Impact Factor ISSN: 1449-2288
Int J Biol Sci 2020; 16(8):1450-1462. doi:10.7150/ijbs.44414 This issue Cite
Research Paper
Stagewise keratinocyte differentiation from human embryonic stem cells by defined signal transduction modulators
1. Centre of Reproduction, Development and Aging, Faculty of Health Sciences, University of Macau, Taipa, Macau
2. Cancer Centre, Faculty of Health Sciences, University of Macau, Taipa, Macau
3. Department of Urology Surgery, Zhuhai People's Hospital, Jinan University, Zhuhai, Guangdong 519000, China
4. Zhuhai Precision Medical Center, Zhuhai People's Hospital, Jinan University, Zhuhai, Guangdong 519000, China
5. Center of Interventional radiology, Zhuhai People's Hospital, Jinan University, Zhuhai, Guangdong 519000, China
6. State Key Laboratory of Quality Research in Chinese Medicine, Institute of Chinese Medical Sciences, University of Macau, Taipa, Macau
7. Bioimaging and Stem Cell Core Facility, Faculty of Health Sciences, University of Macau, Taipa, Macau
Abstract
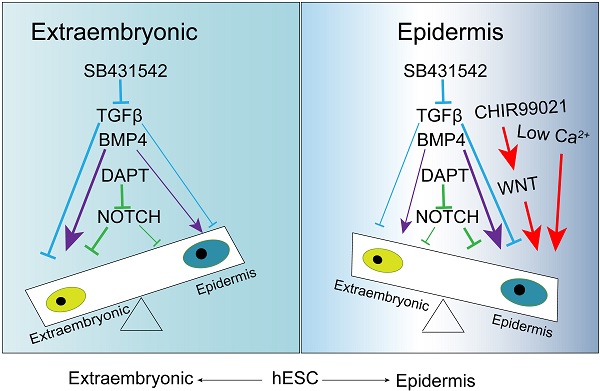
Keratinocyte is the predominant cell type in the epidermis of skin, and it provides the protective barrier function for the body. Various signaling pathways have been implicated in keratinocyte differentiation in animal models; However, their temporal regulation and interactions are still to be explored in pluripotent stem cell models. In this report, we use human embryonic stem cells to demonstrate that epidermal ectoderm and subsequent keratinocyte cell fate can be determined step by step under the regulation of defined factors. The inhibition of TGFβ initiates ectodermal lineage differentiation, and the activation of BMP pathway drives epidermal TP63 expression. Meanwhile, the timely activation of WNT pathway suppresses extraembryonic lineage, and promotes epidermal cell fate. With further specification by NOTCH inhibition, more than 90% of cells become TP63-positive stage Ⅱ keratinocytes. Finally, stage Ⅲ keratinocytes are produced under defined hypo-calcium keratinocyte culture conditions, and are further matured in mouse xenograft model. This study not only establishes an in vitro platform to study keratinocyte cell fate determination, but also provides an efficient protocol to produce keratinocytes for disease models and clinical applications.
Keywords: human embryonic stem cells, keratinocyte differentiation, defined factors

 Global reach, higher impact
Global reach, higher impact