2F binding sites be thought of as activators of gene expression in S phase or as elements that confer cell-cycle regulated repression in G0/G1? Mutational analysis of putative E2F-binding sites in relatively small fragments of genomic DNA suggests that E2F-binding sites regulate gene expression in different ways in different promoter contexts [11] [14, 15]. For example, promoter mutation experiments have shown that alteration in the E2F-1 binding sites of the c-myc, cdc2 and human DHFR promoters resulted in 50-90% loss of transcription activity [16] [17, 18]. Here the E2F-1-binding sites appear to act as activator elements during the S phase of the cell cycle. Release of E2F-1 from pRB results in activation of gene expression during the S phase of the cell cycle. In contrast to the experiments described above, similar mutations of these sites in the promoters of other genes, such as b-myb [19] and mouse DHFR [20] caused increased transcriptional activity. Here the E2F-1-binding sites appear to act as repressor elements during G0/G1 and gene expression results from the release of repression. It is still unclear how active repression by the E2F-1/pRB complex contributes to control of transcription, compared to pRB-mediated inhibition of transcriptional activation by free E2F-1 on individual target gene expression.
In this study, cell lines overexpressing E2F-1/wt and E2F-1/411, which doesn't complex with pRB [21, 22], were used to better understand the role of E2F-1/pRB interaction on cell phenotype and gene expression. This paper presents a novel finding that the E2F-1/pRB complex acts in different ways not only dependent on the individual target genes, but also on the same target genes in cycling cells as compared to cells re-entering the cell cycle. The findings show that E2F-1 acts as a part of the repression complex with pRB on the expression of DHFR, b-myb, TK and cdc2 in cycling cells; on the other hand, E2F-1 acts as an activator on the expression of the same genes of cells that are re-entering the cycle. DHFR, b-myb, TK and cdc2 are also actively growth regulated by E2F-1, and free E2F increased the expression of these target genes in all stages of the cells re-entering the cycle. However, c-myc and cyclin D1 are not significantly growth regulated by E2F-1 in any stage of the cell cycle. Rb is also growth regulated by E2F-1. Interestingly, free E2F-1 acts as a repressor of Rb expression at G1 and as an activator at S phase of the cell cycle.
2. MATERIALS AND METHODS
2.1. Cells and viruses
ψ-CRE, a murine fibroblast cell line was used in these experiments. The cells were grown in DMEM supplemented with 5% (vol/vol) fetal bovine serum and 5% calf serum in a 5% CO2 atmosphere at 370C [23]. Retroviral vector, Linker Neo CMV E2F-1 was used to express E2F-1/wt and E2F-1/mutant genes as described below. Linker Neo CMV E2F-1 is identical to Linker CMV T [24]. except that the large T antigen gene from simian virus 40 was replaced by a cDNA, approximately 1326 bps long, encoding E2F-1/wt [25] or E2F-1/Y411A [2] (obtained from Dr. K. Helin). pX17 is a retroviral vector [24, 26] that encodes the neomycin resistance gene but does not encode E2F-1 gene. pX17 was used as negative control in the experiments. To produce infectious virus, the recombinant plasmids encoding Linker Neo CMV E2F-1s were transfected together with helper virus containing vector (SV-A-MLV-env) which encoded genes needed to complement the linker virus, but did not encode whole virus, [27] into Cos-7 packaging cells using the calcium-phosphate transfection method [24]. Two to three days later, virus containing medium was collected from these cells and used to infect established ψ-CRE murine cell lines [23].
2.2. Antibodies
The antibodies used in immunoblotting for detection of E2F-1, were E2F-1 (C-20), cdcp34 (cdc2; B-6), cyclin D1 (HD 11), c-myc (9E10) Santa Cruz Biotechnology, respectively. The antibodies used for immunoprecipitation of E2F-1 and pRb were KH95 (Santa Cruz Biotechnology) and G3-245 (Pharmingen; San Diego, CA) monoclonal antibodies, respectively
2.3. Immunoblotting
For Western blotting, cells were lysed by addition of 1 ml of lysis buffer (50 mM Tris [pH 8.0], 5 mM EDTA, 150 mM NaCl, 0.5% NP-40, 1 mM PMSF and 0.01 mg of aprotinin per ml to the cell monolayer. Cell free extracts prepared from 4x104 cells were electrophoresed on sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels and transferred to Hybond ECL membranes (Amersham; Arlington Heights, IL). Antibodies described above were used for immunoblotting.
2.4. Immunoprecipitation
Immunoprecipitations were performed using cell-free extract prepared as described in the immunoblotting procedure. The extracts were precleared by incubation with Protein G Plus-/Protein A-Agarose beads (Oncogene Science; Uniondale, NY) that had been treated with BSA. E2F-1 or pRB was immunoprecipitated from precleared extracts by adding 2 µg of specific antibodies and 20 µl of BSA treated Protein G Plus-/Protein A-Agarose beads. This mixture was incubated for 15 hours and centrifuged as described above. The immune complexes were washed 4 times with lysis buffer and then analyzed by SDS-PAGE and immunoblotted as described above.
2.5. Soft agar assays
Five or ten thousand cells were plated in 1 ml of top agar composed of DMEM supplemented with 5% fetal bovine serum and 5% calf serum and containing 0.38% Noble agar. Dishes were maintained at 37°C in a humidified 5% CO2 atmosphere. After 3 weeks, colonies were either photographed or counted. For photomicrographs, dishes were stained with p-iodonitrotetrazolium violet and photographed. For counting, colonies were visualized with a microscope using low power magnification and those greater than 50 µm in diameter were counted. Six microscope fields per dish were counted and used to calculate the total number of colonies on the entire dish (based on dish surface area). Colony counts were obtained from four dishes each cell line for per experiment. The data are expressed as the means of counts from five independent experiments.
2.6. Cell cycle synchronization
To synchronize cells by serum starvation, 5 x 105 cells were seeded into 10-cm dishes. Forty-eight hours later, cells were washed with PBS and DMEM supplemented with 0.5% fetal bovine serum was then added. After 60 h, cells were stimulated into the cell cycle by addition of complete medium (DMEM supplemented with 5% (vol/vol) fetal bovine serum and 5% calf serum). Samples for total RNA preparation and flow cytometry were collected at time 0 and appropriate intervals thereafter as indicated.
2.7. Total RNA isolation
RNA STAT-60 Total RNA (TEL-TEST, INC.; Friendswood, TX) isolation reagent kit was used and its protocol followed for total RNA isolation. Cells in culture dishes were homogenized with RNA STAT-60, followed by RNA extraction using chloroform. RNA was precipitated with 0.5 vol. of isopropanol followed by washing with 75% ethanol. The quality of the samples was checked electrophoretically, and quantification was done spectrophotometrically. Two independent RNA preparations were used in the experiment.
