Impact Factor ISSN: 1449-2288
Int J Biol Sci 2013; 9(10):1070-1078. doi:10.7150/ijbs.6662 This issue Cite
Research Paper
Cells Isolated from Human Periapical Cysts Express Mesenchymal Stem Cell-like Properties
1. Unit of Maxillofacial Surgery, Calabrodental, Crotone, Italy;
2. Tecnologica Research Institute, Biomedical Section, Crotone, Italy.
* All authors equally contributed to this research.
Received 2013-5-9; Accepted 2013-11-5; Published 2013-11-16
Abstract
We provide a detailed description of mesenchymal stem cells (MSCs) isolated from human periapical cysts, which we have termed hPCy-MSCs. These cells have a fibroblast-like shape and adhere to tissue culture plastic surfaces. hPCy-MSCs possess high proliferative potential and self-renewal capacity properties. We characterised the immunophenotype of hPCy-MSCs (CD73+, CD90+, CD105+, CD13+, CD29+, CD44+, CD45-, STRO-1+, CD146+) by flow cytometry and immunofluorescence. hPCy-MSCs possess the potential to differentiate into osteoblast- and adipocyte-like cells in vitro. Multi-potentiality was evaluated with culture-specific staining and quantitative reverse transcription-polymerase chain reaction (qRT-PCR) analysis for osteo/odontogenic and adipogenic markers. This is the first report to indicate that human periapical cysts contain cells with MSC-like properties. Taken together, our findings indicate that human periapical cysts could be a rich source of MSCs.
Keywords: Mesenchymal stem cells (MSCs), cyst, periapical inflammatory tissues, mesenchymal stem cells isolated from human periapical cyst (hPCy-MSCs), osteogenic differentiation, adipogenic differentiation, odontogenic cyst.
Introduction
Human periapical inflammatory tissues that are formed as a consequence of endodontic infections are commonly termed periapical granuloma, and the condition is referred to as apical periodontitis.
Periapical cysts are a direct outcome of chronic apical periodontitis; however, not every chronic lesion develops into a cyst, and the pathogenic pathways leading to cyst formation may vary under different circumstances.
Histologically, periapical inflammatory tissues are typically fibrous and are infiltrated by inflammatory cells, including macrophages, neutrophils, and lymphocytes [1]. Once the infection is cured, the surrounding tissues will undergo a healing process, and a variable amount of new bone will be regenerated in the area affected by the inflammatory osteolysis.
A previous study by Maeda et al. [2] showed that some periapical lesions may contain osteogenic cells that have the potential to differentiate into mature osteoblastic cells; undoubtedly, such cells led to better osseous healing after treatment of the infection, thereby affecting the dental pulp. Although the typology of these cells was not characterised specifically, these results nonetheless suggested that the periapical granulation tissue contains a number of cells that function as osteogenic progenitors. Another interesting study demonstrated that mesenchymal stem cells (MSCs) were present in the granulation tissue that developed in response to foreign bodies in an animal model [3].
Periapical cysts are the most common odontogenic cystic lesions of inflammatory origin. These cysts are found at the root apices of involved teeth, and are defined as odontogenic periapical cysts of inflammatory origin that are preceded by a chronic periapical granuloma [4].
Recently, Liao et al. [5] showed that human periapical inflammatory tissues expressed MSC markers, suggesting the presence of MSCs. They further demonstrated that isolated cells exhibited a mesenchymal cell immunophenotype with the capacity to differentiate into osteogenic and adipogenic lineages.
To date, several types of MSCs have been isolated from teeth, including dental pulp stem cells (DPSCs) [6], stem cells from human exfoliated deciduous teeth (SHEDs) [7], periodontal ligament stem cells (PDLSCs) [8], dental follicle progenitor stem cells (DFPCs) [9], and stem cells from apical papilla (SCAPs) [10]. In addition, it was recently demonstrated that mesenchymal stem/progenitor cells could be successfully isolated from inflamed adult human dental pulp [11] and inflamed human periodontal ligament (PDL) tissue [12]. This latter study also demonstrated that MSCs isolated from pathological dental tissues possess many similarities with MSCs isolated from healthy tissues.
Therefore, in light of the results of previous studies, and in the search for alternative sources of MSCs, inflamed periapical cysts were assessed for their potency. The aim of this study was to determine whether cells isolated from human periapical inflammatory cysts could express MSC-like properties. Herein, we describe MSCs isolated from inflamed cysts (hPCy-MSCs) and demonstrate their highly proliferative capacity and differentiation into osteogenic and adipogenic lineages in vitro. Therefore, we suggest that hPCy-MSCs may represent a rich source of MSCs that could be used in human regenerative medicine.
