Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(13):2815-2829. doi:10.7150/ijbs.31781 This issue Cite
Research Paper
Spexin alleviates insulin resistance and inhibits hepatic gluconeogenesis via the FoxO1/PGC-1α pathway in high-fat-diet-induced rats and insulin resistant cells
1. Department of Endocrinology and Metabolism, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China.
2. Shanghai Intertek Medical diagnostic Testing Center Co; Ltd, Shanghai 200436, China.
3. School of Pharmaceutical Engineering& Life Science, Changzhou University, Changzhou, 213164 China.
* These authors contributed equally to this study.
Abstract
Objective: Recent studies demonstrate circulating serum spexin levels are reduced in obesity or type 2 diabetes mellitus (T2DM) patients and may play a role in glucose metabolism. The mechanism underlying is not known. In this study, we explore whether spexin has a role in insulin resistance and hepatic glucose metabolism.
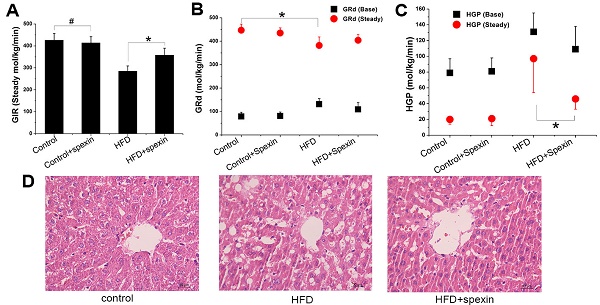
Methods: The correlation between serum spexin levels and the homeostasis model assessment of insulin resistance (HOMA-IR) was studied in newly diagnosed T2DM patients. After intraperitoneal injection of exogenous spexin for 8 weeks, the effect of spexin on exogenous glucose infusion rates (GIR), and hepatic glucose production (HGP) were assessed by extended hyperinsulinemic-euglycemic clamp in high-fat-diet (HFD)-induced rats. Glucose concentration with CRISPR/Cas9-mediated disruption of spexin expression in HepG2 cells culture was observed. Expression of transcription factors (Forkhead box O1, FoxO1 and peroxisome proliferator-activated receptor gamma coactivator 1-alpha, PGC-1α) and key enzymes (G-6-Pase and PEPCK) of gluconeogenesis pathway were observed in vitro and in vivo.
Results: The serum spexin level was significantly low in newly diagnosed T2DM patients as compared with healthy patients and significantly negatively correlated with the HOMA-IR values. Exogenous spexin treatment resulted in weight loss and decrease of HOMA-IR value in high-fat-diet (HFD)-induced rats. The exogenous glucose infusion rates (GIR) were higher in the HFD + spexin group than that in the HFD group (358 ± 32 vs. 285 ± 24 μmol/kg/min, P < 0.05). Steady-state hepatic glucose production (HGP) was also suppressed by ~50% in the HFD + spexin group as compared with that in the HFD group. Furthermore, spexin inhibited gluconeogenesis in dose-dependent and time-dependent manner in the insulin-resistant cell model. CRISPR/Cas9-mediated knockdown of spexin in HepG2 cells activated gluconeogenesis. Moreover, spexin was shown regulating gluconeogenesis by inhibiting FoxO1/PGC-1α pathway, and key gluconeogenic enzymes, (PEPCK and G-6-Pase) in both HFD-induced rats and insulin-resistant cells.
Conclusions: Spexin plays an important role in insulin resistance in HFD-induced rats and insulin-resistant cells. Regulation of the effects of spexin on insulin resistance may hold therapeutic value for metabolic diseases.
Keywords: spexin, insulin resistance, hepatic gluconeogenesis, FoxO1

 Global reach, higher impact
Global reach, higher impact