2.8. Primers used in the MRT-PCR
All primers for E2F-1 target genes were designed with the Primer3: WWW primer tool program (http://biotools.umassmed.edu/bioapps/primer3_www.cgi) and are shown in the table 1. Primers were made by IDT, Inc. (Coralville, IA).
Oligonucleotides used in this study
| Primer | n-mer | sequence | PCR fragment size (bp) |
|---|---|---|---|
| FBmyb (forward) | 21 | 5'-GCC AGT CAC CCT TTT AAC CGC-3' | 713 |
| RBmyb (reverse) | 23 | 5'-CAA ATT TAC TCA GGT CGC ACC AA-3' | |
| FDHFR (forward) | 21 | 5'-GAC CAT TGA ACT GCA TCG TCG-3' | 452 |
| RDHFR (reverse) | 25 | 5'-AAT CAA TTT CTG GGA AAA ACG TGT C-3' | |
| FcycD1 (forward) | 23 | 5'-CTG ACA CCA ATC TCC TCA ACG AC-3' | 647 |
| RcycD1 (reverse) | 22 | 5'-AAA GAA AGT GCG TTG TGC GGT A-3' | |
| Fcdc2 (forward) | 23 | 5'-AGA GCG TTT GGA ATA CCG ATA CG-3' | 423 |
| Rcdc2 (reverse) | 24 | 5'-TGT CCA AGT CAT CAA AGT ACG GGT-3' | |
| Fc-myc (forward) | 23 | 5'-ATA AAA GAA GCT TTT CGG GCG TT-3' | 508 |
| Rc-myc (reverse) | 22 | 5'-GGC TGT ACG GAG TCG TAG TCG A-3' | |
| FTK (forward) | 21 | 5'-GTA TGC CAA AGA CAC GCG CTA-3' | 395 |
| RTK (reverse) | 22 | 5'-ACA CGG AGT GAT ACT TGT CGG C-3' | |
| FRb (forward) | 25 | 5'-GCG ATA TAA ACT TGG AGT CCG ATT G-3' | 569 |
| RRb (reverse) | 24 | 5'-GCG TGC ACA GAG TGT ATT TAG TCG-3' |
2.9. Semiquantitative Multiplex RT-PCR
Multiplex RT-PCR was performed using Promega's Access RT-PCR system. RT-PCR was used first to determine the linear range of target genes. Second, the linear range of the internal standard was determined using β-actin and β-actin competimer (QuantumRNA β-actin internal standards, Ambion). Conditions for MRT-PCR were optimized after determining the β-actin and β-actin competimer ratio. PCR cycles were optimized so that the target gene would be in the linear range. Reactions contained AMV-RT and Tfl DNA polymerase, AMV/Tfl Reaction Buffer, dNTP mix, specific upstream and downstream primers, β-actin and β-actin competimer primers, MgSO4, and RNA template. The PCR machine was programmed as follows: 480C for 45 minutes to synthesize first strand cDNA; 940C for 2 minutes to denature template; 940C 45 seconds for denaturation; at 600C for 45 seconds for annealing; and 680C for 1 minute for extension. The PCR products were run in a 1.5% agarose gel and the gel was stained with EtBr. EtBr signals were analyzed by computer assisted densitometry.
2.10. Fluorescence-activated cell sorting (FACS) analysis
To analyze cellular DNA content, cells were fixed and stained [28]. Briefly, 106 trypsinized cells were washed three times with PBS and then fixed by incubation with 50 μl of 0.125% paraformaldehyde at 370C for 5 minutes. Then 450 μl of ice-cold methanol was added to the sample. The fixed cells were washed three times with 0.1% Triton X-100 in PBS and treated with RNase A (0.04 Kunitz units) for 30 minutes. The cells were then stained with 50 μg of propidium iodide per ml. Cell analysis was performed using a Coulter EPICS Elite EPS flow cytometer (Coulter Electronics; Miami, FL). The fraction of the total cell population present in each of the G1, S and G2/M cell cycle phases was obtained from DNA histograms by mathematical modeling using MPLUS software (Phoenix Flow Systems; San Diego, CA [28]).
3. Results
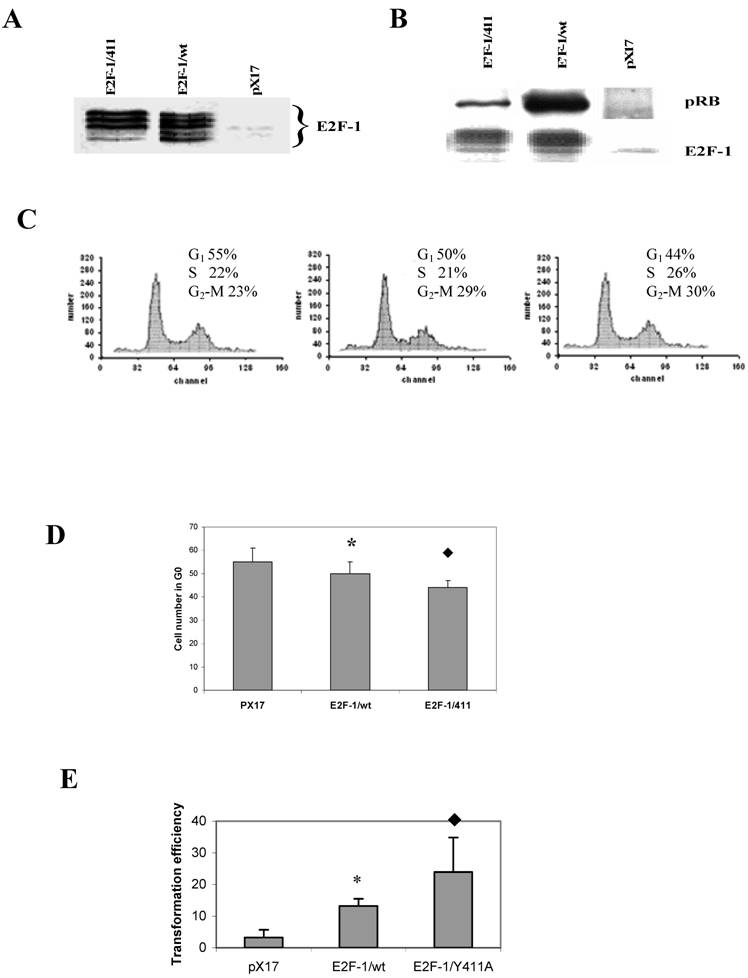
3.1. Overexpression of E2F-1/wt and E2F-1/411 genes in ψ-CRE cell lines changes the cell growth phenotype differentially depending on interaction with pRB
For overexpression of E2Fs, the mouse fibroblast ψ-CRE cell line was used since it had the highest amount of pRB protein among the cell lines C3H10T1/2, BALB/c-3T3 and NIH 3T3 (data not shown) and we wanted to examine interaction between pRB and E2F-1. Using retroviral vectors, three cell lines were established that overexpress E2F-1/wt, E2F-1/411 or the control, pX17 (the retroviral vector with no insert, negative control). Cell clones were pooled from cells growing in the presence of G418. Figure 1A shows immunoblots of the cells expressing E2F-1. Multiple closely-migrating protein bands were seen. These are differentially-phosphorylated forms of E2F-1 protein [29]. In addition, cells infected with pX17 were used as negative controls. These cell lines were used for the remaining experiments of this study. The E2F-1/411 pRB interactions were described previously in vitro and in the yeast two-hybrid system and a single mutation of E2F-1 411 site decreased binding of E2F-1 to pRb without affecting E2F-1 transcription activity [21]. Therefore it would be important to show E2F-1/411 pRB binding acts the same way in the cell. To test the in vivo affinity of the E2F-1/411 for pRB, E2F-1 was immunoprecipitated from extracts made from asynchronously growing E2F-1/wt or E2F-1/411 cell lines using an anti-E2F-1-specific antibody as explained in detail in Material and Methods. It was found that E2F-1/411 pRB binding is five fold less than E2F-1/wt pRB binding (Fig. 1B).
