Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(2):493-506. doi:10.7150/ijbs.29404 This issue Cite
Research Paper
CD9 regulates keratinocyte migration by negatively modulating the sheddase activity of ADAM17
1. Institute of Burn Research, State Key Laboratory of Trauma, Burns and Combined Injury, Southwest Hospital, Third Military Medical University(Army Medical University), Chongqing, China.
2. Department of plastic Surgery, Southwest Hospital, Third Military Medical University(Army Medical University), Chongqing, China.
Abstract
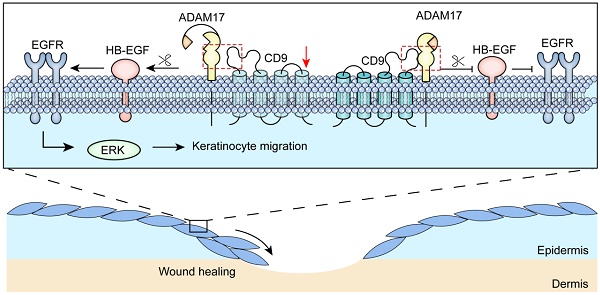
CD9 is a trans-membrane protein, and has recently been implicated in different physiological and cellular processes, such as cell migration and adhesion. According to previous study, down-regulation of CD9 contributes to keratinocyte migration, critical for wound re-epithelialization. Nevertheless, it is widely believed that tetraspanin CD9 does not have ligands or function as the cell surface receptor, rather it is thought to associate with other transmembrane molecules, thereby mediate keratinocyte migration. Little is known about how CD9 associates with transmembrane molecules in migratory keratinocytes. Here, using confocal microscopy, we observed that tetraspanin CD9 and ADAM17 co-localized on the surface of keratinocytes in the course of wound repair in vivo and in vitro. Co-immunoprecipitation experiments demonstrated a direct association between CD9 and ADAM17 in HaCaT cells and C57-MKs. Functional studies revealed that down-regulation or over-expression of CD9 exerted negative regulatory effects on ADAM17 sheddase activity. This activity is involved in CD9-regulated cell motility and migration. Further studies found that ADAM17 inhibitor-TAPI-2 or siADAM17 significantly abolished the enhanced effect of keratinocyte migration induced by CD9 down-regulation. Meanwhile, the sheddase activity of ADAM17 was inhibited by TAPI-2, which decreased this release of AREG and HB-EGF in CD9-silenced HaCat cells and C57-MKs. Importantly, neutralizing antibody against HB-EGF significant weakened keratinocyte migration and motility in CD9-silenced keratinocytes, and the inhibition of CD9-regulated keratinocyte migration by siADAM17 was rescued by addition of recombinant HB-EGF, activating EGFR/ERK pathway. Collectively, our results suggest that ADAM17 sheddase activity is activated by down-regulation of CD9, thereby mediating shedding of HB-EGF and activation of EGFR/ERK signaling, which crucially affects the keratinocyte migration and wound healing.
Keywords: CD9, ADAM17, HB-EGF, EGFR, keratinocyte migration, wound healing

 Global reach, higher impact
Global reach, higher impact