Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2020; 16(9):1616-1628. doi:10.7150/ijbs.40896 This issue Cite
Research Paper
Noninvasive monitoring of liver metastasis development via combined multispectral photoacoustic imaging and fluorescence diffuse optical tomography
1. OPTIMAL, Small animal Imaging Platform, F-38000 Grenoble, France.
2. INSERM U1209, CNRS UMR5309, Univ. Grenoble Alpes, Institute for Advanced Biosciences, F-38000 Grenoble, France.
3. Fluoptics, Grenoble, France.
4. CNRS UMR5525 ; TIMC-IMAG, Univ. Grenoble Alpes, F-38000 Grenoble, France.
Abstract
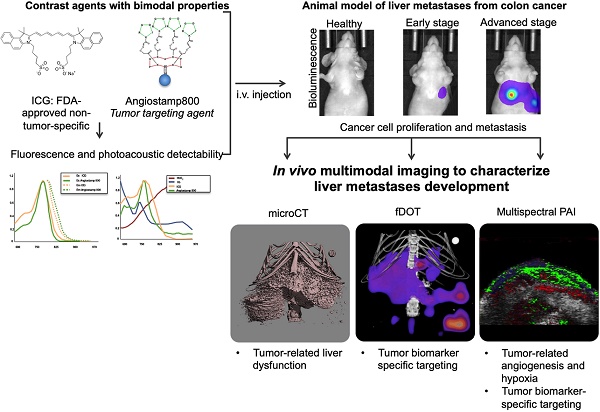
Rationale: In vivo molecular imaging in preclinical animal models is a tool of choice for understanding the pathophysiological mechanisms involved in cancer development and for conducting drug development research. Moreover, combining several imaging modalities can provide multifaceted, complementary and cross-validated information. Photoacoustic imaging (PAI) is a promising imaging modality that can reflect blood vasculature and tissue oxygenation as well as detect exogenous molecules, but one shortcoming of PAI is a lack of organic photoacoustic contrast agents capable of providing tumor contrast.
Methods: In the present study, we designed an animal model of liver metastases from colon cancer and monitored metastasis development by in vivo bioluminescence and X-ray microcomputed tomography. Contrast-agent-free PAI was used to detect the respective amounts of oxy- and deoxyhemoglobin and, thus, liver tissue oxygenation. two contrast agents, Angiostamp800 and indocyanin green (ICG), respectively with and without tumor targeting specificity, were then evaluated for their dual fluorescence and photoacoustic detectability and were then used for combined PAI and fluorescence diffuse optical tomography (fDOT) at various disease development stages.
Findings: Contrast-agent-free PAI reflected tumor angiogenesis and gradual hypoxia during metastasis development. Multispectral PAI enabled noninvasive real-time monitoring of ICG blood pharmacokinetics, which demonstrated tumor-related liver dysfunction. Both PAI and fluorescence ICG signals were clearly modified in metastasis-bearing livers but did not allow for differentiation between different disease stages. In contrast, there was a significant improvement achieved by using the tumor-specific marker Angiostamp800, which provided gradually increasing PAI and fDOT signals during metastasis development.
Conclusion: We demonstrated for the first time the value of using Angiostamp800 as a bimodal tumor-targeting contrast agent for combined PAI and fluorescence imaging of liver metastasis progression in vivo.
Keywords: photoacoustic imaging, liver metastases

 Global reach, higher impact
Global reach, higher impact