3.2. Overexpression of E2F-1/wt and E2F-1/411 changes the cell cycle of asynchronously growing cells and increased cell transformation
Overexpression of E2F-1 is sufficient to induce S-phase in most quiescent cells, as reported previously [30, 31]. However, cycling E2F-1 null mutant cells showed no difference in the cell cycle compared to cycling normal cells [32]. Therefore, we first determined the effects of E2F-1/wt and free E2F-1 in cycling cells using flow cytometry. It was found that the overexpression of E2F-1/wt and E2F-1/411 significantly decreased the cell number in G1 phase compared to the control (pX17) cells. Furthermore, E2F-1/Y411A showed fewer cells in G1 as compared to E2F-1/wt-expressing cells (Fig. 1C and D).
As shown in this and some other laboratories previously [33, 34], overexpression of E2F-1 increases the transformation of cells. Therefore we aimed to assess how free E2F-1 (E2F-1/411) would change the transformation efficiency compared to E2F-1/wt. Anchorage-independent growth of E2F-1-overexpressing cells was tested by plating the cells in soft agar medium and assaying the ability of the cells to form colonies. E2F-1/wt- and E2F-1/411- overexpressing cell lines showed significant increases in transformation, compared to the control (pX17) cell line. The mutant E2F-1/Y411A showed a significant increase compared to E2F-1/wt overexpressing cell line (Fig. 1E). Therefore, free E2F-1, unbound to pRB, decreased the G1 phase and increased the transformation of ψ-CRE cell more than E2F-1 bound to pRB.
A. Overexpression of E2F-1/wt and E2F-1/411. Cell free extracts from an equivalent number of cells (5 X 104) of E2F-1/wt and E2F-1/411 were analyzed by immunoblotting using anti-E2F-1 specific polyclonal antibody after separation on 10% SDS-PAGE. The position of the E2F-1 bands are indicated on the right. B. Co-immunoprecipitation of pRb and E2F-1. Cell free extracts from the ψ-CRE cell lines overexpressing E2F-1/wt, E2F-1/411, or control (pX17) were co-precipitated with a monoclonal antibody specific for E2F-1 and followed by western blotting with E2F-1 polyclonal antibody or pRB polyclonal antibody as described in Materials and Methods. C. Histograms of cell cycle analysis of pX17, E2F-1/wt and E2F-1/411 overexpressing asynchronously growing cells are shown with the mean of cell numbers in G1, S and G2/M phases. D. Number of pX17, E2F-1/wt and E2F-1/411 overexpressing asynchronously growing cells in G1. G1 values are mean ± S.E.M., n=4, * P< 0.05 compared to control (pX17) ψ-CRE cell line, u P< 0.05 compared to ψ-CRE E2F-1/wt cell lines. E. Cells were plated in soft agar medium and stained as described in Materials and Methods. Colonies greater than 50 μm in diameter were scored after three weeks of growth. Transformation frequency was expressed as the percentage of total plated cells that formed colonies. Values are mean ± S.E.M., n=5, * P< 0.05 compared to control (pX17) ψ-CRE cell line, u P< 0.05 compared to ψ-CRE E2F-1/wt cell lines.
3.3. Overexpression of E2F-1/wt and E2F-1/411 changed the target gene expression in asynchronously growing cells
It is likely that the different phenotypic changes that occurred in the E2F-1/wt and E2F-1/411-overexpressing cells are a result of changes produced by alterations in the expression of E2F-1 target genes. E2F-1 target genes, such as RB, c-myc, b-myb, DHFR, TK, cdc2, cyclin D and others, are important cell cycle regulators. Therefore, the effects of E2F-1/wt and E2F-1/411 on target gene expression using semiquantitative multiplex RT-PCR (MRT-PCR) were compared.
Results obtained from asynchronized proliferating cells showed that overexpression of E2F-1/wt repressed the expression of DHFR, b-myb, cyclin D1, cdc2, c-myc and TK (Fig. 2). This finding correlates well with most of the previous studies [35, 36]. In addition, DHFR, b-myb, cyclin D1, cdc2, c-myc and TK gene expression were less repressed in cell lines overexpressing E2F-1/Y411A compared to cell lines overexpressing E2F-1/wt. However, none of these genes in E2F-1/411-overexpressing cells had expression levels exceeding those of the control (pX17). Therefore these data imply that in the asynchronously growing cells for DHFR, b-myb, cdc2, TK and cyclin D1 expression, the functional consequence of the pRB-E2F-1 interaction is not that E2F-1 acts as activator and is repressed by pRB binding. Instead, E2F-1 might have a role as a protein helping the E2F-1/pRB complex possibly bind to the promoter and actively repress transcription.
