Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(7):1557-1570. doi:10.7150/ijbs.31904 This issue Cite
Research Paper
miR-155-5p Promotes Dorsal Root Ganglion Neuron Axonal Growth in an Inhibitory Microenvironment via the cAMP/PKA Pathway
1. Department of Orthopedics, The 981st Hospital of the Chinese People's Liberation Army, Chengde 067000, Hebei Province, P.R. China
2. Department of Orthopedics, Tianjin Medical University General Hospital, Tianjin 300052, P.R. China
3. Department of Pediatric Internal Medicine, Affiliated Hospital of Chengde Medical University, Chengde 067000, Hebei Province, P.R. China
4. Department of Spine Surgery, Beijing Luhe Hospital, Capital Medical University, Beijing 100000, P.R. China
5. Department of Orthopedics, Chengde Central Hospital, Chengde 067000, Hebei Province, P.R. China
6. Department of Orthopedics, The Second Hospital of Tianjin Medical University, Tianjin 300211, P.R. China
7. Chengde Medical University, Chengde 067000, Hebei Province, P.R. China
8. Leukemia Center, Chinese Academy of Medical Sciences & Peking Union of Medical College, Institute of Hematology & Hospital of Blood Diseases, Tianjin 30020, P.R. China
9. Department of Neurology, The 981st Hospital of the Chinese People's Liberation Army, Chengde 067000, Hebei Province, P.R. China
10. Tianjin Neurological Institute, Key Laboratory of Post-Neuroinjury Neuro-repair and Regeneration in Central Nervous System, Ministry of Education and Tianjin City, Tianjin 300052, P.R. China
* Tianyi Wang, Bo Li, Zhijie Wang, Xin Yuan, Chuanjie Chen contributed equally to this article.
Abstract
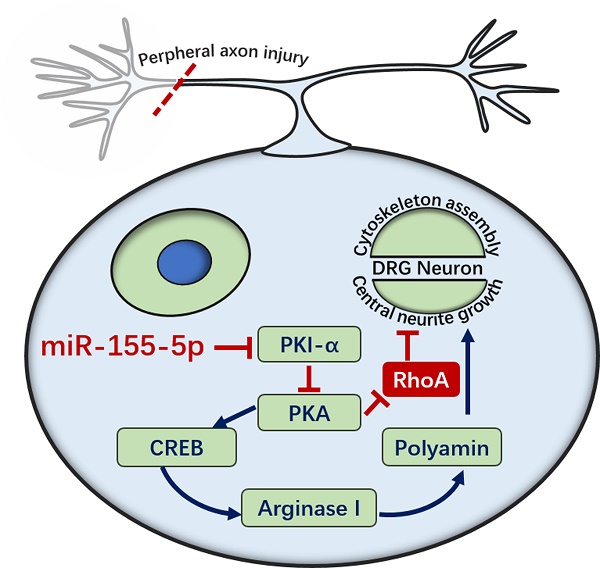
Sensory dysfunction post spinal cord injury causes patients great distress. Sciatic nerve conditioning injury (SNCI) has been shown to restore sensory function after spinal cord dorsal column injury (SDCL); however, the underlying mechanism of this recovery remains unclear. We performed a microarray assay to determine the associated miRNAs that might regulate the process of SNCI promoting SDCL repair. In total, 13 miRNAs were identified according to our inclusion criteria, and RT-qPCR was used to verify the microarray results. Among the 13 miRNAs, the miR-155-5p levels were decreased at 9 h, 3 d, 7 d, 14 d, 28 d, 2 m and 3 m timepoints in the SDCL group, while the SNCI group had a smaller decrease. Thus, miR-155-5p was chosen for further study after a literature review and an analysis with the TargetScan online tool. Specifically, miR-155-5p targets PKI-α, and the expression pattern of PKI-α was opposite that of miR-155-5p in both the SDCL and SNCI groups. Interestingly, miR-155-5p could promote dorsal root ganglion (DRG) neuron axon growth via the cAMP/PKA pathway and in a TNF-α, IL-1β or MAG inhibitory microenvironment in vitro. Furthermore, miR-155-5p could regulate the cAMP/PKA pathway and promote sensory conduction function recovery post dorsal column injury as detected by NF-200 immunohistochemistry, somatosensory-evoked potentials, BBB scale and tape removal test. Collectively, our results demonstrated that miR-155-5p participates in the molecular mechanism by which SNCI promotes the repair of SDCL and that upregulated miR-155-5p can repair SDCL by enhancing DRG neuron axon growth via the cAMP/PKA pathway. These findings suggest a novel treatment target for spinal cord injury.
Keywords: spinal cord dorsal column injury, microRNA, sensory function, cAMP/PKA, miR-155-5p

 Global reach, higher impact
Global reach, higher impact