Impact Factor ISSN: 1449-2288
Int J Biol Sci 2019; 15(2):369-385. doi:10.7150/ijbs.28422 This issue Cite
Research Paper
ING4 suppresses hepatocellular carcinoma via a NF-κB/miR-155/FOXO3a signaling axis
1. Center for Systems Biology, Soochow University, Suzhou 215006, China
2. Department of Oncology, the First Affiliated Hospital of Soochow University, Suzhou 215006, China
3. Department of Oncology, Suzhou Science & Technology Town Hospital, Suzhou 215153, China
4. Department of General Surgery, the First Affiliated Hospital of Soochow University, Suzhou 215006, China
*These authors contributed equally to this work.
Abstract
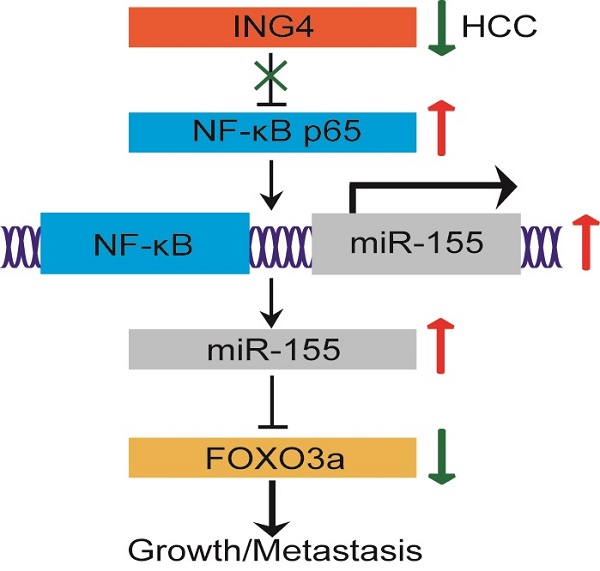
The tumor suppressor ING4 has been shown to be reduced in human HCC. The alteration of ING4 contributes to HCC progression. However, its effect in HCC and the potential mechanism is largely unclear. Herein, we found that downregulation of ING4 in HCC tumor tissues was closely associated with cancer staging, tumor size and vascular invasion. Lentivirus-mediated ING4 overexpression significantly inhibited proliferation, migration and invasion, and induced cell cycle G1 phase arrest and apoptosis in MHCC97H human HCC cells. Moreover, overexpression of ING4 dramatically suppressed MHCC97H tumor cell growth and metastasis to lung in vivo in athymic BALB/c nude mice. Mechanistic studies revealed that overexpression of ING4 markedly increased expression of FOXO3a both at the mRNA and protein level as well as enhanced nuclear level and transcriptional activity of FOXO3a in MHCC97H tumor cells. In addition, ING4 repressed transcriptional activity of NF-κB and expression of miR-155 targeting FOXO3a. Knockdown of ING4 exhibited opposing effects in MHCC97L human HCC cells. Interestingly, knockdown of FOXO3a attenuated not only ING4-elicited tumor suppression but also ING4-mediated regulatory effect on FOXO3a downstream targets, confirming that FOXO3a is involved in ING4-directed tumor-inhibitory effect in HCC. Overexpression of miR-155 attenuated ING4-induced upregulation of FOXO3a, whereas inhibition of miR-155 blunted ING4 knockdown-induced reduction of FOXO3a. Furthermore, inhibition of NF-κB markedly impaired ING4 knockdown-induced upregulation of miR-155 and downregulation of FOXO3a. Taken together, our study provided the first compelling evidence that ING4 can suppress human HCC growth and metastasis to a great extent via a NF-κB/miR-155/FOXO3a pathway.
Keywords: hepatocellular carcinoma (HCC), inhibitor of growth 4 (ING4), forkhead box 3a (FOXO3a), nuclear factor-kappa B (NF-κB), microRNA-155 (miRNA/miR-155)

 Global reach, higher impact
Global reach, higher impact