E2F-1 target gene expression levels in asynchronously growing ψ-CRE cells overexpressing E2F-1/wt, E2F-1/411 and control (pX17). A. (Left Column) MRT-PCR of the total RNA of the ψ-CRE cells overexpressing E2F-1/wt , 411 and control was carried out as described in Materials and Methods and products were analyzed on a 1.5% agarose gel. The gels were stained with EtBr, and EtBr signals were analyzed by computer assisted densitometry. B. (Right Column) Results are presented as fold increases in expression of target gene for each cell line compared to control (pX17), which was given the value of 100. Values are mean ± S.E.M., n>3, * P< 0.05 compared to control (pX17) ψ-CRE cells. In both columns, from top to bottom, the genes analyzed were DHFR, B-myb, Cyclin D1, Cdc2, c-myc, TK and RB.
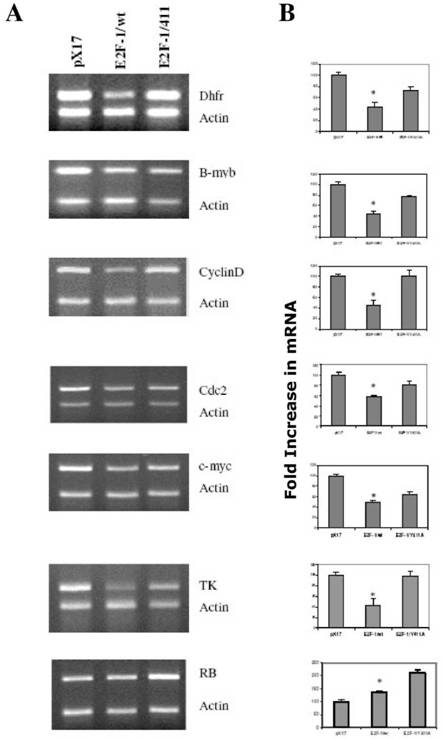
From this follows that, first, if E2F-1 acts as activator, target gene expression would be higher in E2F-1/wt- overexpressing cells compared to that of controls (pX17). Secondly and more importantly, target gene promoters will have only free E2F-1 (unbound to pRB) in E2F-1/411- overexpressing cells. Therefore, if E2F-1 acts as activator, target gene expression levels would be higher than the controls, pX17. However, the data shows that free E2F-1 did not increase target gene expression more than the control levels, and it only decreased the repression of the E2F-1/pRB complex by replacing E2F-1/pRB complex on the promoter. Therefore, E2F-1 does not act as activator, but it helps pRB repress the promoter of target genes including DHFR, b-myb, cyclin D1, cdc2, and TK in the asynchronously growing cells.
Very different results were observed with RB expression (Fig. 2) from those described for the other target genes. The data show that E2F-1 acts as activator for the expression of RB. First, overexpression of E2F-1, even bound to pRB, increased the expression of RB; second, E2F-1/411 (unbound to pRB) increased RB expression more than did E2F-1/wt. These data, therefore, show that E2F-1 has different roles for the expression of different target genes and pRB has a role in controlling the E2F-1 effects on all target genes that were examined in asynchronously growing cells.
E2F-1 target protein expression levels in asynchronously growing ψ-CRE cells overexpressing E2F-1/wt or E2F-1/411. Cell free extracts from an equivalent number of cells (5 X 104) of E2F-1/wt and E2F-1/411 were analyzed by immunoblotting using specific antibody for the indicated individual protein after separation on 10% SDS-PAGE.
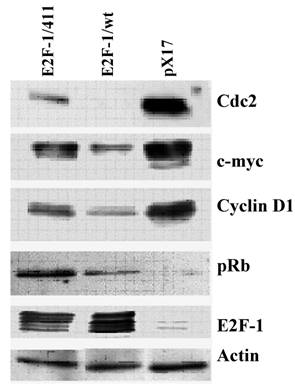
3.4. The E2F-1/wt and E2F-1/411 overexpression changes the protein expression of E2F-1 target genes in asynchronously growing cells
Next, to see the effects of E2F-1/wt and 411 on target genes at the level of protein, the protein levels of some of the E2F target genes used in previous experiments were checked. Western Blot analysis (Fig. 3) showed similar results to RNA levels from RT-PCR. E2F-1/wt decreased the protein levels of cyclin D1, cdc2, c-myc and increased the pRB level. E2F-1/411 overexpression showed less repression compared to E2F-1/wt on the level of cdc2, c-myc and cyclin D1, and increased the pRB level more than E2F-1/wt. Difference in protein expression in different cells is more remarkable compared to the RNA expression analysis. This difference might be related with the sensitivity of the techniques.
3.5. E2F-1/wt and E2F-1/411 change G0/G1 checkpoint controls and S-phase entry
The previous experiment showed that overexpression of E2F-1 in cells decreased the percentage of cells in the G1 phase in cycling cells (Fig. 1). In this experiment the effect of overexpression of E2F-1/wt and free E2F-1 on cells re-entering the cell cycle was examined. E2F-1/wt and 411 showed significantly less accumulation in G0/G1, compared to the control (pX17) cells after serum starvation (Fig. 4A and B). Furthermore, E2F-1/411 showed less accumulation in G0/G1 as compared to E2F-1/wt-expressing cells, similar to the result with cycling cells. Fig. 4C shows the time course of S phase entrance of E2F-1/wt- and mutant E2F-1/411- after resupplying cells with serum. The E2F-1/411 mutant overexpressing cells showed the fastest S-phase entry compared to the other cell lines.
3.6. E2F-1 and pRB have different roles for the growth regulation of the different target genes
To better understand the function of free E2F-1 and the E2F-1/pRB complex on individual target gene expression in different stages of the cell cycle, growth regulation of chosen target genes was investigated in control, E2F-1/wt and E2F-1/411 overexpressing cell lines. Growth regulation of these genes was expressed as the ratio of the expression levels of the target gene in S phase to that in G0/G1 phase [20].
RNA was extracted from cell lines arrested in G0/G1. After serum stimulation, RNA was extracted at various times. Expression of E2F-1 target genes was measured by MRT-PCR. Subsequently, the growth regulation of target genes in E2F-1/wt and E2F-1/411 overexpressing cell lines was compared in Fig. 5.
Cell cycle analysis of pX17, E2F-1/wt and E2F-1/411 overexpressing cells. A. Cells were starved in 0.5% FBS for 60 hours, at which time samples were taken and analyzed by flow cytometry. The histograms from pX17, E2F-1/wt and E2F-1/411 overexpressing cells are shown with the mean of cell numbers in G0/G1, S and G2/M phases. B. Number of pX17, E2F-1/wt or E2F-1/411 overexpressing cells in G0/G1. G0/G1 values are mean ± S.E.M., n=4, * P< 0.05 compared to control (pX17) ψ-CRE cell line, u P< 0.05 compared to ψ-CRE E2F-1/wt cell lines. C. Cells were collected at various times after serum stimulation and the percentage of cells in S phase of the cell cycle was determined. Values are mean ± S.E.M., n=3.
