Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2020; 16(11):2001-2013. doi:10.7150/ijbs.43825 This issue Cite
Research Paper
Cilostazol alleviate nicotine induced cardiomyocytes hypertrophy through modulation of autophagy by CTSB/ROS/p38MAPK/JNK feedback loop
1. Jinan Central Hospital Affiliated to Shandong University, Jinan, China.
2. Central Hospital Affiliated to Shandong First Medical University, Jinan, China.
3. Affiliated Hospital of Shandong University of Traditional Chinese Medicine, Jinan, China.
4. School of Biological Science and Technology, University of Jinan, China.
5. Development Biology, School of Life Science, Shandong University, Jinan, China.
Abstract
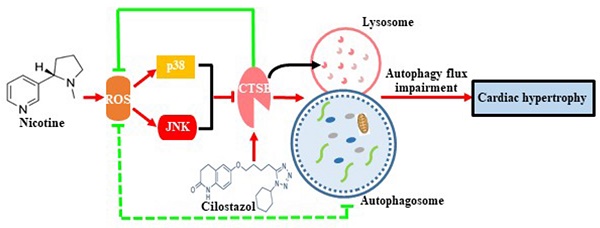
Nicotine is proved to be an important factor for cardiac hypertrophy. Autophagy is important cell recycling system involved in the regulation of cardiac hypertrophy. Cilostazol, which is often used in the management of peripheral vascular disease. However, the effects of cilostazol on nicotine induced autophagy and cardiac hypertrophy are unclear. Here, we aim to determine the role and molecular mechanism of cilostazol in alleviating nicotine-induced cardiomyocytes hypertrophy through modulating autophagy and the underlying mechanisms. Our results clarified that nicotine stimulation caused cardiomyocytes hypertrophy and autophagy flux impairment significantly in neonatal rat ventricular myocytes (NRVMs), which were evidenced by augments of LC3-II and p62 levels, and impaired autophagosomes clearance. Interestingly, cathepsin B (CTSB) activity decreased dramatically after stimulation with nicotine in NRVMs, which was crucial for substrate degradation in the late stage of autophagy process, and cilostazol could reverse this effect dramatically. Intracellular ROS levels were increased significantly after nicotine exposure. Meanwhile, p38MAPK and JNK were activated after nicotine treatment. By using ROS scavenger N-acetyl-cysteine (NAC) could reverse the effects of nicotine by down-regulation the phosphorylation of p38MAPK and JNK pathways, and pretreatment of specific inhibitors of p38MAPK and JNK could restore the autophagy impairment and cardiomyocytes hypertrophy induced by nicotine. Moreover, CTSB activity of lysosome regained after the treatment with cilostazol. Cilostazol also inhibited the ROS accumulation and the activation of p38MAPK and JNK, which providing novel connection between lysosome CTSB and ROS/p38MAPK/JNK related oxidative stress pathway. This is the first demonstration that cilostazol could alleviate nicotine induced cardiomyocytes hypertrophy through restoration of autophagy flux by activation of CTSB and inhibiting ROS/p38/JNK pathway, exhibiting a feedback loop on regulation of autophagy and cardiomyocytes hypertrophy.
Keywords: nicotine, cardiomyocytes hypertrophy, autophagy, CTSB, ROS, p38MAPK/JNK.

 Global reach, higher impact
Global reach, higher impact