Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2020; 16(11):1888-1900. doi:10.7150/ijbs.40917 This issue Cite
Research Paper
Steroid-induced osteonecrosis of the femoral head reveals enhanced reactive oxygen species and hyperactive osteoclasts
1. School of Biomedical Sciences, The University of Western Australia, Perth, WA 6009, Australia
2. Department of Joint Orthopaedics, The First Affiliated Hospital, Guangzhou University of Chinese Medicine, Guangzhou, Guangdong 510405, China
3. The Lab of Orthopaedics of Chinese Medicine, Lingnan Medical Research Center, Guangzhou University of Chinese Medicine, Guangzhou, Guangdong 510405, China
4. Research Institute of Orthopaedics of Chinese Medicine, Guangzhou University of Chinese Medicine, Guangzhou, Guangdong 510378, China
*Kai Chen and Yuhao Liu contributed equally to this work.
Abstract
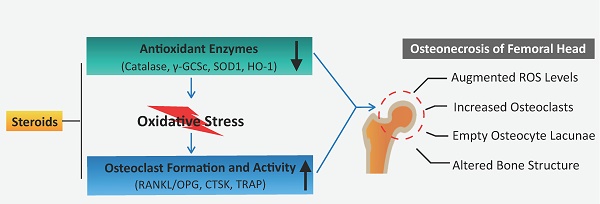
Steroid-induced osteonecrosis of the femoral head (ONFH) is a progressive bone disorder which typically results in femoral head collapse and hip joint dysfunction. It is well-accepted that abnormal osteoclast activity contributes to loss of bone structural integrity and subchondral fracture in ONFH. However, the pathophysiologic mechanisms underlying the recruitment and hyperactivation of osteoclasts in ONFH remain incompletely understood. We assessed the changes of reactive oxygen species (ROS) level and subsequent osteoclast alterations in steroid-induced osteonecrotic femoral heads from both patients and rat ONFH models. When compared with healthy neighboring bone, the necrotic region of human femoral head was characterized by robust up-regulated expression of osteoclast-related proteins [cathepsin K and tartrate-resistant acid phosphatase(TRAP)] but pronounced down-regulation of antioxidant enzymes (catalase, γ-glutamylcysteine synthetase [γ-GCSc], and superoxide dismutase 1 [SOD1]). In addition, the ratio of TNFSF11 (encoding RANKL)/TNFRSF11B (encoding OPG) was increased within the necrotic bone. Consistently, in rat ONFH models induced by methylprednisolone (MPSL) and imiquimod (IMI), significant bone loss in the femoral head was observed, attributable to increased numbers of TRAP positive osteoclasts. Furthermore, the decreased expression of antioxidant enzymes observed by immunoblotting was accompanied by increased ex-vivo ROS fluorescence signals of dihydroethidium (DHE) in rat ONFH models. Therefore, this study lends support to the rationale that antioxidant agents may be a promising therapeutic avenue to prevent or mitigate the progression of steroid-induced ONFH by inhibiting ROS level and hyperactive osteoclasts.
Keywords: Reactive oxygen species (ROS), Osteonecrosis of femoral head (ONFH), Osteoclasts, Antioxidant enzymes

 Global reach, higher impact
Global reach, higher impact