10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(10):2037-2050. doi:10.7150/ijbs.35245 This issue Cite
Research Paper
Vascular Protection of TPE-CA on Hyperhomocysteinemia-induced Vascular Endothelial Dysfunction through AA Metabolism Modulated CYPs Pathway
1. School of Chinese Materia Medica, Beijing University of Chinese Medicine, Beijing 100029, China
2. Institution of Basic Theory, China Academy of Chinese Medical Sciences, Beijing 100700, China
3. Institute of Basic Research in Clinical Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China
4. School of Chinese Medicine, Hong Kong Baptist University, Kowloon, Hongkong, China
Abstract
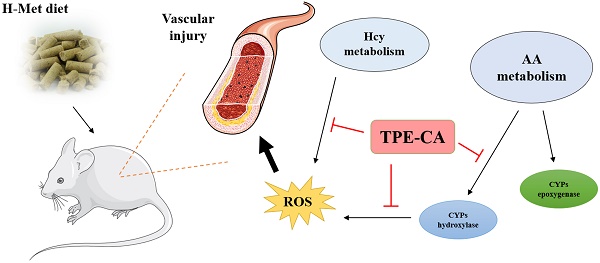
A high concentration of homocysteine (Hcy) in plasma induces vascular endothelial dysfunction, and it may ultimately accelerate the development of cardiovascular diseases (CVDs). Although several B vitamins have been clinically applied for hyperhomocysteinemia (HHcy) treatment, the outcomes are not satisfied due to their limited therapeutic mechanism. Hence, in order to improve the curative effect, development of new effective therapeutic strategies should be put on the agenda. Total phenolic extracts of Citrus aurantium L. (TPE-CA) is a naturally obtained phenolic mixture, mainly containing flavones, flavanones and their glycosyl derivatives, flavonols, polymethoxyflavones and coumarins. Previous reports indicated that bioactive phenolic compounds possessed potent vascular protective effects and regarded as a protective agent against CVDs. Intriguingly, the exact mechanism underlying the suppressed effects of TPE-CA on HHcy could assist in revealing their therapy on CVDs. Here, the multi-targeted synergistic mechanism of TPE-CA on HHcy-induced vascular endothelial dysfunction was uncovered in a deduced manner. TPE-CA treatment exhibited an obvious superiority than that of B vitamins treatment. Network pharmacology was employed to identify the interrelationships among compounds, potential targets and putative pathways. Further experimental validation suggested that the treatment of TPE-CA for HHcy could not only effectively reduce the Hcy level in plasma through up-regulating transsulfuration pathway in Hcy metabolism, but also restore the HHcy-induced vascular endothelial dysfunction by activating cytochrome P450 enzymes (CYPs) epoxygenase signal cascades and inhibiting CYPs hydroxylase signal cascades in arachidonic acid (AA) metabolism.
Keywords: Hyperhomocysteinemia, Vascular endothelial dysfunction, Total phenolic extracts of Citrus aurantium L., CYPs signal pathway, Arachidonic acid metabolism

 Global reach, higher impact
Global reach, higher impact