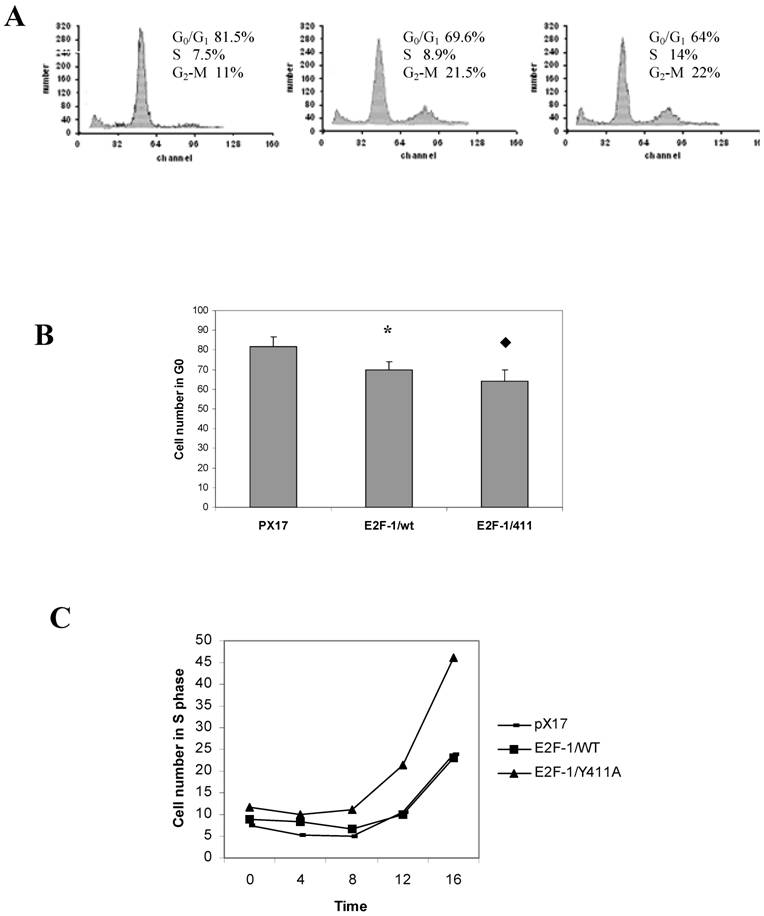
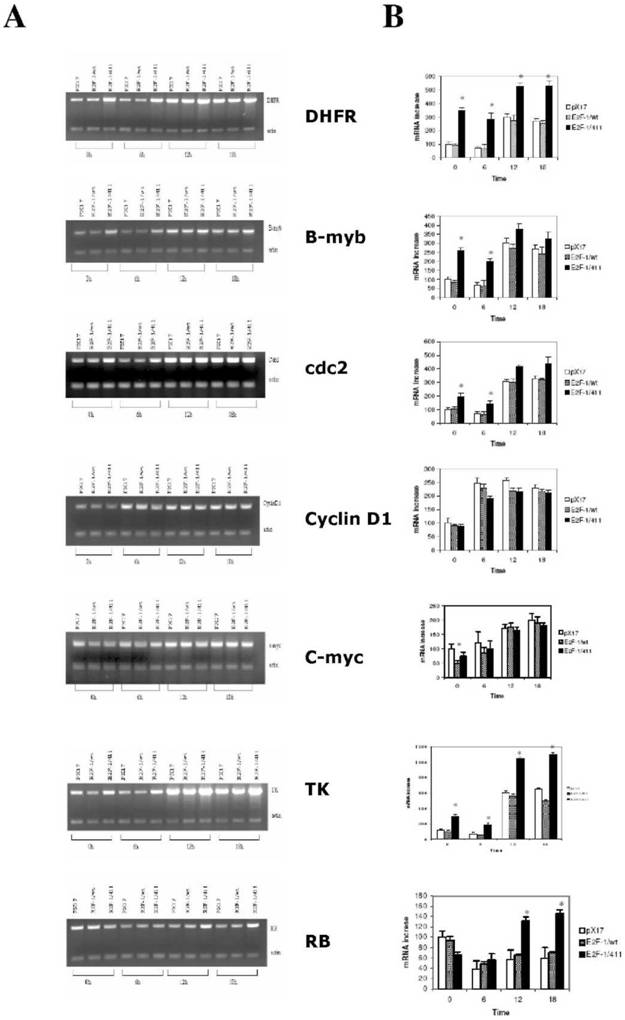
Target genes, including b-myb, DHFR, cdc2, and TK, showed similar results for growth regulation (Fig. 5). For these genes, expression in control (pX17) cells was similar to that in cells expressing E2F-1/wt in both G0/G1 and S phase. In contrast, to that, expression of these genes in the E2F-1/411 overexpressing cells in G0/G1 was generally 3-4-fold higher than that of controls (pX17) and E2F-1/wt cells. In S phase, expression of DHFR and TK was increased as compared to pX17 and E2F-1/wt cells. Expression of c-myc was decreased in cells expressing E2F-1/wt in G0/G1 as compared to cells expressing E2F-1/411. For RB, expression in E2F-1/411 expressing cells was increased in S phase as compared to cells expressing E2F-1/wt (Fig. 5).
Roles of E2F-1 and pRb on target gene expression levels in synchronously growing ψ-CRE cells. A. (Left Column) MRT-PCR of the total RNA of control ψ-CRE cells (pX17) and ψ-CRE cells overexpressing E2F-1/wt and E2F-1/-411. The cells were starved and then stimulated back into the cell cycle at time 0 as described in Materials and Methods. Time points analyzed were time 0, 6 hours, 12 hours and 18 hours. Products were analyzed on 1.5% agarose gels. The gels were stained with EtBr, and EtBr signals were analyzed by computer assisted densitometry. B. (Right Column) Results from A are presented as fold increases in expression (y-axis) of each bar graph of target genes for each cell line compared to control (pX17), which was given the value of 100 at time 0. Values are mean ± S.E.M., n>3, * P< 0.05 compared to control (pX17) ψ-CRE cells at the same time point. In both columns, from top to bottom, the genes analyzed were DHFR, B-myb, Cdc2, Cyclin D1, c-myc, TK and RB.
