Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(12):2654-2663. doi:10.7150/ijbs.39534 This issue Cite
Research Paper
Gene signature predictive of hepatocellular carcinoma patient response to transarterial chemoembolization
1. Laboratory of Human Carcinogenesis;
2. Liver Cancer Program, Center for Cancer Research, National Cancer Institute, Bethesda, Maryland, USA;
3. Departments of Pathology;
4. Surgery, and State Key Laboratory for Liver Research, The University of Hong Kong, Hong Kong, China;
5. Life Sciences Institute, Zhejiang University, Hangzhou, China;
6. Shandong Cancer Hospital and Institute, Jinan, China;
7. Fudan University, Shanghai, China;
8. University Medical Center Mainz, Mainz, Germany;
9. Thoracic and GI Malignancies Branch, Center for Cancer Research, National Cancer Institute, Bethesda, Maryland, USA
*Present address for YP and JC: Chulabhorn Research Institute, Bangkok, Thailand
Abstract
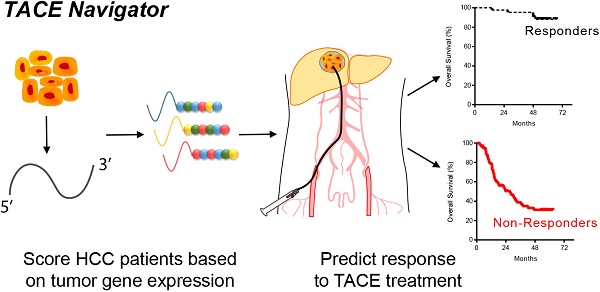
Transarterial chemoembolization (TACE) is a commonly used treatment modality in hepatocellular carcinoma (HCC). The ability to identify patients who will respond to TACE represents an important clinical need, and tumor gene expression patterns may be associated with TACE response. We investigated whether tumor transcriptome is associated with TACE response in patients with HCC. We analyzed transcriptome data of treatment-naïve tumor tissues from a Chinese cohort of 191 HCC patients, including 105 patients who underwent TACE following resection with curative intent. We then developed a gene signature, TACE Navigator, which was associated with improved survival in patients that received either adjuvant or post-relapse TACE. To validate our findings, we applied our signature in a blinded manner to three independent cohorts comprising an additional 130 patients with diverse ethnic backgrounds enrolled in three different hospitals who received either adjuvant TACE or palliative TACE.
TACE Navigator stratified patients into Responders and Non-Responders which was associated with improved survival following TACE in our test cohort (Responders: 67 months vs Non-Responders: 39.5 months, p<0.0001). In addition, multivariable Cox model demonstrates that TACE Navigator was independently associated with survival (HR: 9.31, 95% CI: 3.46-25.0, p<0.001). In our validation cohorts, the association between TACE Navigator and survival remained robust in both Asian patients who received adjuvant TACE (Hong Kong: 60 months vs 25.6 months p=0.007; Shandong: 61.3 months vs 32.1 months, p=0.027) and European patients who received TACE as primary therapy (Mainz: 60 months vs 41.5 months, p=0.041). These results indicate that a TACE-specific molecular classifier is robust in predicting TACE response. This gene signature can be used to identify patients who will have the greatest survival benefit after TACE treatment and enable personalized treatment modalities for patients with HCC.
Keywords: Transarterial Chemoembolization, hepatocellular carcinoma, precision oncology, gene signature, treatment response, hypoxia signaling

 Global reach, higher impact
Global reach, higher impact