Materials and methods
Cell preparation and culture
Human periapical cystic tissue was obtained from 12 healthy male volunteers, 25 ± 3 years of age, who provided informed consent at the Calabrodental dental clinic (Crotone, Italy). The study was carried out under approved guidelines of the Ethical Committee. Before teeth extraction, every subject was checked for oral infections or other diseases, and all subjects were pre-treated with professional dental hygiene one week before surgery, followed by daily oral rinses with mouthwash containing 0.12% Chlorhexidine. The same surgical protocol and surgeon were used in all 12 treated cases. The enucleated cyst was immediately placed in sterile physiological saline buffer and transferred to the laboratory (Tecnologica srl Research Institute, Crotone, Italy) within 5 min, owing to the proximity between the dental clinic and research laboratories. The odontogenic periapical cysts were immediately washed several times in sterile phosphate-buffered saline (PBS; Invitrogen) containing antibiotics. The culture of cells isolated from cystic tissue was conducted as described in previous reports [6, 13, 14].
The entire cystic wall, regardless of whether the cyst was complete or broken up, was removed and placed in 10-mm tissue culture dishes for mechanical disruption. Samples were minced into small pieces (approximately 0.1 mm in diameter) with a sterile Bard-Parker scalpel with a 15c blade, placed in the growth medium, and digested in a solution containing 3 mg/mL type I collagenase and 4 mg/mL dispase (Sigma) in PBS for 1 h at 37°C. The solution was then filtered through 70-μM Falcon strainers (Becton and Dickinson). After filtration, the cells were pelleted, seeded in culture dishes, and incubated in alpha-minimal essential medium (α-MEM) culture medium with 10% foetal bovine serum (FBS; Invitrogen), 2 mM glutamine, 100 μM L-ascorbic acid-2-phosphate, 100 U/ml penicillin, and 100 mg/ml streptomycin (Invitrogen). Cells were incubated at 37°C and 5% CO2 and the medium was changed twice a week.
Cell counting
To determine the population doubling (PD) rate, cells isolated from human odontogenic periapical cysts were initially seeded at a density of 150 × 103 cells/well in a 6-well plate in culture medium. Cells were passaged and counted once they reached a sub-confluence of 80%. At each passage, cells were re-plated at the initial density and cultures were maintained until passage 11. The following formula was applied: PD = [log10 (N) - log10(Ns)]/ log10(2), where N is the harvested cell number and Ns is the initial plated cell number [15]. The cumulative population doubling (cPD) index for each passage was obtained by adding the PD of each passage to the PD of the previous passages.
Self-renewal capacity: colony-forming unit-fibroblast (CFU-F) assays
A CFU assay was performed to determine the presence of putative MSCs in the tissues taken from odontogenic periapical cysts. The clonogenic potential of cells isolated from human periapical cysts was tested by the CFU-F assay [16, 17]. The cells were seeded into a 6-well plate (Costar) at 100 cells/well. After 10 days of culture, the cells were fixed with 4% paraformaldehyde (Sigma) and then stained with 0.1% toluidine blue (Sigma). Aggregates of more than 50 cells were scored as colonies. Cells were observed under a light microscope (Leica). All experiments were performed in triplicate.
Flow cytometric analysis
Cells were trypsinised, resuspended, and aliquoted at 500,000/100 μL PBS into a fluorescence-activated cell sorting (FACS) tube. Each aliquot was incubated with saturating concentrations of fluorochrome-conjugated antibodies on ice for 30 min. At the end of incubation, the tube was centrifuged at 200 × g for 5 min and inverted onto a paper towel to drain the supernatant. The cells were washed twice, and 0.5 mL of PBS was added to each tube. The samples were kept on ice in the dark until used for FACS. CD73-FITC, CD90-PE, CD105-APC, CD13-PE, CD29-APC, CD44-FITC, and CD45-APC-H7 (Becton Dickinson - BD) antibodies were used for our analysis. Samples were analysed with FACSCanto II instruments (BD), and data were analysed by using FlowJo software (Tree Star).
Confocal microscopy
Fixed monolayer cells were permeabilised with 0.1% Triton X-100 in PBS for 5 min. Samples were then blocked with 3% BSA in PBS for 30 min at room temperature and then incubated with fluorescein isothiocyanate (FITC)-conjugated STRO-1 (Santa Cruz) and with Phycoerythrin (PE)-conjugated CD146 (Santa Cruz) antibodies, diluted at 1:50 in PBS containing 3% BSA, for 1 h at room temperature. After washing in PBS, samples were stained with 1 mg/mL 4,6-diamidino-2-phenylindole (DAPI; Sigma) in PBS for 1 min, and then mounted with anti-fading medium (ProLong Antifade; Invitrogen) and visualised by confocal microscopy (Leica; TCS SP5). For detection, the samples were sequentially excited with the following laser wavelengths: 405 nm lines of a diode laser for DAPI and 488 nm lines of the argon laser for FITC and PE. The excitation and the detection of the samples were carried out in sequential mode to avoid overlapping of the signals. Optical sections were obtained at increments of 0.3 mm in the Z-axis, and were digitized with a scanning mode format of 1024 × 1024 pixels.
