Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2020; 16(15):2853-2867. doi:10.7150/ijbs.44365 This issue Cite
Research Paper
miR-550-1 functions as a tumor suppressor in acute myeloid leukemia via the hippo signaling pathway
1. Department of Hematology, the First Affiliated Hospital, College of Medicine, Zhejiang University, 79 Qingchun Road, Hangzhou, 310003, P.R. China.
2. Department of Cancer Biology, University of Cincinnati, Cincinnati, OH 45219, USA.
3. Section of Hematology/Oncology, Department of Medicine, University of Chicago, Chicago, Illinois 60637, USA.
4. Department of Hematology, Affiliated Hangzhou First People's Hospital, Zhejiang University School of Medicine, 216 Huansha Road, Hangzhou, 310006, P.R. China.
5. Department of Systems Biology & the Gehr Family Center for Leukemia Research, Beckman Research Institute of City of Hope, Monrovia, CA 91016, USA.
*These authors contributed equally to this work.
Abstract
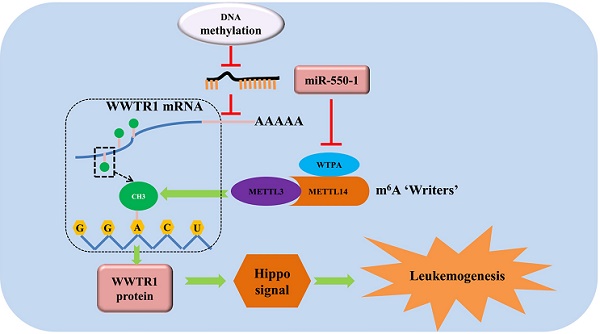
MicroRNAs (miRNAs) and N6-methyladenosine (m6A) are known to serve as key regulators of acute myeloid leukemia (AML). Our previous microarray analysis indicated miR-550-1 was significantly downregulated in AML. The specific biological roles of miR-550-1 and its indirect interactions and regulation of m6A in AML, however, remain poorly understood. At the present study, we found that miR-550-1 was significantly down-regulated in primary AML samples from human patients, likely owing to hypermethylation of the associated CpG islands. When miR-550-1 expression was induced, it impaired AML cell proliferation both in vitro and in vivo, thus suppressing tumor development. When ectopically expressed, miR-550-1 drove the G0/1 cell cycle phase arrest, differentiation, and apoptotic death of affected cells. We confirmed mechanistically that WW-domain containing transcription regulator-1 (WWTR1) gene was a downstream target of miR-550-1. Moreover, we also identified Wilms tumor 1-associated protein (WTAP), a vital component of the m6A methyltransferase complex, as a target of miR-550-1. These data indicated that miR-550-1 might mediate a decrease in m6A levels via targeting WTAP, which led to a further reduction in WWTR1 stability. Using gain- and loss-of-function approaches, we were able to determine that miR-550-1 disrupted the proliferation and tumorigenesis of AML cells at least in part via the direct targeting of WWTR1. Taken together, our results provide direct evidence that miR-550-1 acts as a tumor suppressor in the context of AML pathogenesis, suggesting that efforts to bolster miR-550-1 expression in AML patients may thus be a viable clinical strategy to improve patient outcomes.
Keywords: acute myeloid leukemia, miR-550-1, WWTR1, apoptosis, proliferation

 Global reach, higher impact
Global reach, higher impact