Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(1):188-203. doi:10.7150/ijbs.50810 This issue Cite
Research Paper
PRPF6 promotes androgen receptor/androgen receptor-variant 7 actions in castration-resistant prostate cancer cells
1. Department of Cell Biology, Key laboratory of Cell Biology, Ministry of Public Health, and Key laboratory of Medical Cell Biology, Ministry of Education, School of Life Sciences, China Medical University, Shenyang City, Liaoning Province110122, China.
2. Department of Molecular Oncology, Liao Ning Tumor Hospital, Shenyang, Liaoning 110042, China.
3. Department of Breast Surgery, the First Affiliated Hospital of China Medical University, Shenyang City 110001, Liaoning Province, China.
Abstract
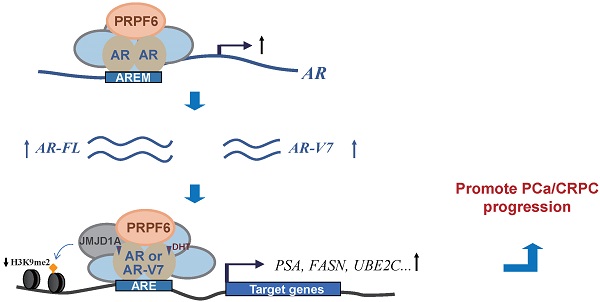
Androgen receptor (AR) and its variants play vital roles in development and progression of prostate cancer. To clarify the mechanisms involved in the enhancement of their actions would be crucial for understanding the process in prostate cancer and castration-resistant prostate cancer transformation. Here, we provided the evidence to show that pre-mRNA processing factor 6 (PRPF6) acts as a key regulator for action of both AR full length (AR-FL) and AR variant 7 (AR-V7), thereby participating in the enhancement of AR-FL and AR-V7-induced transactivation in prostate cancer. In addition, PRPF6 is recruited to cis-regulatory elements in AR target genes and associates with JMJD1A to enhance AR-induced transactivation. PRPF6 also promotes expression of AR-FL and AR-V7. Moreover, PRPF6 depletion reduces tumor growth in prostate cancer-derived cell lines and results in significant suppression of xenograft tumors even under castration condition in mouse model. Furthermore, PRPF6 is obviously highly expressed in human prostate cancer samples. Collectively, our results suggest PRPF6 is involved in enhancement of oncogenic AR signaling, which support a previously unknown role of PRPF6 during progression of prostate cancer and castration-resistant prostate cancers.
Keywords: androgen receptor, pre-mRNA processing factor 6, alternative splicing, transcriptional regulation, prostate cancer

 Global reach, higher impact
Global reach, higher impact