In vitro osteogenic differentiation
Cells were detached with 0.25% trypsin-EDTA, resuspended in growth medium, and plated at 1 × 104 cells/well in a 96-well plate. The next day, the growth medium was replaced by osteogenic medium [α-MEM (Sigma), 20% FBS (Invitrogen), 0.2 mM L-ascorbic acid-2-phosphate (Sigma), 100 nM dexamethasone (Sigma), 10 mM β-glicerophosphate (Sigma), 100 U/mL penicillin, 0.1 mg/mL streptomycin, and 0.25 mg/mL amphotericin B]. The cells were grown for various periods of time and the medium was changed twice a week [18]. α-MEM supplemented with 10% FBS was used in the control group. Cells isolated from human cystic tissue, which were grown in osteogenic medium for 3 weeks, were washed once with PBS and fixed with 4% paraformaldehyde (Sigma) for 15 min at room temperature. After washing with PBS three times, an aqueous solution of 5 mg/mL Alizarin Red S (Sigma) was added to the cells for 30 min. Then, cells were washed with H2O three times for 5 min each while shaking, and were analysed by microscopy. For quantification, the Alizarin Red precipitates were solubilized. Briefly, stained samples were incubated with 800 mL acetic acid (10%) for 30 min. Then, the supernatant was transferred into a 1.5-mL tube and boiled for 10 min at 85°C. After centrifugation (15 min, 15,000 × g), 150 μL samples were transferred into a 96-well microtiter plate and the optical density was measured at 405 nm using a Multiskan FC ELISA reader (Thermo Scientific).
In vitro adipogenic differentiation
For adipogenesis, the cultures were incubated in α-MEM supplemented with 10% FBS, 100 U/mL penicillin, 100 μg/mL streptomycin, 12 mM L-glutamine, 10 μM insulin (Sigma), 200 μM indomethacin (Sigma), 1 μM dexamethasone, and 0.5 mM 3-isobutyl-1-methylxanthine (IBMX; Sigma). The medium was changed twice a week for 3 weeks. α-MEM supplemented with 10% FBS was used in the control group.
Adipogenic differentiation was evaluated using Oil Red O staining (Sigma), which shows the presence of triglyceride deposits. In brief, for evaluating the generation of oil droplets, the hPCy-MSCs were fixed in 10% formalin for 10 min at room temperature and washed twice with water. Oil Red O (Sigma-Aldrich) working solution was prepared by adding 6 mL of stock solution (0.5 g Oil Red in 100 mL isopropanol) to 4 mL distilled water, mixed, and filtered through Whatman filter paper. Next, Oil Red O stain was added and incubated for 1 h at room temperature. Finally, the cells were rinsed several times with water and observed under an inverted microscope [19-21].
RNA preparation and quantitative reverse transcription-polymerase chain reaction (qRT-PCR) analysis
RNA extraction was performed with the Purelink™ RNA mini kit (Applied Biosystems), following the manufacturer's instructions, and total RNA was quantified by using a Multiskan Go spectrophotometer (Thermo scientific). Total RNA samples (250 ng) were subjected to the reverse-transcription reaction using the High Capacity RNA-to-cDNA Kit (Applied Biosystems), following the manufacturer's instructions. Five hundred nanolitres of cDNAs were amplified by real-time PCR with the power SYBR green PCR Master Mix (Applied Biosystems) with 2 pmol of primers in a total volume of 10 µL.
Real-time PCR reactions were carried out in a Pikoreal 96 (Thermo scientific) apparatus with the following conditions: initial denaturation step at 95°C for 10 min, followed by 40 cycles of 10 s at 95°C and 1 min at 60°C. The specificity of PCR products was checked by melting curve analysis and gel electrophoresis. The efficiencies of real-time PCRs were calculated by constructing cycle threshold (Ct) curves obtained from amplifications of serial ten-fold dilutions of a cDNA sample, which showed that the efficiencies were higher than 90%. The expression of each gene was determined from the Ct value, and relative expression levels were calculated using the ΔΔCt method after normalization to the expression of the HRPT housekeeping gene. Primer sequences for the adiponectin (ADIPOQ), alkaline phosphatase (ALP), dentin matrix protein 1 (DMP-1), glucose transporter type 4 (GLUT4), hypoxanthine phosphoribosyltransferase (HPRT), lipoprotein lipase (LPL), osteocalcin (OSC), osteopontin (OPN), and peroxisome proliferator-activated receptor gamma (PPARg) genes are listed in Table 1. The experiment was performed three times.
Oligonucleotide primer sequences utilized in the qRT-PCR analysis.

Statistical analysis
Results are expressed as the mean ± standard deviation (SD) of three separate experiments. Statistical analysis was performed using the Student's t-test with the GraphPad Prism statistical package (version 5.0), and differences were considered statistically significant at P < 0.05 (*), P < 0.01 (**), and P < 0.001 (***).