4. Discussion
It has been known that E2F-1 can be a transactivator and a transrepressor depending on the target genes and cell lines. Here, it is shown that E2F-1 could be both transactivator and transrepressor on the same target gene expression in the same cell depending on the cell cycle. Although cell cycle phenotypic effects of E2F-1 are the same on both cycling cells and cells re-entering the cell cycle (Fig. 1C and 4A and B), the target genes we examined are regulated differently in re-entering cells compared to cycling cells. It was found that on DHFR, b-myb, cdc2 and TK expression, the functional consequence of the pRB-E2F-1 interaction is that E2F acts as activator and is repressed by pRB binding in cells re-entering the cycle. During the re-entry into the cell cycle, free E2F-1 increases the transcription levels of the same genes more than 3-4 fold compared to cells in which the E2F-1/pRB complex was present. However, free E2F-1 (E2F-1/411- overexpressing cells) only de-repressed the expression of DHFR, b-myb, cdc-2, Cyclin D1, C-myc and TK up to the control (pX17) level in asynchronously growing cells. In addition, while there was no significant effect of E2F-1/wt over control (pX17) on the expression of DHFR, B-myb, cdc-2, TK, cyclin D1 and RB in cells re entering the cell cycle, overexpression of E2F-1/wt significantly repressed the expression of the DHFR, B-myb, cdc-2, Cyclin D1, C-myc and TK in cycling cells. Therefore, E2F-1 acts as part of the repression complex for the the expression of the DHFR, B-myb, cdc-2, Cyclin D1, C-myc and TK in cycling cells and as transactivator in cells re-entering the cycle on the expression of DHFR, b-myb, cdc-2, and TK. This study shows that free E2F-1 increased the expression of B-myb, DHFR, cdc2 and TK in cells re-entering the cell cycle and E2F-1 overexpression de-repressed the expression of the same genes in cycling cells.
It may be assumed in this study that E2F-1 overexpression may replace other E2F proteins which have a role for repression of cell cycle at G0/G1. In contrast to the activator E2Fs, E2F4 and E2F5 lack NLS nuclear localization signals (NLS) [37]. So, without the association with pRb2/p130 and pRb/p107, E2F4 and E2F5 can no longer access the cell nucleus [38]. In addition to this mechanism, there is a second mechanism that completely eliminates the possibility of forming pRb2/p130-E2F4/5 complexes. This second mechanism contributed by Skp2, which is the ubiquitin ligase of pRb2/p130 and belongs to the SCF (Skp1, Cullin, F-box protein) family. Skp2 recognizes hyperphosphorylated pRb2/p130, ubiquitin-ligases it and, therefore, causes the quick removal of pRb2/p130 by proteosomes [39, 40]. At this point, the promoter regions become vacant for the activator E2Fs, which, in turn, express the set of gene to initiate the S phase entry [41].
It is obvious that E2Fs and related proteins are not the only factors related with gene expression related with the cell proliferation. Some other factors or changes are different in synchronously growing and asynchronously growing cells that give the differences for the target gene expression in cycling cells versus cells re-entering the cycle. As previous reports indicate, some other elements besides pRB might be involved in regulation of the target genes. For example, in the c-myc promoter, transcription factors Sp1, ATF1, ATF2 and a novel factor distinct from E2F mediates C-MYC promoter activation through its E2F element during exit from quiescence, [42-44] for the b-myb promoter, CHR (cell cycle gene homology regions), which is also found in some other target genes, and CDE (cell cycle-dependent elements) have been shown to be regulatory factors [45] effecting E2F regulation.
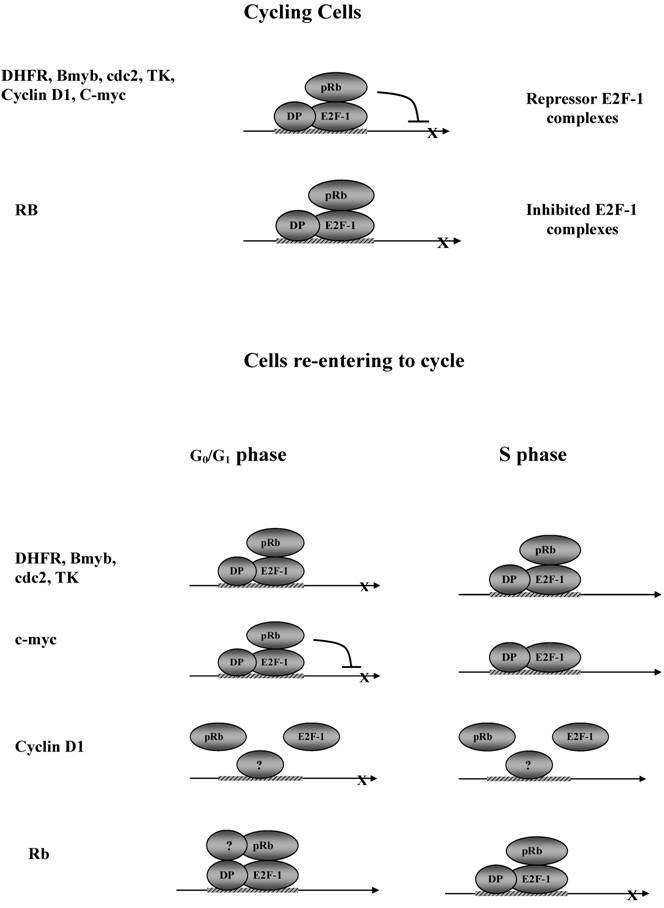
Fig. 6 is proposed to explain the data obtained in these studies. For the target genes DHFR, b-myb, cdc-2, Cyclin D1, C-myc and TK, in cycling cells, the model shows pRB brought to the DNA by E2F-1, and repressing transcription. In this model, E2F-1 without pRB (free E2F) has no effect on expression of these targets. For DHFR, b-myb, cdc-2, and TK, in G0/G1, the functional consequence of the pRB-E2F-1 interaction is that E2F acts as activator and is repressed by pRB binding in cells re-entering the cycle. In here, repression is due to the effect of pRB in repressing the transactivation function of E2F (because transcription is increased in cells expressing E2F-1/411). Therefore, the inhibited E2F complexes model most accurately describes the results from this group, showing that E2F-1 bound to pRB does not stimulate expression of these target genes. But, when pRB cannot bind E2F-1 (in the E2F-1/411 cells) it does stimulate target gene expression.
Interestingly, this study showed that B-myb, DHFR, cdc2 and TK expression was higher in cells overexpressing E2F-1/411 than in cells overexpressing E2F-1/wt not only in the G0/G1 but also in the S phases. Therefore, the E2F-1/pRB complex also regulated these genes during S phase.
Models for the E2F-1 and pRb roles for target gene expression. Models were designed from the data presented in this paper. While repressor E2F complex generic types is the most appropriate for the target genes including, DHFR, b-myb, cdc-2, Cyclin D1, C-myc and TK in cycling cells, the inhibited E2F complex is the most appropriate generic types of E2F complexes for the RB. Possible models were designed for individual genes in cells re-entering the cycle according to the results.
