Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(10):2198-2210. doi:10.7150/ijbs.35092 This issue Cite
Research Paper
P21 and P27 promote tumorigenesis and progression via cell cycle acceleration in seminal vesicles of TRAMP mice
1. School of Life Sciences, Jiangsu Key Laboratory for Molecular and Medical Biotechnology, Nanjing Normal University, Nanjing, 210023, China;
2. ABSL-3 Laboratory at the Center for Animal Experiment and State Key Laboratory of Virology, Wuhan University School of Medicine, Wuhan, 430071, China.
Abstract
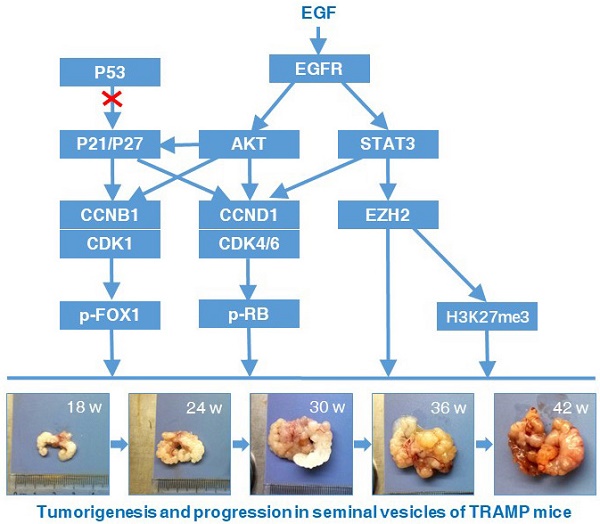
Transgenic adenocarcinoma mouse prostate (TRAMP) model is established to mimic human prostate cancer progression, where seminal vesicle lesions often occur and has been described as phyllodes-like epithelial-stromal tumors. However, the molecular mechanism regulating tumorigenesis and progression in seminal vesicles of TRAMP mice remains largely unknown. In this study, C57BL/6 TRAMP mice were found to have a significantly shorter lifespan than wild-type (WT) mice and all of the seminal vesicles were markedly increased in size and weight with age from 24 weeks exhibiting a clearly papillary-phyllode pattern, though no obvious difference was observed in multiple organs including heart, liver, spleen, lungs, kidneys, testicles and bone between TRAMP and WT mice, and less than 10% of TRAMP mice developed prostate tumors. Western blotting showed Cyclin (CCN) B1 and CCND1 were remarkably overexpressed in seminal vesicle tumors of TRAMP mice at 24 weeks of age and increased with age till the end of trial, which was confirmed by Immunohistochemistry (IHC). P21 and P27 were also significantly augmented, whereas P53 and phosphorylated P53 (p-P53) were constantly expressed in normal controls and P53 did not appear to be mutated. Not only cyclin-dependent kinase (CDK) 1 and phosphorylated forkhead box protein (FOX) O1 but also CDK4, CDK6 and phosphorylated retinoblastoma-associated protein (RB) had similar increase trends, so did epidermal growth factor receptor (EGFR), AKT serine/threonine kinase (AKT), and their respective phosphorylation levels. Signal transducer and activator of transcription (STAT) 3, p-STAT3, enhancer of zeste homolog 2 (EZH2) and EZH2 mediated trimethylation of histone H3 lysine 27 (H3K27me3) were considerably elevated, too. Taken together, this finding suggests P21 and P27 promote carcinogenesis and development in seminal vesicles of TRAMP mice via accelerating cell cycle progression, in which oncogenic transformation of P21 and P27 might be through regulation of EGFR-AKT signaling.
Keywords: tumorigenesis, transgenic adenocarcinoma mouse prostate, epidermal growth factor receptor

 Global reach, higher impact
Global reach, higher impact