Impact Factor ISSN: 1449-2288
Int J Biol Sci 2020; 16(11):1972-1988. doi:10.7150/ijbs.39259 This issue Cite
Research Paper
Protective role of silibinin against myocardial ischemia/reperfusion injury-induced cardiac dysfunction
1. Department of Cardiology, The First Affiliated Hospital of Wenzhou Medical University, 325000, Nanbaixiang, Wenzhou, Zhejiang, China.
2. Department of Respiratory, The Second Affiliated Hospital and Yuying Children's Hospital of Wenzhou Medical University, 325000, Wenzhou, Zhejiang, China.
3. Department of Cardiology, Affiliated Xinhua Hospital, Shanghai Jiaotong University (SJTU) School of Medicine, Shanghai, China.
*The authors contributed equally to this article.
Abstract
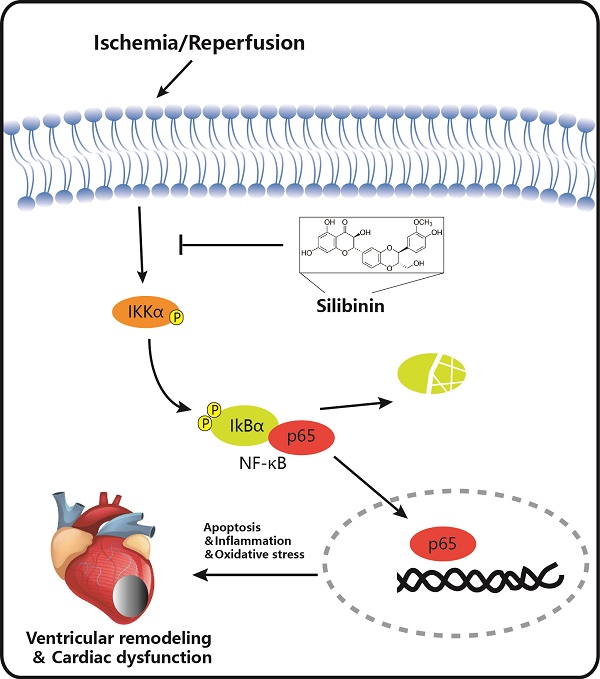
Silibinin is a traditional medicine and utilized for liver protection with antioxidant, anti-inflammation and anti-apoptosis properties. However, its role in myocardial I/R injury and the mechanism involved is currently unknown. In the present study, Silibinin treatment improves cardiac function and limits infarct size, and subsequently inhibits fibrotic remodeling in mice with myocardial I/R injury. Mechanistically, silibinin reduces cardiomyocytes apoptosis, attenuates mitochondrial impairment and endoplasmic reticulum (ER) stress, alleviates ROS generation, neutrophil infiltration and cytokines release. Consistently, silibinin prevents H9C2 cells from hypoxia/reperfusion-induced cell death, oxidative stress and inflammation in vitro. Furthermore, H9C2 cells treated with silibinin blocks NF-κB signaling activation by inhibiting IKKα phosphorylation, IκBα degradation and p65 NF-κB nuclear translocation during hypoxia/ reperfusion. In addition, silibinin plus BAY 11-7082 (a selected NF-κB inhibitor) do not provide incremental benefits in improving myocytes apoptosis, oxidative stress and inflammation in comparison with NF-κB signaling inhibition only. Thus, silibinin-mediated cardioprotection in myocardial I/R injury is associated with decreased apoptosis, oxidative stress and inflammatory response through deactivation of NF-κB pathway.
Keywords: Myocardial I/R injury, Silibinin, Apoptosis, Oxidative stress, Inflammation, NF-κB

 Global reach, higher impact
Global reach, higher impact