Previously it was known that pRB is phosphorylated during S phase in the cells overexpressing E2F-1/wt, and therefore E2F-1 would not be complexed with pRB. This implies that pRB would not have a function in the regulation of target genes in S phase. Our results contradict most of the previous studies in which it was shown that during S phase, the E2F-1 target gene promoter was not protected by a protein complex [46, 47]. However, results of this study showing that DHFR, cdc-2, and TK are regulated during S phase, albeit to a lesser extent than in G0/G1 are mostly consistent with the finding of Wells et al. [48, 49]. They showed with chromosomal immunoprecipitation that some of the genes have protein complexes on the E2F-1 sites not only at G0/G1 but also at S phase.
The E2F-1/pRB complex regulates c-myc only during G0/G1 phase, and no significant effect of the E2F-1/pRB complex on c-myc is seen during S phase. Since c-myc expression is lower in E2F-1/wt and E2F-1/411-overexpressing cells compared to control, active repressor complex model is the most appropriate for regulation of c-myc by the E2F-1/pRB complex and E2F-1 (Fig. 5 and 6).
Although cyclin D1 showed growth regulation in both cell lines overexpressing E2F-1/wt and E2F-1/Y411A, the E2F-1/pRB complex does not play any role in this regulation.
Regulation of RB gene expression was more complex. Contrary to previous reports, our study showed that E2F-1 activates RB expression in asynchronously growing cells. The E2F-1/411-mutant-overexpressing cells showed that in G0/G1, free E2F-1 decreased RB expression, but the opposite happened in S phase. This result may indicate that the RB expression was controlled by E2F-1/pRB and E2F-1 was inhibited by pRB during S phase but not in G0/G1. This fits well with the role of pRB in the cell cycle. During G0/G1, a cell needs pRB to repress genes involved in cell proliferation. However, in S phase, pRB effect should be reduced on the target gene promoter so that gene expression may occur. This might be another way of regulating transcription-related proteins by pRB in S phase, distinct from pRB phosphorylation.
It is possible that some additional factors may be involved with the E2F-1/pRB complex. For example, Udvadia et al. showed that pRB and Sp1 increased the activation of cyclin D1 [50]. It was presented previously [50] that RB promoter also has Sp1 site on promoter close to one of the E2F-1 site and deletion of Sp1 site decreased the activity of the promoter. Although, not significantly, our data also shows that cyclin D1 expression in the E2F-1/wt- expressing cell is higher than that of E2F-1/411 similar to the pRB expression in the cells re-entering the cycle. These may suggest that pRB may be an activator of certain promoters that contain Sp1 sites. Similar finding was also reported previously by Wells et all [48].
These novel findings about interaction of E2F-1 -pRb complex at different cell cycle provide new insight into the understanding of the molecular mechanism of pRB-E2F-1 interaction. In addition, this finding may represents a new characteristic of the transcription factors, which may act differently at different part of the cell cycle.
Conflict of Interest
There are no conflicts of interest and financial disclosure in the subject matter of this paper.
References
1. Hallstrom TC, Nevins JR. Balancing the decision of cell proliferation and cell fate. Cell Cycle. 2009;8:532-535
2. Helin K, Harlow E, Fattaey A. Inhibition of E2F-1 transactivation by direct binding of the retinoblastoma protein. Mol Cell Biol. 1993;13:6501-6508
3. Johnson DG, Ohtani K, Nevins JR. Autoregulatory control of E2F1 expression in response to positive and negative regulators of cell cycle progression. Genes Dev. 1994;8:1514-1525
4. Sherr CJ. The Pezcoller lecture: cancer cell cycles revisited. Cancer Res. 2000;60:3689-3695
5. Dyson N. The regulation of E2F by pRB-family proteins. Genes Dev. 1998;12:2245-2262
6. DeGregori J, Johnson DG. Distinct and Overlapping Roles for E2F Family Members in Transcription, Proliferation and Apoptosis. Curr Mol Med. 2006;6:739-748
7. Yee AS, Reichel R, Kovesdi I, Nevins JR. Promoter interaction of the E1A-inducible factor E2F and its potential role in the formation of a multi-component complex. Embo J. 1987;6:2061-2068
8. Muller H, Bracken AP, Vernell R. et al. E2Fs regulate the expression of genes involved in differentiation, development, proliferation, and apoptosis. Genes Dev. 2001;15:267-285
9. Ren B, Cam H, Takahashi Y. et al. E2F integrates cell cycle progression with DNA repair, replication, and G(2)/M checkpoints. Genes Dev. 2002;16:245-256
10. Weinmann AS, Yan PS, Oberley MJ, Huang TH, Farnham PJ. Isolating human transcription factor targets by coupling chromatin immunoprecipitation and CpG island microarray analysis. Genes Dev. 2002;16:235-244
11. Black AR, Azizkhan-Clifford J. Regulation of E2F: a family of transcription factors involved in proliferation control. Gene. 1999;237:281-302
12. Weintraub SJ, Prater CA, Dean DC. Retinoblastoma protein switches the E2F site from positive to negative element. Nature. 1992;358:259-261
13. Weintraub SJ, Chow KN, Luo RX, Zhang SH, He S, Dean DC. Mechanism of active transcriptional repression by the retinoblastoma protein. Nature. 1995;375:812-815
14. Helin K. Regulation of cell proliferation by the E2F transcription factors. Curr Opin Genet Dev. 1998;8:28-35
15. Muller H, Helin K. The E2F transcription factors: key regulators of cell proliferation. Biochim Biophys Acta. 2000;1470:M1-12
16. Batsche E, Lipp M, Cremisi C. Transcriptional repression and activation in the same cell type of the human c-MYC promoter by the retinoblastoma gene protein: antagonisation of both effects by SV40 T antigen. Oncogene. 1994;9:2235-2243
17. Jensen DE, Black AR, Swick AG, Azizkhan JC. Distinct roles for Sp1 and E2F sites in the growth/cell cycle regulation of the DHFR promoter. J Cell Biochem. 1997;67:24-31
