Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(1):50-61. doi:10.7150/ijbs.50041 This issue Cite
Research Paper
Eomesodermin in CD4+T cells is essential for Ginkgolide K ameliorating disease progression in experimental autoimmune encephalomyelitis
1. Department of Neurology, Huashan Hospital, Fudan University.
2. Department of Neurology and Research Center of Neurology, Second Affiliated Hospital, Key Laboratory of Medical Neurobiology of Zhejiang Province, Zhejiang University School of Medicine.
3. Department of Neurology, Fujian Medical University Union Hospital.
4. Department of Neurology and Institute of Neurology, First Affiliated Hospital, Fujian Medical University.
5. Key Laboratory of New-tech for Chinese Medicine Pharmaceutical Process, Lianyungang, China.
* These authors contributed equally.
Abstract
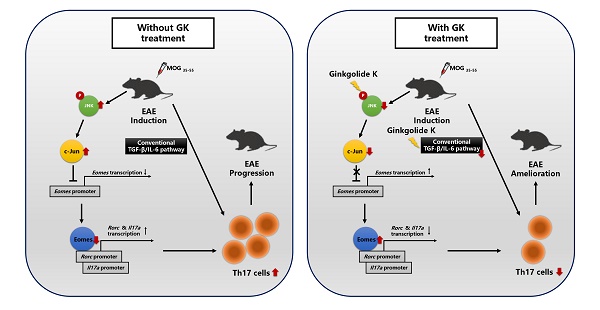
Eomesodermin (Eomes), a transcription factor, could suppress the Th17 cell differentiation and proliferation through directly binding to the promoter zone of the Rorc and Il17a gene, meanwhile the expression of Eomes is suppressed when c-Jun directly binds to its promoter zone. Ginkgolide K (1,10-dihydroxy-3,14-didehydroginkgolide, GK) is a diterpene lactone isolated from the leaves of Ginkgo biloba. A previous study indicated that GK could decrease the level of phospho JNK (c-Jun N-terminal kinase). Here, we reported the therapeutic potential of Ginkgolide K (GK) treatment to ameliorate experimental autoimmune encephalomyelitis (EAE) disease progression.
Methods: EAE was induced in both wildtype and CD4-Eomes conditional knockout mice. GK was injected intraperitoneally. Disease severity, inflammation, and tissue damage were assessed by clinical evaluation, flow cytometry of mononuclear cells (MNCs), and histopathological evaluation. Dual-luciferase reporter assays were performed to measure Eomes transcription activity in vitro. The potency of GK (IC50) was determined using JNK1 Kinase Enzyme System.
Results: We revealed that GK could ameliorate EAE disease progression by the inhibition of the Th17 cells. Further mechanism studies demonstrated that the level of phospho JNK was decreased and the level of Eomes in CD4+T cells was dramatically increased. This therapeutic effect of GK was almost completely interrupted in CD4-Eomes conditional knockout mice.
Conclusions: These results provided the therapeutic potential of GK treatment in EAE, and further suggested that Eomes expression in CD4+T cells might be essential in this process.
Keywords: Multiple sclerosis· Transcription factors· EAE· Therapeutic target

 Global reach, higher impact
Global reach, higher impact