Results
Isolation and growth of hPCy-MSCs
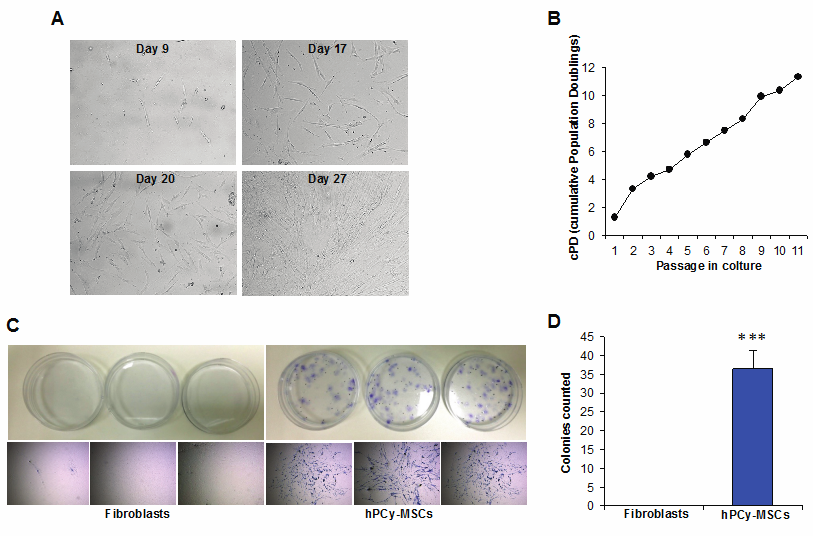
The first plastic-adherent cells were detected one week after tissue preparation. Primary cell cultures reached a confluence of 70-90% at day 27 (Figure 1A). Cells from human inflamed oral cysts were seeded at Low density on a 6-well plate and allowed to grow until 80% confluence. Cells were passaged at the same cell density. The PD rate was calculated at every passage to determine the ultimate PDs and the cPDs (Figure 1B) until the cells ceased dividing. During passaging, the cell viability and cell numbers were measured. Cell kinetics characterisation with cPDs was used to identify changes occurring during the growth of cells isolated from human oral inflamed periapical cysts.
Morphological characterization, growth, and self-renewal capacity of hPCy-MSCs. A) Growth of human periapical cyst mesenchymal stem cells (hPCy-MSCs) at passage 0 at indicated days. B) Cumulative population doublings (cPDs) of cells isolated from oral cysts. The number of population doublings was calculated as follows: PD = [log10(N) - log10(Ns)] / log10(2), where N is the harvested cell number and Ns is the initial plated cell number. The cPD index for each passage was obtained by adding the PD of each passage to the PD of the previous passages. C) Representative CFU-F assay. Fibroblasts and hPCy-MSCs (100 cells each) were cultured in 60-mm dishes. After 10 days of culture, the cells were fixed with 4% paraformaldehyde and then stained with 0.1% toluidine blue. All experiments were performed in triplicate. D) Quantification of CFU-F from fibroblasts and cells isolated from inflamed cysts. Aggregates of more than 50 cells were scored as colonies. The data shown represent means ± SDs, ***P < 0.001 using the unpaired Student's t-test.
Self-renewal capacity of hPCy-MSCs
The in vitro self-renewal capacity of the hPCy-MSCs was demonstrated based on the CFU-F assay, which provides a convenient means of assessing the proliferative capacity that MSCs retain after the cells have been expanded in culture [22].
Therefore, cells isolated from human periapical cysts that were expanded to passage 5 were subjected to the CFU-F assay, in which 100 cells were deposited into a 6-well plate. The results of the CFU-F assays demonstrated that cultures contained subpopulations of cells capable of generating new colonies from single cells (Figure 1C). As indicated in Figure 1D, we obtained several hPCy-MSC colonies (36 ± 4), whereas we did not observe any colonies in the fibroblasts used as controls. These results suggested that some of the cells from the initially formed colonies detached and generated new colonies. Similar results were previously observed by Liao et al. [5] in CFU-F experiments using cells isolated from inflamed periapical tissues.
Immunophenotype of hPCy-MSCs
Specific cell-surface markers were selected to evaluate whether the isolated cells contained the MSC population. These experiments were carried out with the goal of isolating a population of pluripotent-like stem cells derived from human oral inflamed cysts. In order to characterise the population, we performed a flow cytometric analysis to detect the expressions of markers in cells isolated from inflamed oral cysts. We found that the population was CD73+, CD90+, CD105+, CD13+, CD29+, CD44+, and CD45- (Figure 2A), which indicated characteristics of MSCs [6, 23, 24]. The cells at passage 2 expressed high levels of CD73 (99.6%), CD90 (100%), CD105 (36.6%), CD13 (100%), CD29 (100%), and CD44 (99.6%). However, they stained negatively for the hematopoietic marker CD45 (1.2%), indicating the mesenchymal lineage of these cells.
Cell surface markers of hPCy-MSCs. A) Immunophenotype by FACS analysis of cells isolated from oral cysts. A heterogeneous, pooled population of cells at passage 4 was analysed with flow cytometry to detect the expressions of cell surface markers. Results reported forward and side scatter, and in each plot, the X-axis shows relative fluorescence and the Y-axis shows the number of events. Histograms show the isotype control (red peaks) versus specific antibody staining (blue peaks) and the percentage of cells positive for the selected molecules. B) Immunofluorescence analysis of STRO-1 and CD146 expressions in hPCy-MSCs. Surface antigens were stained with fluorescein isothiocyanate (FITC, green) for STRO-1 and Phycoerythrin (PE, red) for CD146, and the nuclei were stained with 4,6-diamidino-2-phenylindole (DAPI, blue); bar scale = 100 μm.