18. Jun D, Park HK, Nordin AA, Nagel JE, Kim YH. Characterization of the murine cdc2 gene. Mol Cells. 1998;8:731-740
19. Lam EW, Bennett JD, Watson RJ. Cell-cycle regulation of human B-myb transcription. Gene. 1995;160:277-281
20. van Ginkel PR, Hsiao KM, Schjerven H, Farnham PJ. E2F-mediated growth regulation requires transcription factor cooperation. J Biol Chem. 1997;272:18367-18374
21. Shan B, Lee WH. Deregulated expression of E2F-1 induces S-phase entry and leads to apoptosis. Mol Cell Biol. 1994;14:8166-8173
22. Shan B, Durfee T, Lee WH. Disruption of RB/E2F-1 interaction by single point mutations in E2F-1 enhances S-phase entry and apoptosis. Proc Natl Acad Sci U S A. 1996;93:679-684
23. Sladek TL, Jacobberger JW. Rapid titration of retroviral vectors encoding intracellular antigens by flow cytometry. J Virol. 1990;64:3135-3138
24. Sladek TL, Jacobberger JW. Dependence of SV40 large T-antigen cell cycle regulation on T-antigen expression levels. Oncogene. 1992;7:1305-1313
25. Helin K, Lees JA, Vidal M, Dyson N, Harlow E, Fattaey A. A cDNA encoding a pRB-binding protein with properties of the transcription factor E2F. Cell. 1992;70:337-350
26. Sladek TL, Jacobberger JW. Simian virus 40 large T-antigen expression decreases the G1 and increases the G2 + M cell cycle phase durations in exponentially growing cells. J Virol. 1992;66:1059-1065
27. Landau NR, Littman DR. Packaging system for rapid production of murine leukemia virus vectors with variable tropism. J Virol. 1992;66:5110-5113
28. Jacobberger JW, Fogleman D, Lehman JM. Analysis of intracellular antigens by flow cytometry. Cytometry. 1986;7:356-364
29. Yang XH, Sladek TL. Novel phosphorylated forms of E2F-1 transcription factor bind to the retinoblastoma protein in cells overexpressing an E2F-1 cDNA. Biochem Biophys Res Commun. 1997;232:336-339
30. Lukas J, Petersen BO, Holm K, Bartek J, Helin K. Deregulated expression of E2F family members induces S-phase entry and overcomes p16INK4A-mediated growth suppression. Mol Cell Biol. 1996;16:1047-1057
31. DeGregori J, Leone G, Ohtani K, Miron A, Nevins JR. E2F-1 accumulation bypasses a G1 arrest resulting from the inhibition of G1 cyclin-dependent kinase activity. Genes Dev. 1995;9:2873-2887
32. Wang ZM, Yang H, Livingston DM. Endogenous E2F-1 promotes timely G0 exit of resting mouse embryo fibroblasts. Proc Natl Acad Sci U S A. 1998;95:15583-15586
33. Johnson DG, Cress WD, Jakoi L, Nevins JR. Oncogenic capacity of the E2F1 gene. Proc Natl Acad Sci U S A. 1994;91:12823-12827
34. Yang XH, Sladek TL. Overexpression of the E2F-1 transcription factor gene mediates cell transformation. Gene Expr. 1995;4:195-204
35. Hurford RKJr, Cobrinik D, Lee MH, Dyson N. pRB and p107/p130 are required for the regulated expression of different sets of E2F responsive genes. Genes Dev. 1997;11:1447-1463
36. Tao Y, Kassatly RF, Cress WD, Horowitz JM. Subunit composition determines E2F DNA-binding site specificity. Mol Cell Biol. 1997;17:6994-7007
37. Chestukhin A, Litovchick L, Rudich K, DeCaprio JA. Nucleocytoplasmic shuttling of p130/RBL2: novel regulatory mechanism. Mol Cell Biol. 2002;22:453-468
38. Puri PL, Cimino L, Fulco M. et al. Regulation of E2F4 mitogenic activity during terminal differentiation by its heterodimerization partners for nuclear translocation. Cancer Res. 1998;58:1325-1331
39. Tedesco D, Lukas J, Reed SI. The pRb-related protein p130 is regulated by phosphorylation-dependent proteolysis via the protein-ubiquitin ligase SCF(Skp2). Genes Dev. 2002;16:2946-2957
40. Bhattacharya S, Garriga J, Calbo J, Yong T, Haines DS, Grana X. SKP2 associates with p130 and accelerates p130 ubiquitylation and degradation in human cells. Oncogene. 2003;22:2443-2451
41. Sun A, Bagella L, Tutton S, Romano G, Giordano A. From G0 to S phase: a view of the roles played by the retinoblastoma (Rb) family members in the Rb-E2F pathway. J Cell Biochem. 2007;102:1400-1404
42. Alvaro-Blanco J, Martinez-Gac L, Calonge E. et al. A novel factor distinct from E2F mediates C-MYC promoter activation through its E2F element during exit from quiescence. Carcinogenesis. 2009;30:440-448
43. Moberg KH, Logan TJ, Tyndall WA, Hall DJ. Three distinct elements within the murine c-myc promoter are required for transcription. Oncogene. 1992;7:411-421
44. Campanero MR, Armstrong M, Flemington E. Distinct cellular factors regulate the c-myb promoter through its E2F element. Mol Cell Biol. 1999;19:8442-8450
45. Liu N, Lucibello FC, Zwicker J, Engeland K, Muller R. Cell cycle-regulated repression of B-myb transcription: cooperation of an E2F site with a contiguous corepressor element. Nucleic Acids Res. 1996;24:2905-2910
46. Lam EW, Watson RJ. An E2F-binding site mediates cell-cycle regulated repression of mouse B-myb transcription. Embo J. 1993;12:2705-2713
47. Lanker S, Valdivieso MH, Wittenberg C. Rapid degradation of the G1 cyclin Cln2 induced by CDK-dependent phosphorylation. Science. 1996;271:1597-1601
48. Wells J, Boyd KE, Fry CJ, Bartley SM, Farnham PJ. Target gene specificity of E2F and pocket protein family members in living cells. Mol Cell Biol. 2000;20:5797-5807
49. Stengel KR, Thangavel C, Solomon DA, Angus SP, Zheng Y, Knudsen ES. RB/p107/130 pocket proteins: Protein dynamics and interactions with target gene promoters. J Biol Chem. 2009;284:19265-19271
50. Udvadia AJ, Templeton DJ, Horowitz JM. Functional interactions between the retinoblastoma (Rb) protein and Sp-family members: superactivation by Rb requires amino acids necessary for growth suppression. Proc Natl Acad Sci U S A. 1995;92:3953-3957
Author contact
Corresponding author: Fikret Sahin, MD. PhD., Microbiology Department, School of Medicine, Ankara University, Basic Science Buildings, 3rd floor, Sihhiye-Ankara, Turkey 06100. Home: 011 90-312 491 1951; Cell phone: 0535 721 2043; Fax: 011 90- 312 310 6370; fsahin29com.

 Global reach, higher impact
Global reach, higher impact