Impact Factor ISSN: 1449-2288
Int J Biol Sci 2018; 14(2):137-146. doi:10.7150/ijbs.23182 This issue Cite
Research Paper
A Prognostic Model Based on Circulating Tumour Cells is Useful for Identifying the Poorest Survival Outcome in Patients with Metastatic Colorectal Cancer
1. Circulating Tumor Cell Lab, Division of Medical Oncology, Department of Internal Medicine, Chang Gung Memorial Hospital at Linkou, College of Medicine, Chang Gung University, Taoyuan, Taiwan;
2. Graduate Institute of Clinical Medical Sciences, College of Medicine, Chang Gung University, Taiwan;
3. Graduate Institute of Biochemical and Biomedical Engineering, Chang Gung University, Taoyuan City, Taiwan;
4. Department of Chemical and Materials Engineering, Chang Gung University, Taoyuan, Taiwan;
5. Division of Hematology-Oncology, Department of Internal Medicine, Chang Gung Memorial Hospital, Keelung and Chang Gung University, College of Medicine, Taiwan;
6. Cancer Center, Chang Gung Memorial Hospital, Keelung, Taiwan.
7. Department of Oncology-Pathology, Karolinska Institutet, Stockholm, Sweden; Cancer Center Karolinska, Karolinska University Hospital, Stockholm SE-17176, Sweden.
8. Northern Institute for Cancer Research, School of Medicine, Newcastle University, Newcastle, United Kingdom.
*Drs. Chou, Wu and Chang contributed equally to this work.
Abstract
Background: There is an urgency to develop robust prognostic biomarkers for metastatic colorectal cancer (mCRC) patients receiving chemotherapy. The current study aimed to examine the prognostic significance of circulating tumour cells (CTCs) and to develop a prognostic model incorporating CTCs in predicting the outcomes of mCRC patients treated with chemotherapy.
Methods: Our study prospectively enrolled 55 mCRC patients who had undergone palliative chemotherapy between 2011 and 2014. Baseline CTCs and clinicopathological variables predictive of survival outcome were identified using univariate analysis. Negative selection-based protocol plus flow cytometry was used for CTC identification.
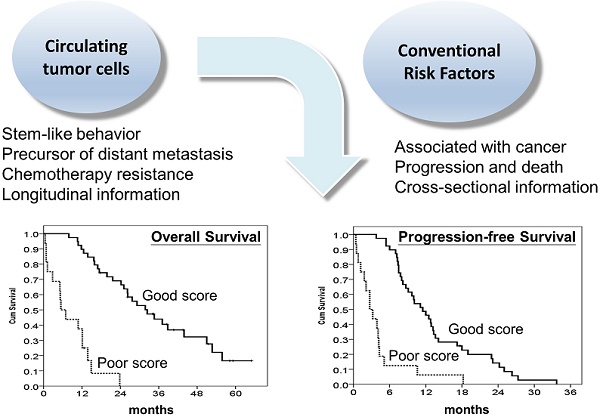
Results: The median overall survival (OS) and progression-free survival (PFS) were 24.2 months and 8.7 months, respectively. CTCs were detected in all the patients, and the median number of CTCs was 30.8/mL (range: 5.8-431.3/mL). The median OS and PFS were 37.1 and 13.3 months, respectively, for patients with CTC number ≤30/mL, while the median OS and PFS were 14.9 months and 5.1 months, respectively, for patients with CTC number >30/mL (both P<0.001). A prognostic model using CTCs in conjunction with other independent clinical variables further stratified patients into good and poor prognostic groups. The median OS and PFS were 32.4 and 11.5 months, respectively, in the good prognostic group and 5.4 and 2.7 months, respectively, in the poor prognostic group.
Conclusions: We developed a reliable CTC-based prognostic model for the prediction of clinical outcomes in mCRC patients treated with chemotherapy. This model may be used to assist clinicians in identifying those with the poorest prognosis before treatment.
Keywords: metastatic colorectal cancer, circulating tumour cell, palliative chemotherapy, prognosis, model

 Global reach, higher impact
Global reach, higher impact