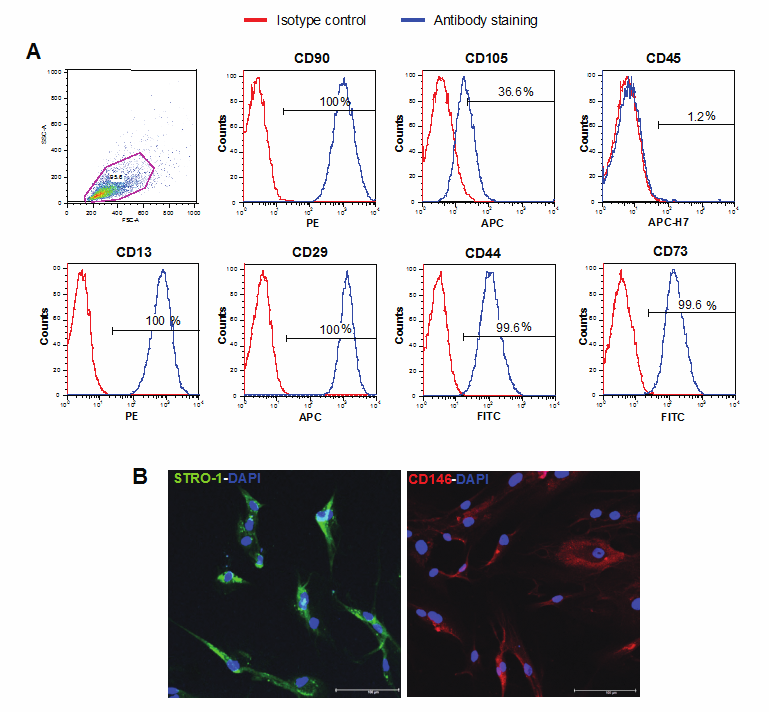
In order to further characterise the population, the expressions of STRO-1 and CD146 were analysed by confocal microscopy. The isolated cell populations expressed the cell surface molecules STRO-1 and CD146, two early MSC markers that are present on both DPSCs and bone marrow MSCs [25, 26] (Figure 2B). Together, these results demonstrate that cells isolated from inflamed periapical cyst tissues have MSC characteristics with respect to the expressions of markers.
Differentiation in mesenchymal lineages
In addition to their colony forming ability and the expression of specific antigens on the cell surface, the capacity for differentiation into different mesenchymal tissues is one of the key properties of MSCs [27]. The differentiation potential of hPCy-MSCs was evaluated by culturing the cells in osteogenic and adipogenic media. Long-term cultures (3 weeks) of cells grown in the presence of osteogenic media demonstrated the capacity to form Alizarin Red-positive condensed nodules with high levels of calcium covering the entire wells (Figure 3A). The deposits were sparsely scattered throughout the adherent layer as single mineralized zones (Figure 3B). To quantify the differentiation status, the stain from all cells was eluted and the absorbance was measured. The differentiated cells displayed approximately 34-fold greater values than control cells (Figure 3C).
Osteogenic differentiation of hPCy-MSCs. A) Mineralized deposit identified by Alizarin Red staining in cells grown in osteogenic medium for 3 weeks. Undiff, untreated control; Osteogenic, osteogenic stimulation. B) Alizarin Red staining of hPCy-MSCs as viewed under the light microscope at 10× magnification, panel B (right) displays a higher-magnification image of cells. Scale bar = 100 μm. C) Quantification of Alizarin Red staining via dissolving the dye and subsequent absorption measurement (405 nm); *** P < 0.001 compared to untreated control cells (Undiff) by the unpaired two-tailed Student's t-test. D) qRT-PCR was used to determine the difference in gene expression profiles of osteoblastic-specific markers (ALP, OPN, OSC, DMP-1) between 1 month osteoblastic-differentiated cells and undifferentiated cells (Undiff). Relative expression levels were calculated by using the 2-ΔΔCT method. Values are expressed as mean ± SD and were normalized using HPRT expression levels. The Y-axis shows the fold-change in expression and the X-axis shows the tested markers, *P < 0.05 compared with undifferentiated cells based on the two-tailed Student's t-test.
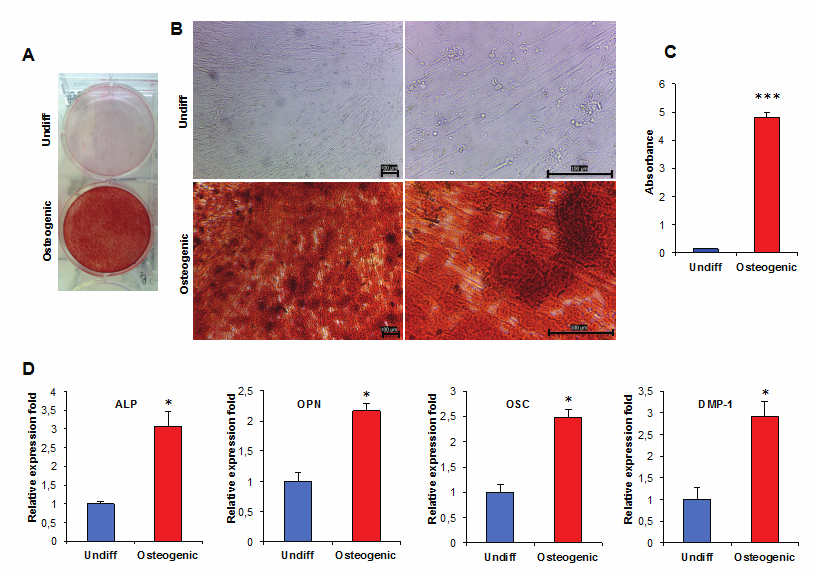
In addition, the expression patterns of osteo-specific genes, including the alkaline phosphatase (ALP), osteopontin (OPN), osteocalcin (OSC), and dentin matrix protein 1 (DMP-1) genes, were analysed at the mRNA level after 3 weeks of osteo-induction [28, 29].
