Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(13):2774-2782. doi:10.7150/ijbs.34888 This issue Cite
Research Paper
Obstructive sleep apnea syndrome promotes the progression of aortic dissection via a ROS- HIF-1α-MMPs associated pathway
1. Division of Cardiology, Department of Internal Medicine, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430030, PR China
2. Hubei Key Laboratory of Genetics and Molecular Mechanisms of Cardiological Disorders, Wuhan, 430030, PR China
3. Department of Cardiology, Affiliated Hospital of Weifang Medical University, Weifang, Shandong, 261000, PR China
4. Department of Forensic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430030, PR China
* W.L. and W.Z. contribute equally to this manuscript.
Abstract
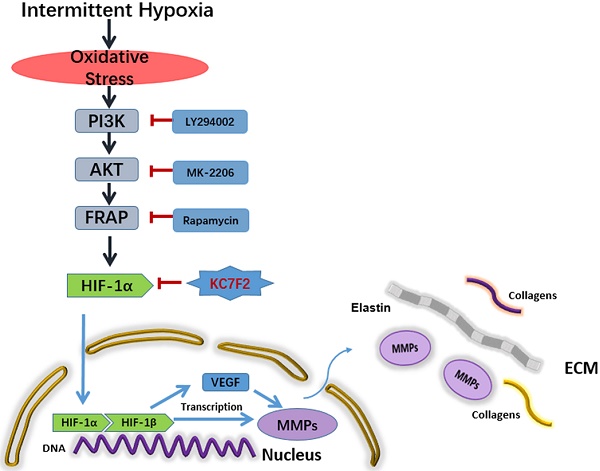
Aims: Obstructive sleep apnea syndrome (OSAS) has been increasingly recognized as an independent risk factor for aortic dissection (AD) and it is strongly associated with the extent of intermittent hypoxia and re-oxygenation (IH). This study aimed to clarify role of ROS- HIF-1α-MMPs pathway in the pathogenesis of AD and whether the HIF-1α inhibitor attenuates AD formation.
Methods and results: 8-week-old male ApoE-/- mice were given β-aminopropionitrile at a concentration of 0.1 % for 3 weeks and infused via osmotic mini pumps with either saline or 2,500 ng/min/kg angiotensin II (Ang II) for 2 weeks. To mimic the OSAS, one group was exposed to IH, which consisted of alternating cycles of 20.9% O2/8% O2 FiO2 (30 episodes per hour) with 20 s at the nadir FiO2 during the 12-h light phase, 2 weeks before Ang II infusion. After Ang II infusion, we assessed remodeling in the aorta by echocardiography, histological and immunohistochemical analysis. IH treatment resulted in significant enlargement of the luminal area, destruction of the media, marked thickening of the adventitia, higher incidence of AD formation and lower survival rate in compared with the Ang II only group. Moreover, IH exposure markedly increased the aortic ROS production and subsequent HIF-1α expression, which in turn promoted the expressions of VEGF, MMP2 and MMP9 and finally leading to the progression of AD. Besides, in vitro study confirmed that IH induced HIF-1α expression plays an important role in the induction of MMPs and that is regulated by the PI3K/AKT/FRAP pathway. Intriguingly, a selective HIF-1α inhibitor KC7F2 could significantly ameliorate IH exposure induced aforementioned deleterious effects in vitro and in vivo.
Conclusion: OSAS induced IH can promote the occurrence and progression of AD via a ROS- HIF-1α-MMPs associated pathway. The selective HIF-1α inhibitor KC7F2 could be a novel therapeutic agent for AD patient with OSAS.
Keywords: Aortic dissection, OSAS, HIF-1α, KC7F2, ROS, MMP2/9

 Global reach, higher impact
Global reach, higher impact