Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(12):2548-2560. doi:10.7150/ijbs.34213 This issue Cite
Review
Current Progress in CAR-T Cell Therapy for Solid Tumors
1. Shanghai Baize Medical Laboratory, Shanghai, China
2. Department of Pathology and Laboratory Medicine, Indiana University School of Medicine, Indianapolis, Indiana, USA
3. Shanghai Cell Therapy Research Institute, Shanghai, China
4. Shanghai Engineering Research Center for Cell Therapy, Shanghai, China
* These authors contributed equally to this work.
Abstract
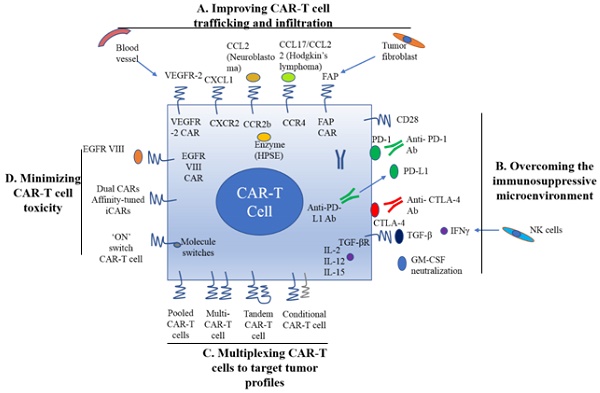
Cancer immunotherapy by chimeric antigen receptor-modified T (CAR-T) cells has shown exhilarative clinical efficacy for hematological malignancies. Recently two CAR-T cell based therapeutics, Kymriah (Tisagenlecleucel) and Yescarta (Axicabtagene ciloleucel) approved by US FDA (US Food and Drug Administration) are now used for treatment of B cell acute lymphoblastic leukemia (B-ALL) and diffuse large B-cell lymphoma (DLBCL) respectively in the US. Despite the progresses made in treating hematological malignancies, challenges still remain for use of CAR-T cell therapy to treat solid tumors. In this landscape, most studies have primarily focused on improving CAR-T cells and overcoming the unfavorable effects of tumor microenvironment on solid tumors. To further understand the current status and trend for developing CAR-T cell based therapies for various solid tumors, this review emphasizes on CAR-T techniques, current obstacles, and strategies for application, as well as necessary companion diagnostics for treatment of solid tumors with CAR-T cells.
Keywords: CAR-T cells, chimeric antigen receptor, solid tumors, companion diagnostics, CTC

 Global reach, higher impact
Global reach, higher impact