The OPN and DMP-1 proteins are present in mineralized tissues such as bone, dentin, and cementum [30]. The real-time RT-PCR data, expressed as the amount in differentiated hPCy-MSCs relative to that in undifferentiated hPCy-MSCs, showed increased mRNA expression of ALP (3.07 ± 0.4-fold), OPN (2.17 ± 0.11-fold), OSC (2.47 ± 0.16-fold), and DMP-1 (2.91 ± 0.36-fold) (Figure 3D). In addition, the qRT-PCR results showed that hPCy-MSCs could express markers related to dentinogenesis, including DMP-1, ALP, and OPN [31]. These results suggest that hPCy-MSCs cultured in osteogenic media can differentiate into osteoblast/odontoblast-like cells.
To further confirm the differentiation capacity of hPCy-MSCs, adipogenic differentiation was induced. MSCs isolated from inflamed periapical cysts treated by the adipogenic differentiation protocol showed the presence of intracellular lipid droplets, which were confirmed by Oil Red O staining (Figure 4A and 4B). The hPCy-MSCs grown in culture medium alone (Undiff) did not show any lipid droplets at any of the time points examined, and maintained their typical fibroblast-like shape. We evaluated the gene expressions of the following specific adipogenic markers by real-time RT-PCR: glucose transporter type 4 (GLUT4), lipoprotein lipase (LPL), adiponectin (ADIPOQ), and peroxisome proliferator-activated receptor gamma (PPARg) [28, 32]. The gene expressions of all adipogenic markers were significantly higher in induced hPCy-MSCs compared to control cells. In fact, qRT-PCR demonstrated the up-regulation of GLUT4 (4.43 ± 0.77-fold), LPL (7.09 ± 0.75-fold), ADIPOQ (230.3 ± 21.57-fold), and PPARg (4.57 ± 0.38-fold) (Figure 4C).
Adipogenic differentiation of hPCy-MSCs. A) Untreated control hPCy-MSCs (Undiff) and adipogenic-induced cells (Adipogenic) were stained with Oil Red O at 21 days after induction. B) Adipogenic differentiation is evident by the formation of oil droplets stained with Oil Red O; panel B (right) displays a higher-magnification image of cells. Scale bar = 100 μm. C) Relative quantification of GLUT4, LPL, ADIPOQ, and PPARg expressions was evaluated by real-time PCR in untreated hPCy-MSCs (Undiff) and in adipogenic-induced cells (Adipogenic). Relative expression levels were calculated by using the 2-ΔΔCT method. Experiments were performed in triplicate and data are expressed as mean ± SD. Values were normalized to HPRT RNA levels. *P < 0.05, ** P < 0.01 compared with undifferentiated cells by unpaired two-tailed Student's t-test.
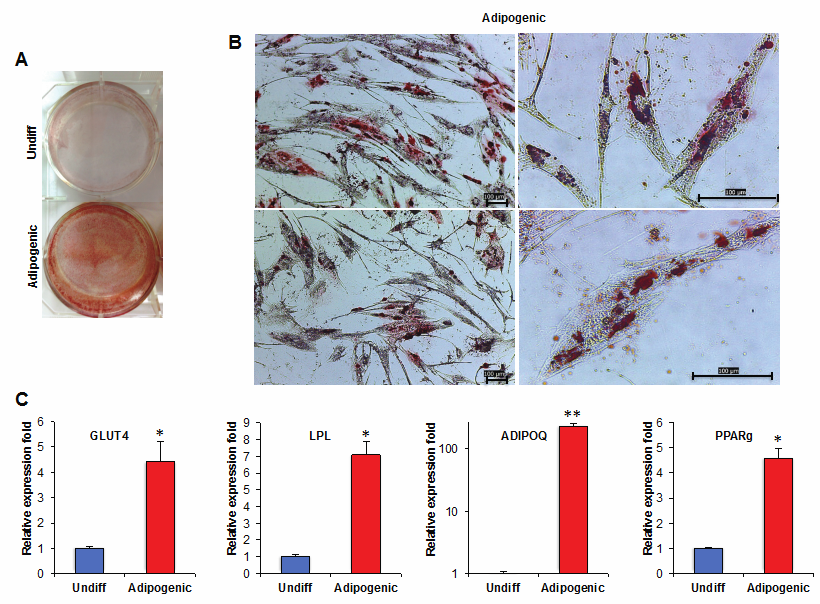
Taken together, these data showed the capacity of MSCs isolated form inflamed periapical cysts to undergo osteogenic and adipogenic differentiation upon in vitro induction. These results suggest that the hPCy-MSCs cell line established in this study has a high level of multipotency.
