Impact Factor ISSN: 1449-2288
Int J Biol Sci 2019; 15(13):2859-2871. doi:10.7150/ijbs.39063 This issue Cite
Research Paper
Macrophage Colony-stimulating Factor Mediates the Recruitment of Macrophages in Triple negative Breast Cancer
1. Breast surgery, the second Hospital of Shanxi Medical University, Taiyuan 030001, China.982602719@qq.com.
2. The second Clinical Medical College, Shanxi Medical University, Taiyuan 030001, China.1456236003@qq.com.
3. Institute of Biomedical Sciences, Shanxi University, Taiyuan 030006, China. zlc@sxu.edu.cn
4. Department of Pathology, Shanxi Cancer Hospital, Taiyuan 030001, China. xiyanfeng1998@163.com.
5. Breast surgery, the second Hospital of Shanxi Medical University, Taiyuan 030001, China. 17747001066@189.cn. 13099096632.
6. Department of Digestive Sciences, Shanxi Cancer Hospital, Taiyuan 030001, China. zhhh31@163.com. 18035119990.
7. Institute of Biotechnology, Key Laboratory of Chemical Biology and Molecular Engineering of National Ministry of Education, Shanxi University, Taiyuan, 030006, China. lzy@sxu.edu.cn. 13934565188.
8. School of Life Science, Shanxi University, Taiyuan, 030006, China. lzy@sxu.edu.cn. 13934565188.
* Equal contributors.
Abstract
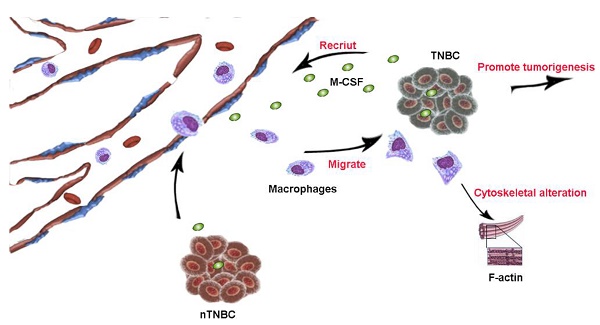
Triple negative breast cancer (TNBC) is characterized by aggressive malignant tumor, poor prognosis and lack of targeted treatment. Several studies have established that macrophages are closely associated with the progression of TNBC. Through immunohistochemical analysis, we found that the infiltration of macrophage in TNBC tissue was more than that in non-triple negative breast cancer (nTNBC) tissue. Furthermore, the conditioned medium (CM) of MDA-MB-231 and HCC1937, the TNBC cell lines, had significant migration-promoted effect on macrophages. However, the macrophages migration-promoted ability of nTNBC cell line MCF-7 was weaker than that of MDA-MB-231 and HCC1937 cells. Mechanistically, MDA-MB-231 and HCC1937 cells secreted more macrophage colony-stimulating factor (M-CSF) than MCF-7, which is the main inducer of macrophage migration, and the secreted M-CSF promoted the increase in actin and the elongation of pseudopodia. When M-CSF was neutralized by antibody, the elongation of macrophage pseudopodia was disappeared and the migration was inhibited. In vivo, there were more macrophages in tumors induced by MDA-MB-231 than MCF-7 did. Therefore, M-CSF specially secreted by TNBC was the important cause of macrophages aggregation in TNBC, which further promoted the aggressiveness of TNBC.
Keywords: triple negative breast cancer, macrophage migration, macrophage colony-stimulating factor, cytoskeleton, filamentous actin

 Global reach, higher impact
Global reach, higher impact