Impact Factor ISSN: 1449-2288
Int J Biol Sci 2020; 16(13):2296-2305. doi:10.7150/ijbs.45457 This issue Cite
Research Paper
Long noncoding RNA LINC01123 promotes the proliferation and invasion of hepatocellular carcinoma cells by modulating the miR-34a-5p/TUFT1 axis
1. The Second Clinical Medical College, Zhejiang Chinese Medical University, Hangzhou, Zhejiang 310053, China.
2. Key Laboratory of Tumor Molecular Diagnosis and Individualized Medicine of Zhejiang Province, Zhejiang Provincial People's Hospital (People's Hospital of Hangzhou Medical College), Hangzhou, Zhejiang 310014, China
3. The Medical College of Qindao University, Qindao, Shandong, 266071, China
4. Graduate Department, BengBu Medical College, BengBu, Anhui 233030, China
5. Department of Hepatobiliary Surgery, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an Shaanxi 710061, China
*Contributed equally
Abstract
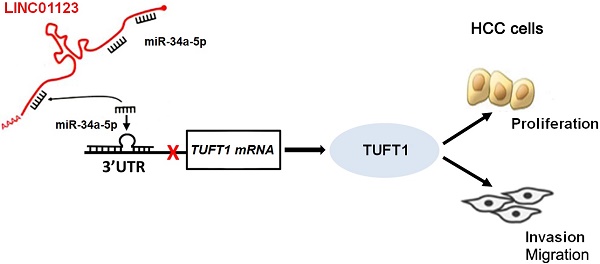
Hepatocellular carcinoma (HCC), one of the main causes of cancer-related deaths globally, is characterized by rapid growth and high invasiveness. Accumulating evidence demonstrates that long noncoding RNAs (lncRNAs) play critical roles in the growth and metastasis of HCC. Recently, lncRNA LINC01123 has been found to contribute to cell proliferation and aerobic glycolysis in lung cancer. However, the function of LINC01123 in HCC, as well as the underlying mechanism of its action, remain unclear. Here, we found that the expression of LINC01123 was clearly upregulated in HCC tissues compared to nontumor tissues. Furthermore, expression of LINC01123 in HCC cells was significantly higher than in LO2 cells. Importantly, the upregulated level of LINC01123 was related to unfavorable clinical features and poor prognosis of HCC. Next, we demonstrated that LINC01123 knockdown suppressed the proliferation, migration and invasion of HCC cells in vitro. Depletion of LINC01123 inhibited HCC xenograft growth in vivo. Conversely, ectopic expression of LINC01123 facilitated HCC cell proliferation and invasion. Mechanistically, LINC01123 acted as a molecular sponge for miR-34a-5p in HCC cells. Tuftelin1 (TUFT1) was identified as the target gene of miR-34a-5p. LINC01123 positively regulated TUFT1 level by targeting of miR-34a-5p in HCC cells. Notably, TUFT1 restoration can abolish miR-34a-5p-induced inhibitory effects on HCC cell proliferation, migration and invasion. In conclusion, LINC01123 was overexpressed in HCC and accelerated cancer cell proliferation and invasion by regulating the miR-34a-5p/TUFT1 axis.
Keywords: hepatocellular carcinoma, LINC01123, miR-34a-5p, TUFT1, tumor progression

 Global reach, higher impact
Global reach, higher impact