Impact Factor ISSN: 1449-2288
Int J Biol Sci 2015; 11(12):1376-1389. doi:10.7150/ijbs.13109 This issue Cite
Research Paper
Regulation of Androgen Receptor Expression Alters AMPK Phosphorylation in the Endometrium: In Vivo and In Vitro Studies in Women with Polycystic Ovary Syndrome
1. Department of Physiology/Endocrinology, Institute of Neuroscience and Physiology, The Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden.
2. Department of Gynecology, Obstetrics and Gynecology Hospital of Fudan University, Shanghai, China;
3. Shanghai Key Laboratory of Female Reproductive Endocrine Related Diseases, Shanghai, China;
4. Department of Integrative Medicine and Neurobiology, State Key Lab of Medical Neurobiology, Shanghai Medical College and Institute of Acupuncture Research (WHO Collaborating Center for Traditional Medicine), Institute of Brain Science, Fudan University, Shanghai, China.
* These authors contributed equally to the study.
Abstract
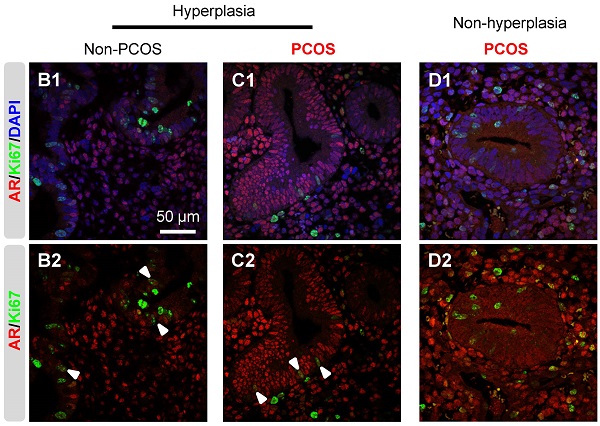
The failure of reproductive success in polycystic ovary syndrome (PCOS) patients could be in part due to endometrial dysfunction. However, no studies have investigated any causality between androgen, androgen receptor (AR) expression, and adenosine monophosphate activated protein kinase (AMPK) activation in the endometrium under physiological and pathological conditions. In the present study, we show that 1) endometrial AR expression levels fluctuate in non-PCOS and PCOS patients during the menstrual cycle; 2) the menstrual phase-dependent alteration of p-AMPKα expression occurs in non-PCOS patients but not in PCOS patients; 3) AR expression is higher in PCOS patients than non-PCOS patients during hyperplasia while AMPKα activation (indicated by the ratio of p-AMPKα to AMPKα); and 4) co-localization of AR and Ki-67 in epithelial cell nuclei is observed in endometrial hyperplasia. Importantly, using in vitro human tissue culture and an in vivo 5α-dihydrotestosterone-treated rat model, we show that the action of androgen on AMPKα activation is likely mediated through nuclear AR, especially in epithelial cells. Collectively, we present evidence that AR expression and AMPKα activation depend on menstrual cycle phase and the presence of PCOS, and the data suggest that AR-mediated regulation of AMPKα activation might play a role in the development of endometrial hyperplasia.
Keywords: 5α-dihydrotestosterone, androgen receptor, AMPKα, endometrium, PCOS.

 Global reach, higher impact
Global reach, higher impact