Discussion
No study has yet evaluated the possible existence of MSCs in human periapical cysts. A recent study demonstrated that mesenchymal progenitor cells were present in inflamed periapical tissue, which expressed markers of MSCs, were highly osteogenic, weakly adipogenic in vitro, and capable of forming mineralized tissue in vivo [5].
In the present study, we demonstrated that cells isolated from human periapical inflammatory cysts display MSC-like properties, as evidenced by the expression of the MSC markers CD13+, CD29+, CD44+, CD73+, CD90+, CD105+, CD45-, STRO-1+, CD146+ as well as their self-renewal capability and osteogenic and adipogenic potential.
The periapical inflammatory cysts were digested with an enzyme treatment for cell isolation. After digestion, hPCy-MSCs were grown for 20 passages. The cells displayed adherence to the plastic surface and their morphology was similar to that of typical MSCs with fibroblast-like cells [6]. To identify the characteristics of MSCs, we assayed their proliferation abilities and capacity to form colonies [6, 25]. The cell proliferation rate of hPCy-MSCs was evaluated via the calculation of cPDs from passage 1 to 11. hPCy-MSCs showed the ability to form CFU-Fs and multipotent differentiation capacity in vitro under various conditions. In the osteogenic differentiation experiment, hPCy-MSCs were positively stained with Alizarin Red and the expression levels of osteogenic marker genes increased after differentiation. We also found that the hPCy-MSCs could differentiate into adipogenic lineage cells in vitro. In fact, in the adipogenesis differentiation experiment, we confirmed that hPCy-MSCs displayed fatty droplets, which were positively stained with Oil Red O. In addition, we showed that the expressions of adipogenic-associated markers increased after induction of adipogenic differentiation.
Our data indicated that the investigated tissues, although usually intended for enucleation and storage, could also be considered as a source of cells with MSC-like properties. Furthermore, previous studies have demonstrated the presence of MSCs in granulated periapical tissue [3]; therefore, the differentiation in cystic fibrous tissue under continuous, untreated pulpo-periodontal infection does not preclude the possibility to find cells with stem cell-like characteristics. This could indicate co-responsibility in the process of bone regeneration to both the presence of osteogenic factors delivered by the blood and to the differentiation in osteoblast-like cells of the cellular elements present in the cyst wall, as demonstrated in our study.
In conclusion, we successfully isolated and characterised hPCy-MSCs from human periapical cysts, without recourse to the removal of biologically healthy tissues, since human periapical cysts are destined for surgical removal to prevent the development of disabling, pathological conditions.
The hPCy-MSCs were shown to possess self-renewal capacity and multi-lineage differentiation potency, which enable their differentiation into osteoblasts and adipocytes. Therefore, hPCy-MSCs could be a useful source of MSCs for research, and we further suggest that these cells have potential therapeutic properties for regenerative medicine and tissue engineering.
Acknowledgements
The present study was supported by “PROMETEO Project - Progettazione e Sviluppo di piattaforme tecnologiche innovative ed ottimizzazione di PROcessi per applicazioni in MEdicina rigenerativa in ambito oromaxillofaciale, emaTologico, nEurologico e cardiOlogico”. PON01_02834
Conflict of Interests
The authors declare that they have no conflict of interest.
References
1. Nair PN. Pathogenesis of apical periodontitis and the causes of endodontic failures. Crit Rev Oral Biol Med. 2004;15:348-81
2. Maeda H, Wada N, Nakamuta H, Akamine A. Human periapical granulation tissue contains osteogenic cells. Cell Tissue Res. 2004;315:203-8
3. Patel J, Gudehithlu KP, Dunea G, Arruda JA, Singh AK. Foreign body-induced granulation tissue is a source of adult stem cells. Transl Res. 2010;155:191-9
4. Shear M. Radicular and residual cysts. Cysts of the Oral Region, 3rd ed. Bristol UK: Wright. 1992:136-162
5. Liao J, Al Shahrani M, Al-Habib M, Tanaka T, Huang GT. Cells isolated from inflamed periapical tissue express mesenchymal stem cell markers and are highly osteogenic. J Endod. 2011;37:1217-24
6. Gronthos S, Mankani M, Brahim J, Robey PG, Shi S. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A. 2000;97:13625-30
7. Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, Robey PG. et al. SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci U S A. 2003;100:5807-12
8. Seo BM, Miura M, Gronthos S, Bartold PM, Batouli S, Brahim J. et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004;364:149-55
