Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2020; 16(15):2775-2787. doi:10.7150/ijbs.45619 This issue Cite
Research Paper
FoxO3a suppresses neuropeptide W expression in neuronal cells and in rat hypothalamus and its implication in hypothalamic-pituitary-adrenal (HPA) axis
1. Center of Reproduction, Development & Aging and Institute of Translation Medicine, Faculty of Health Sciences, University of Macau, Macau, China
2. School of Medical Science, Jinan University, Guangzhou, China
3. National Pharmaceutical Engineering Center for Solid Preparation in Chinese Herbal Medicine, Jiangxi University of Traditional Chinese Medicine, Nanchang 330006, P. R. China
4. Jiangsu Key Laboratory of Neuropsychiatric Diseases and College of Pharmaceutical Sciences, Soochow University, Suzhou, Jiangsu 215123, China
5. School of Pharmacy Institute for Drug Research, Faculty of Medicine, The Hebrew University of Jerusalem, Jerusalem 91120, Israel
#These authors contributed equally to this study.
Abstract
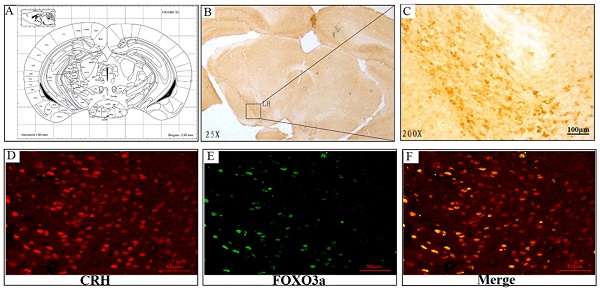
FoxO3a, a forkhead family member of transcription factors, is involved in the regulation of cell metabolism, proliferation, differentiation and apoptosis. However, whether FoxO3a participates in the regulation of glucocorticoids induced-hypothalamic-pituitary-adrenal (HPA) dysfunction is still unknown. Our present results indicate that dexamethasone(DEX) increased FoxO3a expression in PC12 and hypothalamic neuronal cultures in correlation to reduced expression of NPW, a process that could be blocked by GR2 antagonist. DEX restrained the phosphorylation of Akt and FoxO3a, but not ERK1/2 phosphorylation, resulting with FoxO3a nuclear localization. Overexpression of FoxO3a inhibited NPW expression, while FoxO3a knockdown by siRNA had the opposite effect. The regulatory region of NPW promoter contains multiple FoxO3a binding sites, and FoxO3a bonding to these sites inhibited its transcriptional activity. In a rat model, chronic administration of corticosterone reduced animals' body weight and sucrose consumption and caused stress- depression like behavior. Corticosterone treatment induced a marked increase in FoxO3a levels, while decreased the expression of NPW protein in the hypothalamus. Immunofluorescent double labeling demonstrated that FoxO3a and NPW were collocated in the hypothalamus. Taken together, these data indicate that NPW is a new direct downstream target gene of FoxO3a. FoxO3a suppressed the transcription of NPW and modulated glucocorticoids-induced HPA dysfunction by directly regulating the expression of NPW. Thus, present findings suggest that FoxO3a and NPW may be potential therapeutic targets for endocrine and psychiatric disorders.
Keywords: Glucocorticoids, Neuropeptide W (NPW), FoxO3a, HPA axis dysfunction, Hypothalamus

 Global reach, higher impact
Global reach, higher impact