9. Morsczeck C, Gotz W, Schierholz J, Zeilhofer F, Kuhn U, Mohl C. et al. Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biol. 2005;24:155-65
10. Sonoyama W, Liu Y, Fang D, Yamaza T, Seo BM, Zhang C. et al. Mesenchymal stem cell-mediated functional tooth regeneration in swine. PLoS One. 2006;1:e79
11. Alongi DJ, Yamaza T, Song Y, Fouad AF, Romberg EE, Shi S. et al. Stem/progenitor cells from inflamed human dental pulp retain tissue regeneration potential. Regen Med. 2010;5:617-31
12. Park JC, Kim JM, Jung IH, Kim JC, Choi SH, Cho KS. et al. Isolation and characterization of human periodontal ligament (PDL) stem cells (PDLSCs) from the inflamed PDL tissue: in vitro and in vivo evaluations. J Clin Periodontol. 2011;38:721-31
13. Huang GT, Yamaza T, Shea LD, Djouad F, Kuhn NZ, Tuan RS. et al. Stem/progenitor cell-mediated de novo regeneration of dental pulp with newly deposited continuous layer of dentin in an in vivo model. Tissue Eng Part A. 2010;16:605-15
14. Huang GT, Sonoyama W, Chen J, Park SH. In vitro characterization of human dental pulp cells: various isolation methods and culturing environments. Cell Tissue Res. 2006;324:225-36
15. Mokry J, Soukup T, Micuda S, Karbanova J, Visek B, Brcakova E. et al. Telomere attrition occurs during ex vivo expansion of human dental pulp stem cells. J Biomed Biotechnol. 2010;2010:673513
16. Mensing N, Gasse H, Hambruch N, Haeger JD, Pfarrer C, Staszyk C. Isolation and characterization of multipotent mesenchymal stromal cells from the gingiva and the periodontal ligament of the horse. BMC Vet Res. 2011;7:42
17. Staszkiewicz J, Frazier TP, Rowan BG, Bunnell BA, Chiu ES, Gimble JM. et al. Cell growth characteristics, differentiation frequency, and immunophenotype of adult ear mesenchymal stem cells. Stem Cells Dev. 2010;19:83-92
18. Tuli R, Tuli S, Nandi S, Wang ML, Alexander PG, Haleem-Smith H. et al. Characterization of multipotential mesenchymal progenitor cells derived from human trabecular bone. Stem Cells. 2003;21:681-93
19. Wang HS, Hung SC, Peng ST, Huang CC, Wei HM, Guo YJ. et al. Mesenchymal stem cells in the Wharton's jelly of the human umbilical cord. Stem Cells. 2004;22:1330-7
20. Li WW, Wei YH, Li H, Lai DM, Lin TN. Isolation and characterization of a novel strain of mesenchymal stem cells from mouse umbilical cord: potential application in cell-based therapy. PLoS One. 2013;8:e74478
21. Park SB, Seo MS, Kim HS, Kang KS. Isolation and characterization of canine amniotic membrane-derived multipotent stem cells. PLoS One. 2012;7:e44693
22. Digirolamo CM, Stokes D, Colter D, Phinney DG, Class R, Prockop DJ. Propagation and senescence of human marrow stromal cells in culture: a simple colony-forming assay identifies samples with the greatest potential to propagate and differentiate. Br J Haematol. 1999;107:275-81
23. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315-7
24. Mao JJ, Prockop DJ. Stem cells in the face: tooth regeneration and beyond. Cell Stem Cell. 2012;11:291-301
25. Shi S, Gronthos S. Perivascular niche of postnatal mesenchymal stem cells in human bone marrow and dental pulp. J Bone Miner Res. 2003;18:696-704
26. Osathanon T, Sawangmake C, Nowwarote N, Pavasant P. Neurogenic differentiation of human dental pulp stem cells using different induction protocols. Oral Dis. 2013
27. Ren G, Chen X, Dong F, Li W, Ren X, Zhang Y. et al. Concise review: mesenchymal stem cells and translational medicine: emerging issues. Stem Cells Transl Med. 2012;1:51-8
28. Bressan E, Ferroni L, Gardin C, Pinton P, Stellini E, Botticelli D. et al. Donor age-related biological properties of human dental pulp stem cells change in nanostructured scaffolds. PLoS One. 2012;7:e49146
29. Ferro F, Spelat R, Beltrami AP, Cesselli D, Curcio F. Isolation and characterization of human dental pulp derived stem cells by using media containing low human serum percentage as clinical grade substitutes for bovine serum. PLoS One. 2012;7:e48945
30. Feng JQ, Huang H Lu Y, Ye L Xie Y. et al. The Dentin matrix protein 1 (Dmp1) is specifically expressed in mineralized, but not soft, tissues during development. J Dent Res. 2003;82:776-780
31. Guo L, Li J, Qiao X, Yu M, Tang W, Wang H. et al. Comparison of odontogenic differentiation of human dental follicle cells and human dental papilla cells. PLoS One. 2013;8:e62332
32. Battula VL, Chen Y, Cabreira Mda G, Ruvolo V, Wang Z, Ma W. et al. Connective tissue growth factor regulates adipocyte differentiation of mesenchymal stromal cells and facilitates leukemia bone marrow engraftment. Blood. 2013;122:357-66
Author contact
Corresponding author: Dr. Marco Tatullo, PhD. Scientific Director - Tecnologica Research Institute, St. E. Fermi - Crotone, Italy. Tel: 0962-930362; Fax: 0962930362; E-mail: marco.tatullocom.

 Global reach, higher impact
Global reach, higher impact